The creation of very large ring systems is often difficult and challenging. The following macrocyclic ring was created using one of the coupling reactions described in this chap- ter. Which one was used? What was the starting material? Он О НО
The creation of very large ring systems is often difficult and challenging. The following macrocyclic ring was created using one of the coupling reactions described in this chap- ter. Which one was used? What was the starting material? Он О НО
Organic Chemistry
8th Edition
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Chapter24: Catalytic Carbon-carbon Bond Formation
Section: Chapter Questions
Problem 24.30P
Related questions
Question
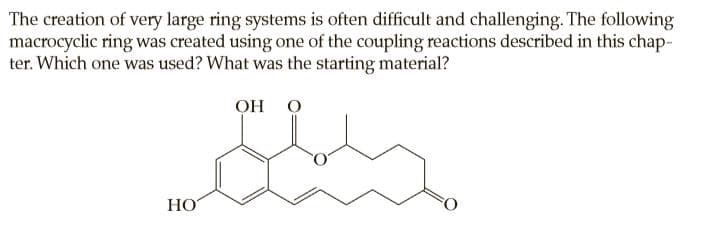
Transcribed Image Text:The creation of very large ring systems is often difficult and challenging. The following
macrocyclic ring was created using one of the coupling reactions described in this chap-
ter. Which one was used? What was the starting material?
Он О
НО
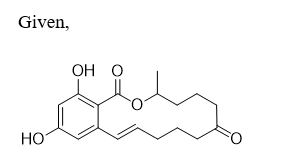
Expert Solution
The starting material has to be given.
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Recommended textbooks for you

Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning

Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning