Q: The triphenylmethyl cation is so stable that a salt such as triphenylmethyl chloride can be isolated…
A: The species in which carbon carries a positive charge is called as carbocation. Carbocations are…
Q: Give the major organic product(s) for each of the following reactions
A: In the first reaction reduction of amide takes place.
Q: How many of the following will undergo a favourable rearrangement to a lower energy carbocation?
A: If migration of atom or group produce more stable carbocation then that rearrangement will lower the…
Q: In the proton transfer step of a Friedel-Crafts alkylation reaction, where does the proton go? O To…
A: Given proton transfer step of Friedel-crafts alkylation To be determined Where does the proton go…
Q: Predict the starting alkyl chloride that would produce the p shown in the SN1 reaction. Assume no…
A: SN1 reaction: The SN1 reaction is a substitution reaction in organic chemistry. "SN" stands for…
Q: Why is there a difference in racemization for the two reactions even though they both include the…
A: Bromine is an excellent leaving group compared to hydroxyl group. Hence, in case of 2-bromobutane,…
Q: Acid-catalyzed transesterification and Fischer esterification take place by nearly identical…
A: Concept introduction: Transesterification involves the interchange of alkoxy groups when ester is…
Q: If the following compound was reacted with Bromine and light under the same reaction conditions as…
A: The rate of the reaction can be described as the time taken by the reactant of certain concentration…
Q: Except for the Tollens test, basic aqueous conditions are generally avoided with sugarsbecause they…
A: The base catalysed epimerization of glucose takes place as follows:
Q: Decriegsing onder of LG ?
A: A leaving group is a nucleophile acting in reverse; it accepts a lone pair as the bond between it…
Q: Write the letters that correspond to the starting material ______ and the product ______ for the…
A: We have to determine the starting material and the product
Q: Which will react in methanol at the highest rate when reacted with CN-? How will the reaction rate…
A: Methanol is a polar protic solvent, and acetone is a polar aprotic solvent.
Q: Which TWO allylic carbocations would give kinetic products?
A: Given compound is: Which double bond reacts faster with HBr and form the product?
Q: Consider the following electrocyclic ring closure. Does the product form by a conrotatory or…
A: Woodward - Hoffmann rules are the set of rules used to vindicate or predict certain aspects of the…
Q: Give the major organic product(s) for the following reaction
A: Alkynes are those hydrocarbons that contain a carbon-carbon triple bond. Alkynes undergo addition…
Q: Which of the following is the rate- determining step in the electrophilic aromatic substitution of…
A: In Electrophilic aromatic substitution of benzene an electrophile replaces the hydrogen atom present…
Q: Which of the following is compatible with a Friedel-Crafts reaction?
A: Friedel-Craft alkylation reaction: When the benzene reacts with an alkyl halide in presence of Lewis…
Q: A) Label the α-carbon and β-carbons in each of the following molecules. Draw the β-elimination…
A: The carbon atom to which the bromine atom is attached is α Carbon. The carbon lying next to α…
Q: Indane can undergo free-radical chlorination at any of the alkyl positions onthe aliphatic ring.…
A: The possible monochlorinated products from the given free-radical chlorination of indane are shown…
Q: Which of the following reacts fastest with methanol by the SN1 mechanism?
A: Tertiary alkyl bromide is the alkyl bromide in which carbon atom bearing bromine atom is attached to…
Q: Which of the following is a proper and reasonable resonance hybrid structure of benzaldehyde that…
A: When a molecule (or) ion can not be represented by single lewis dot structure and more than one…
Q: Which one of the following reactions shows the wrong reaction conditions to give the product shown?
A: The reactions involving organic chemical compounds are called organic reactions. Electrophilic and…
Q: One of the two diastereomers shown below reacts with potassium tertbutoxide significantly faster…
A:
Q: Identify the organohalide which would give the two Friedel-Crafts alkylation products shown below.
A:
Q: Cyanohydrin formation is useful because of the further chemistry that can be carried out on
A: C=O group is called carbonyl group Aldehyde and ketone both are combinedly called carbonyl…
Q: Give complete mechanism of the following reactions, thank you!
A: Substitution reaction- The replacement one atom or group of atom from reactant species with another…
Q: Name the kind of sigmatropic rearrangement that occurs in the following reaction.
A: The kind of sigmatropic rearrangement of the following reaction has to be given.
Q: The creation of very large ring systems is often difficult and challenging. The following…
A:
Q: Which is the correct order of reactivity for the following alkyl bromides in an El reaction?
A: Given : structure of alkyl halide. Reactivity of alkyl halide is directly proportional to the…
Q: How many elimination product(s) is /are possible in the reaction of compound…
A: Here, we have to find the number of elimination products that are possible in the reaction of…
Q: If toluene and vinyl chloride have Friedel - Crafts alkylation, what major product will be?
A: Friedel-Crafts Alkylation reaction is the reaction of toulene with haloalkanes in presence of…
Q: ELOH, A Br
A: Aromatic compounds are the compounds that satisfy the following conditions: 1. The compound must be…
Q: Which term best describes the stereochemical relationship of the products of the reaction shown…
A: The stereochemical relationship of the products of the reaction shown below
Q: Draw the major organic product generated in the reaction below. Pay particular attention to regio-…
A: Given that : We have to draw the major product for the following reaction :
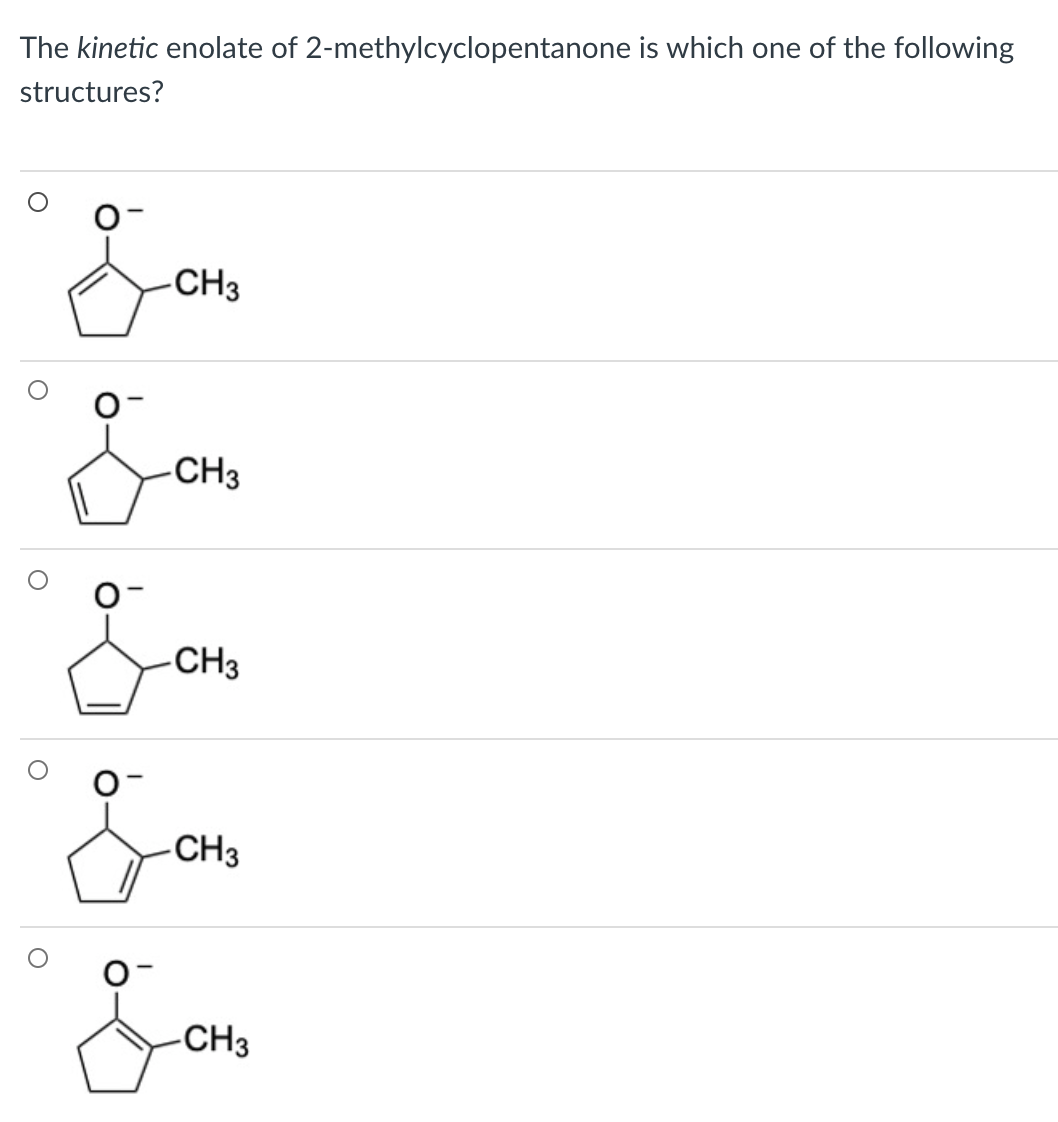
Q: The kinetic enolate of 2-methylcyclopentanone is which one of the following structures? -CH3 -CH3…
A: When 2-methylcyclopentanone is exposed to basic conditions, enolate formation takes place on either…
Q: The following transformation is
A: The convertion of alkene to alkane can be done by hydrogenation of carbon carbon double bonds. The…
Q: Indane can undergo free-radical chlorination at any of the alkyl positions onthe aliphatic ring.Once…
A:
Q: Indane can undergo free-radical chlorination at any of the alkyl positions onthe aliphatic ring.…
A: Indane is an organic compound which two part, one part is an aromatic ring and other part of the…
Q: Which of the following is the product of the reaction sequence below?
A:
Q: her question will stve this respo on 18 Which aly chioride reacts fastest in S2 reactions?
A: The SN2 reacction is most favored in those alkyl halides which forms less stable carbocation.…
Q: In the Friedel-Crafts alkylation of benzene, dialkylation is often a significant by-product. In the…
A:
Q: Intramolecular Diels-Alder reactions are possible when a substrate contains both a 1,3-diene and a…
A:
Q: Consider the following electrocyclic ring closure. Does the product form by a conrotatory or…
A: The starting material has three conjugated double bonds and which proceeds electrocyclic ring…
Q: What will be the ozonolysis product of the given reaction below
A: Given compound is diketone , it is derivied by cycloalkene upon ozonolysis Oxidation of alkenes…
Q: Give detailed mechanism of following reactions?
A: First step is conversion of ethyl 3-oxobutanoate (ester) to 3-oxobutanoic acid (carboxylic acid)…
Q: What is the major intermediate in the Friedel-Crafts alkylation? Why does it imply that…
A: friedel craft alkylation is electrophilic aromatic substitution reaction in which alkyl group is…
Q: Give
A: We have to give product of the given reaction
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

- From the reaction of ethyl cyclohexyl ketone with a base below. Identify the kinetic enolate and thermodynamic enolate respectively.A)There are two plausible enolates that can arise when LDA is used as the base for the ketone shown. In each statement circle what best describes the enolate? The enolate is formed under either a) irreversible or b) reversible conditions The favored is either the a) more stable or b) less stable enolate The favored enolate is either the a) kinetic or the b) thermodynamic enolate B) What is the major enolate? (structure)What type of sigmatropic rearrangement is illustrated in each reaction?
- Which organo-chloride below is most reactive in SN1 reactions?Which of the synthetic procedures shown in Image 31 would carry out the following transformation? A. b B. c C. a D. dwhich newman projection displays the proper antiperiplanar orientation for an E2 elimination of the reaction sequence below
- Rank the following E2 reactions in order of increasing rate. Show the final product.Provide the mechansim using curved arrows of the reaction of p-t-butyl phenol treated with acetic anhydride in AlCl3. Include resonance stabilized intermediates and if more than one product is formed, label them as major, minor, etc. thank you for the helpWhich condition or sequence is ideal for converting ethyne into the epoxide shown below? A. 1. D2 (1 equivalent)/Pd-C 2. H3CC(O)OOH B. 1. D2/Lindlar 2. H3CC(O)OOH C. 1. H3CC(O)OOH 2. D2/ Pd-C D. 1. Na/ND3 2. H3CC(O)OOH
- When allyl bromide is refluxed with magnesium metal in ether solvent, the product formed is 1,5-hexadiene. (C6H10). What is the curved arrow mechanism for this reaction?1. Draw a reasonable arrow-pushing mechanism for the transformation shown along. 2. Identify nucleophiles and electrophiles 3. Name any type of reactions taking place like E2 or E1 4. Account for any regio- or stereoselectivityPlease label all the steps and reagents with their corresponding letter. Thanks!

