The photoelectron spectra of nitrogen and oxygen's 1s electrons are shown below. Which statement best explains the reason that nitrogen's peak appears to the right of oxygen's? * Relative Number of Electrons Relative Number of Electrons Nitrogen (N) 700 + 600 Oxygen (0) 700 600 500 Binding Energy (eV) 500 Binding Energy (CV) 400 400 O Nitrogen atoms have a half-filled p sublevels. Nitrogen's atoms have a smaller nuclear charge than oxygen's atoms. Electrons in the p sublevel of oxygen provide more shielding than electrons in the p sublevel of nitrogen O There are more electron-electron repulsions in oxygen than nitrogen. 300 300
The photoelectron spectra of nitrogen and oxygen's 1s electrons are shown below. Which statement best explains the reason that nitrogen's peak appears to the right of oxygen's? * Relative Number of Electrons Relative Number of Electrons Nitrogen (N) 700 + 600 Oxygen (0) 700 600 500 Binding Energy (eV) 500 Binding Energy (CV) 400 400 O Nitrogen atoms have a half-filled p sublevels. Nitrogen's atoms have a smaller nuclear charge than oxygen's atoms. Electrons in the p sublevel of oxygen provide more shielding than electrons in the p sublevel of nitrogen O There are more electron-electron repulsions in oxygen than nitrogen. 300 300
Introductory Chemistry: A Foundation
9th Edition
ISBN:9781337399425
Author:Steven S. Zumdahl, Donald J. DeCoste
Publisher:Steven S. Zumdahl, Donald J. DeCoste
Chapter11: Modern Atomic Theory
Section: Chapter Questions
Problem 30QAP: Consider the following representation of a set of p orbitals for an atom: mg...
Related questions
Question
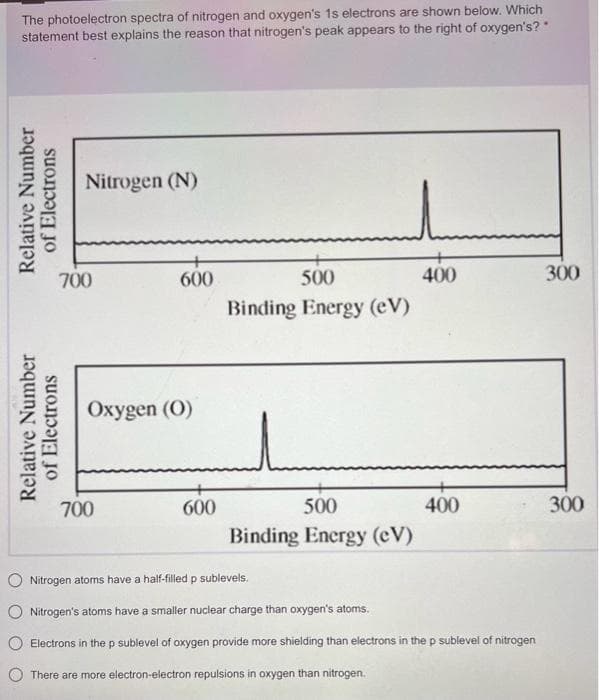
Transcribed Image Text:The photoelectron spectra of nitrogen and oxygen's 1s electrons are shown below. Which
statement best explains the reason that nitrogen's peak appears to the right of oxygen's?"
Relative Number
of Electrons
Relative Number
of Electrons
Nitrogen (N)
700
600
Oxygen (0)
700
600
d
500
Binding Energy (eV)
500
Binding Energy (CV)
400
400
Nitrogen atoms have a half-filled p sublevels.
Nitrogen's atoms have a smaller nuclear charge than oxygen's atoms.
Electrons in the p sublevel of oxygen provide more shielding than electrons in the p sublevel of nitrogen
There are more electron-electron repulsions in oxygen than nitrogen.
300
300
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning
