The solubilities of three salts in water are shown in 1000 Pb(NO,)2 the graph. 900 800 For each salt, determine how much will remain undissolved K,Cr,O, if 500 g is mixed into a liter of pure water at 40 °C. One 700 - significant figure is sufficient in each answer. 600 500 - КСІ Pb(NO,),: 400 300 KCI: 200 100 K,Cr,0;: 10 20 30 40 50 60 70 80 90 100 Temperature (°C) Solubility (g/L)
The solubilities of three salts in water are shown in 1000 Pb(NO,)2 the graph. 900 800 For each salt, determine how much will remain undissolved K,Cr,O, if 500 g is mixed into a liter of pure water at 40 °C. One 700 - significant figure is sufficient in each answer. 600 500 - КСІ Pb(NO,),: 400 300 KCI: 200 100 K,Cr,0;: 10 20 30 40 50 60 70 80 90 100 Temperature (°C) Solubility (g/L)
Chapter4: Calculations Used In Analytical Chemistry
Section: Chapter Questions
Problem 4.24QAP
Related questions
Question
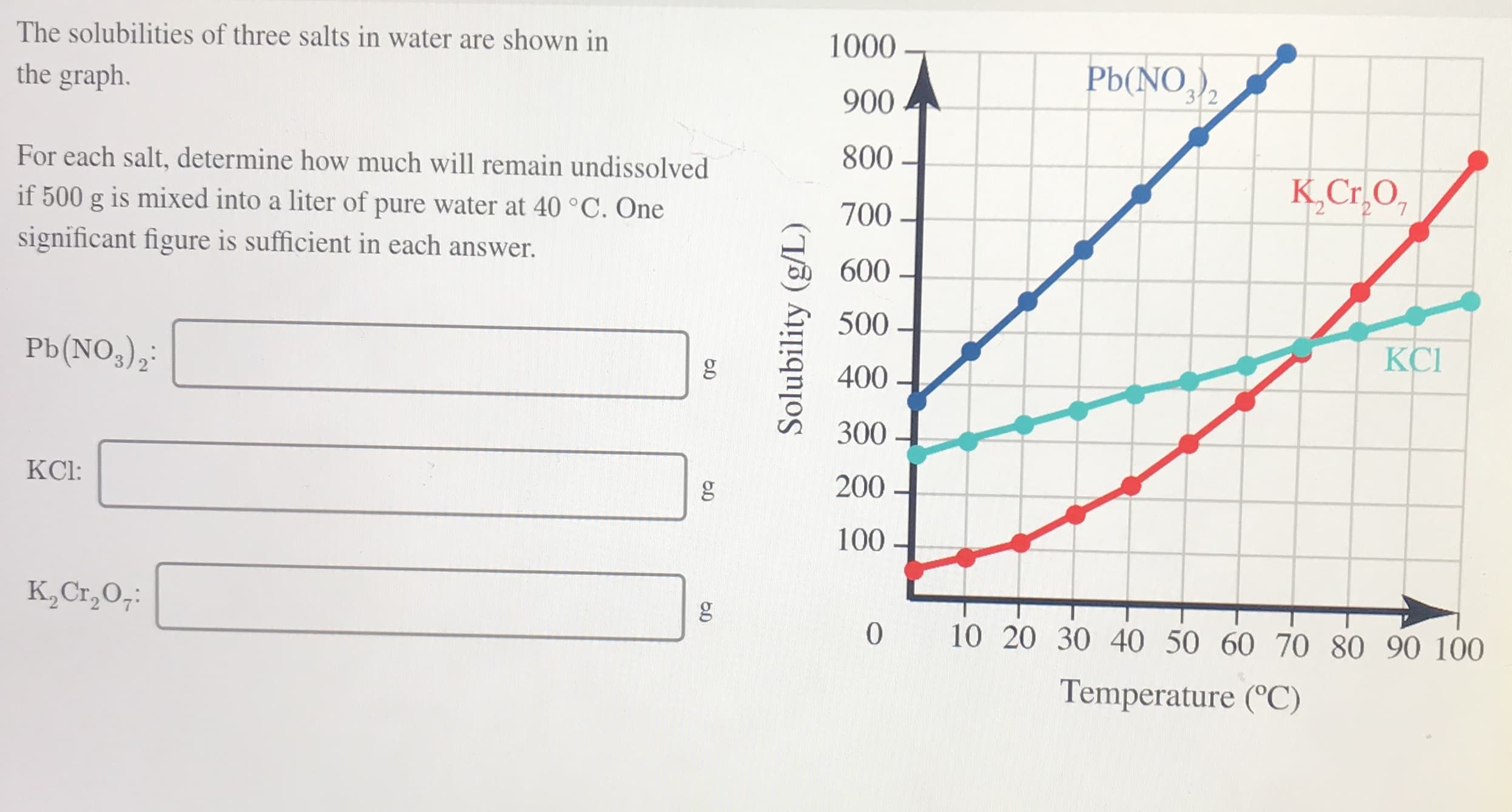
Transcribed Image Text:The solubilities of three salts in water are shown in
1000
Pb(NO,)2
the graph.
900
800
For each salt, determine how much will remain undissolved
K,Cr,O,
if 500 g is mixed into a liter of pure water at 40 °C. One
700 -
significant figure is sufficient in each answer.
600
500 -
КСІ
Pb(NO,),:
400
300
KCI:
200
100
K,Cr,0;:
10 20 30 40 50 60 70 80 90 100
Temperature (°C)
Solubility (g/L)
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 8 steps with 4 images

Recommended textbooks for you


Chemical Principles in the Laboratory
Chemistry
ISBN:
9781305264434
Author:
Emil Slowinski, Wayne C. Wolsey, Robert Rossi
Publisher:
Brooks Cole


Chemical Principles in the Laboratory
Chemistry
ISBN:
9781305264434
Author:
Emil Slowinski, Wayne C. Wolsey, Robert Rossi
Publisher:
Brooks Cole