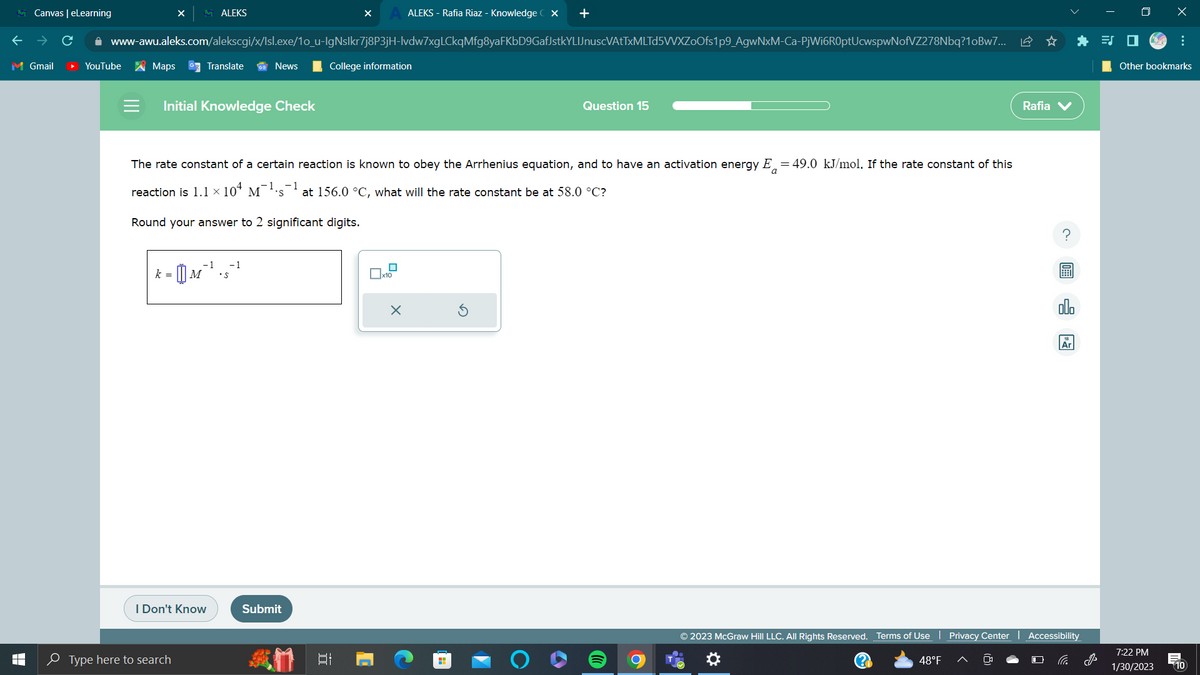
The rate constant of a certain reaction i known to obey the Arrhenius equation, and to have an activation energy E = 49.0 kJ/mol. If the rate constant of this reaction is 1.1 x 104 M¹s¹ at 156.0 °C, what will the rate constant be at 58.0 °C? Round your answer to 2 significant digits. -1 | k = [[M²¹ ³5² ³ X S ? olo
The rate constant of a certain reaction i known to obey the Arrhenius equation, and to have an activation energy E = 49.0 kJ/mol. If the rate constant of this reaction is 1.1 x 104 M¹s¹ at 156.0 °C, what will the rate constant be at 58.0 °C? Round your answer to 2 significant digits. -1 | k = [[M²¹ ³5² ³ X S ? olo
Principles of Instrumental Analysis
7th Edition
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Chapter23: Potentiometry
Section: Chapter Questions
Problem 23.12QAP: What arc the advantages of microfabricated ISEs? Describe typical applications of this type of...
Related questions
Question
Hey,
Can you please help me to solve this problem?
Transcribed Image Text:Canvas | eLearning
M Gmail
T
LI
YouTube
ALEKS
www-awu.aleks.com/alekscgi/x/Isl.exe/1o_u-lgNslkr7j8P3jH-lvdw7xgLCkqMfg8yaFKbD9GafJstkYLIJnuscVAtTxMLTd5VVXZoOfs1p9_AgwNxM-Ca-PjWi6R0ptUcwspwNofVZ278Nbq?1oBw7...
Maps GTranslate
Initial Knowledge Check
-1 -1
k = M •S
I Don't Know
Type here to search
News
The rate constant of a certain reaction is known to obey the Arrhenius equation, and to have an activation energy E = 49.0 kJ/mol. If the rate constant of this
−1
reaction is 1.1 × 10ª M¯¯¹·s¯ at 156.0 °C, what will the rate constant be at 58.0 °C?
Round your answer to 2 significant digits.
X
Submit
College information
I
ALEKS - Rafia Riaz - Knowledge CX +
x10
X
Question 15
H
© 2023 McGraw Hill LLC. All Rights Reserved. Terms of Use | Privacy Center
48°F
Rafia V
Ar
Accessibility
x
Other bookmarks
7:22 PM
1/30/2023
10
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning

Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning