The solubilities of three salts in water are shown in 1000 Pb(NO,), the graph. 900 800 K,Cr.O, For each salt, determine how much will remain undissolved 700 if 500 g is mixed into a liter of pure water at 40 °C. One 600 - significant figure is sufficient in each answer. 500 KCI 400 - Pb(NO,);: g 300 200 100 - KCl: g 10 20 30 40 50 60 70 80 90 100 Temperature (°C) K,Cr,0,: g Solubility (g/L)
The solubilities of three salts in water are shown in 1000 Pb(NO,), the graph. 900 800 K,Cr.O, For each salt, determine how much will remain undissolved 700 if 500 g is mixed into a liter of pure water at 40 °C. One 600 - significant figure is sufficient in each answer. 500 KCI 400 - Pb(NO,);: g 300 200 100 - KCl: g 10 20 30 40 50 60 70 80 90 100 Temperature (°C) K,Cr,0,: g Solubility (g/L)
Principles of Modern Chemistry
8th Edition
ISBN:9781305079113
Author:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Chapter16: Solubility And Precipitation Equilibria
Section: Chapter Questions
Problem 3P
Related questions
Question
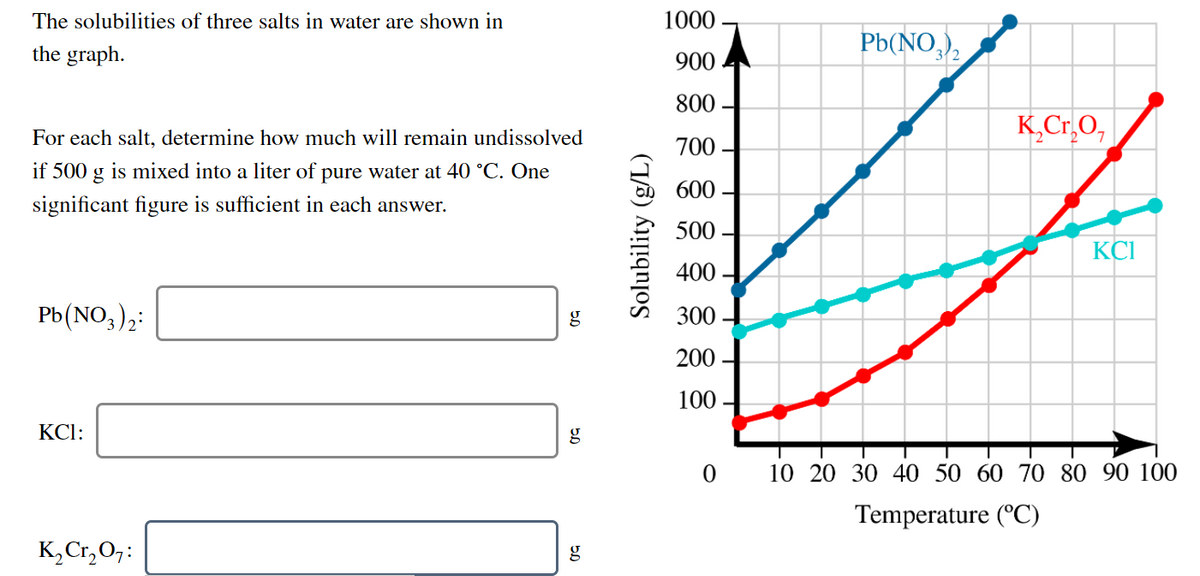
Transcribed Image Text:The solubilities of three salts in water are shown in
1000
Pb(NO,),
the graph.
900
800
K,Cr.O,
For each salt, determine how much will remain undissolved
700
if 500 g is mixed into a liter of pure water at 40 °C. One
600
significant figure is sufficient in each answer.
500
KCI
400
Pb(NO,),:
300
200
100
KCl:
g
10 20 30 40 50 60 70 80 90 100
Temperature (°C)
K,Cr, 0,:
g
Solubility (g/L)
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 8 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning


Chemistry: Matter and Change
Chemistry
ISBN:
9780078746376
Author:
Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:
Glencoe/McGraw-Hill School Pub Co

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning


Chemistry: Matter and Change
Chemistry
ISBN:
9780078746376
Author:
Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:
Glencoe/McGraw-Hill School Pub Co

Chemical Principles in the Laboratory
Chemistry
ISBN:
9781305264434
Author:
Emil Slowinski, Wayne C. Wolsey, Robert Rossi
Publisher:
Brooks Cole

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:
9781305446021
Author:
Lampman
Publisher:
CENGAGE LEARNING - CONSIGNMENT