The oxyacids containing oxygen (O) and phosphorous (P) are named differently than the oxyanions on which they are based. You have just learned that in general -ite endings are changed to -ous while -ate endings are changed to -ic when naming the acid. However, for the acids of oxyanions based on phosphorus, the root also changes. The roots for the oxyanions and oxyacids of phosphorus are shown below. • oxyanion root: phosph oxyacid root: phosphor Although the exception exists for the root, the prefixes and suffixes of these oxyacids still follow the conventions for naming oxyacids. Using the table of oxyanions given in the transition before Part B and the roots shown above, give the name of the oxyacid H3PO4 below. Spell out the full name of the acid.
The oxyacids containing oxygen (O) and phosphorous (P) are named differently than the oxyanions on which they are based. You have just learned that in general -ite endings are changed to -ous while -ate endings are changed to -ic when naming the acid. However, for the acids of oxyanions based on phosphorus, the root also changes. The roots for the oxyanions and oxyacids of phosphorus are shown below. • oxyanion root: phosph oxyacid root: phosphor Although the exception exists for the root, the prefixes and suffixes of these oxyacids still follow the conventions for naming oxyacids. Using the table of oxyanions given in the transition before Part B and the roots shown above, give the name of the oxyacid H3PO4 below. Spell out the full name of the acid.
ChapterU1: Alchemy: Matter, Atomic Structure, And Bonding
Section: Chapter Questions
Problem 8STP
Related questions
Question
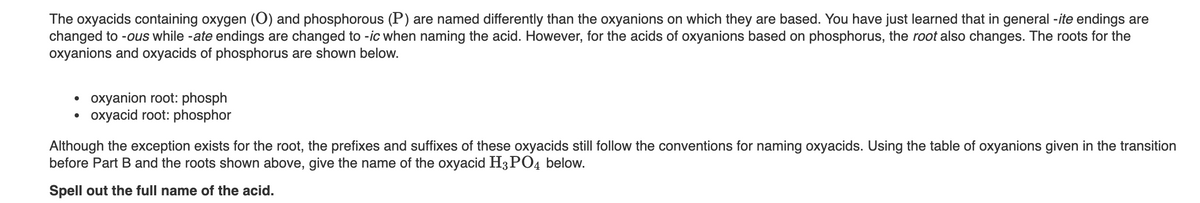
Transcribed Image Text:The oxyacids containing oxygen (O) and phosphorous (P) are named differently than the oxyanions on which they are based. You have just learned that in general -ite endings are
changed to -ous while -ate endings are changed to -ic when naming the acid. However, for the acids of oxyanions based on phosphorus, the root also changes. The roots for the
oxyanions and oxyacids of phosphorus are shown below.
oxyanion root: phosph
oxyacid root: phosphor
Although the exception exists for the root, the prefixes and suffixes of these oxyacids still follow the conventions for naming oxyacids. Using the table of oxyanions given in the transition
before Part B and the roots shown above, give the name of the oxyacid H3PO4 below.
Spell out the full name of the acid.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning

General, Organic, and Biological Chemistry
Chemistry
ISBN:
9781285853918
Author:
H. Stephen Stoker
Publisher:
Cengage Learning


Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning

General, Organic, and Biological Chemistry
Chemistry
ISBN:
9781285853918
Author:
H. Stephen Stoker
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781285199030
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning