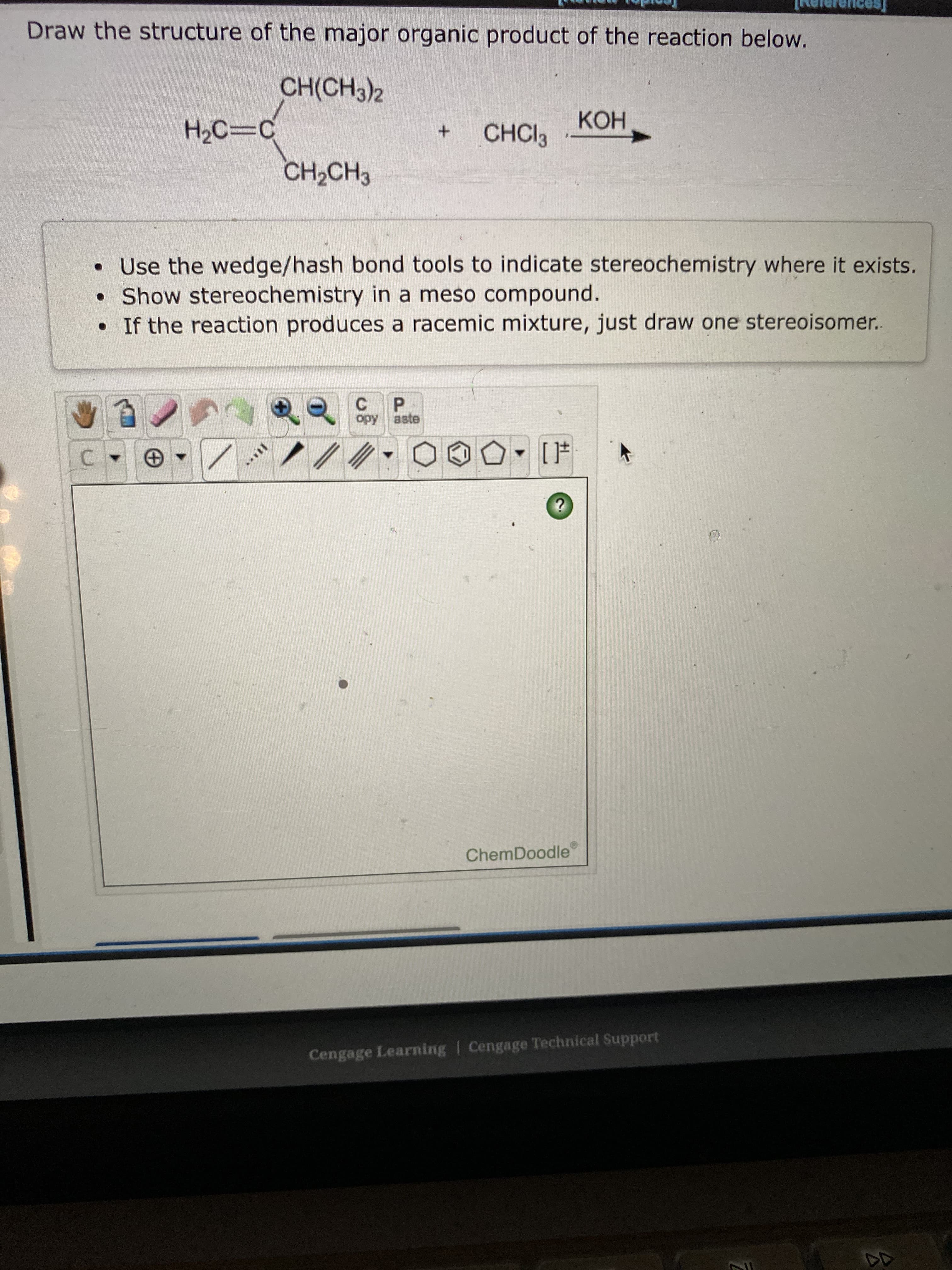
the stru e of the major organic product of the reaction below. CH(CH3)2 КОН H2C=C CHCI3 +. CH2CH3 • Use the wedge/hash bond tools to indicate stereochemistry where it
Q: In the analysis of 1.500 g sample of feldspar, a mixture of the chlorides of sodium and potassium ch...
A: Given: Mass of sample = 1.500 g. Mass of NaCl and KCl mixture formed from sample = 0.3450 g. And the...
Q: Calculate the concentration of iodide ions ( I− ) in a saturated solution of lead iodide at 298 K. (...
A: Given, Ksp of PbI2 = 1.39 × 10-8 PbI2(s) ⇌ Pb2+ (aq) + 2 I-(aq) initial:. - change: ...
Q: A weather balloon calibrated at 0.00 °C to have a volume of 20.0 L has what volume in L at -14.1 °C ...
A: Given ; values of volume and temperature.
Q: What is the molality when a 0.75 mol is dissolved in 2.50 kg of solvent?
A: To solve this question, use the molality formula and calculate molality for given solution.
Q: The equilibrium constant Kp for the interconversion of phosphorous pentachloride and phosphorous tri...
A:
Q: A compound Y is to be determined by UV-visible spectrophotometry. A calibration curve is constructed...
A: The data given is, Concentration (ppm) Absorbance 0.20 0.870 0.40 0.840 0.60 0.800 0.80 0...
Q: At what temperature will 2.00 mol of He fill a 6.00-L container at standard pressure? Enter your ans...
A:
Q: What is the pH at the equivalence point for the titration of 50.0 mL 0.20 M nitrous acid by 0.20 M N...
A:
Q: If 100 ml of a solution contain a mixture of 0.158 g KMnO4 and 0.490 g of K2Cr2O7, a. What is th...
A: Mass of KMnO4 = 0.158 g Mass of K2Cr2O7 = 0.490 g Volume of solution = 100 mL Normality of FeSO4 = 0...
Q: What name is given to the chemical species that changes colour when its pH changes? O Acid Base Indi...
A:
Q: How many grams of O2 gas occupy 19.0 L at STP? Enter your answer in the provided box.
A:
Q: Calculate the average kinetic energy in (kj/mol)and root mean square speeding (m/s) of argon gas at ...
A: Given ; temperature
Q: (Q7) If the initial concentration of the weak base ammonia in a solution is 0.247 M and the solution...
A:
Q: Predict the products of the reaction below. That is, complete the right-hand side of the chemical eq...
A: HClO4 is a strong acid and NaOH is a strong base. When they react each other salt and water formed a...
Q: What mass of aluminum metal can be produced per hour in the electrolysis of a molten aluminum salt b...
A:
Q: Predict the products of the reaction below. That is, complete the right-hand side of the chemical eq...
A: HCl is a strong acid and Ca(OH)2 is a strong base. When they react each other salt and wayer form ...
Q: Give a clear handwritten answer...Find out pH Of Hcl ...given Hcl solution (H+)=0.03?
A:
Q: Given: H2S(aq) + H20(1) HS (aq) + H30*(aq) K = 1.00x10-7 H2S(aq) + 2H2O(1) = s²"(aq) + 2H30*(aq) K =...
A:
Q: 8Ca → 3. 65 Sc 65 4. Be + He C + -> P + H 31 15 32 15 5. 16S + Hе 35 6. 1ci +
A: Dear student, As you have asked multiple question but according to guidelines we will solve first...
Q: The equilibrium constant, Kc, for the following reaction is 1.96x10-2 at 732 K. 2HI(g) H2(g) + I2(g)...
A: First we would make changes to given reaction to get reaction for which Kc is to be determined . For...
Q: Problem 4 IR Spectrum (liquid film) 4000 3000 2000 1600 1200 800 V (cm1) Mass Spectrum 107/109 No si...
A:
Q: Using Hess's Law tó calculate net reaction enthalpy 1/5 The lead-acid storage battery is the oldest ...
A:
Q: Write the equilibrium constant for this reaction in terms of the equilibrium constants, K1 and K2, f...
A: 2S(s) + 3 O2(g) ---> 2SO3(g) K1 SO2(g) + 1/2(O2) ---> SO3(g) K2
Q: A student determines the value of the equilibrium constant to be 4.16x1013 for the following reactio...
A: Given-> 4HCl(g) + O2(g) ---> 2H2O(g) + 2Cl2(g)
Q: The fluorescence of the CQDS when treated with Fe3+ ions does not fluoresce as brightly as the CQDS ...
A:
Q: Select the oxidizing and reducing agent for the reaction represented by the following line notation:...
A: The addition of electrons or decreases in the oxidation state is known as a reduction reaction and t...
Q: Use a Jablonski diagram or potential energy curves to show the theoretical difference between fluore...
A: Diagram that illustrates the electronic states of a molecule and the transitions between them is sho...
Q: Predict the products and balance the following reaction: Ag2SO4 (aq) + NaCl(aq)
A:
Q: A gas at STP occupies 16 L of space. If the pressure changes to 2.6 atm and the temperature increase...
A: Given that Initial volume (V1) = 16L Final pressure (p2) = 2.6 atm Final temperature (T2) = 195°C Ne...
Q: Draw and name all possible skeletal isomers of compounds having the molecular formula C6H14 . All co...
A:
Q: Draw the major product of this reaction. Ignore any inorganic byproducts HBr (1 equiv) %3D %3D %3D B...
A:
Q: What is the % of water in magnesium sulfate heptahydrate
A:
Q: Calculate AG° (in kJ) for the following reaction: 31, (s) + 2Fe(s) →2FE** (aq) +6I¯(aq) 232 O -111 O...
A:
Q: • Part A A lead-acid battery uses a redox reaction in which lead(0) and lead(IV) are both converted ...
A: The reaction given is, => Given: Time passed = 5.00 hours = 5 × 60 × 60 = 18000 s ...
Q: Answer in KJ/mol C,H,(g) + H, (g) =2CH,(g) standard conditions are 298 K and a partial pressure of I...
A:
Q: When KNO3 is dissolved in water, the resulting solution is significantly colder than the water was o...
A:
Q: Part A d for the following second-order reaction: E(9) Use an Arrhenius plot to determine the activa...
A:
Q: The equilibrium constant, Kc, for the following reaction is 6.33x10 5 at 536 K. NH4CI(s) NH3(g) + HC...
A: Given :- NH4Cl(s) <----> NH3(g) + HCl(g) Kc = 6.33 × 10-5 To calculate :- Kc for NH3(g) ...
Q: Which of the following conditions would be consistent to produce a buffer? Strong acid and a strong ...
A: Here, we have to find the condition that is consistent to produce a buffer.
Q: Balance the following chemical equation (if necessary): C,H(g) + O,(9) → H,O(g) + CO,(g) 10
A: Given the unbalanced equation: C5H10(g) + O2(g) → H2O(g) + CO2(g) We have to balance this equation. ...
Q: The equilibrium constant, Kc, for the following reaction is 1.80x102 at 698 K. 2HI(g) H2(g) + I2(g) ...
A: Since you have asked multiple questions, we will solve the first question for you. If you want any s...
Q: If the balloon described in Question 10.46 is released into the air and rises to an altitude of 10,0...
A: Given Data : 1. pressure = 523 Torr 2. Temperature = 7.50 ºC = (7.50+273.15)K = ...
Q: A high-pressure weather system occurs when atmospheric pressure increases above ambient air pressure...
A:
Q: Disaccharides are joined by glycosidic bonds formed between the anomeric carbon on one monosaccharid...
A: The bond between two sugar molecule is known as glycosidic bond. This bond is formed between 2 OH g...
Q: How does how the molecular identity of the element change in space and/or time? Be sure to make the ...
A: A name that uniquely identifies a compound, molecule or substance is known as chemical identity. In ...
Q: How is Polycarbonate made? What is the structure of the Monomer? What is the structure of the polyme...
A: Polycarbonate is a type of polyester made by condensation of carbonic acid or carbonyl chloride and...
Q: The equilibrium constant, Kp, for the following reaction is 0.215 at 673 K. Calculate K. for this re...
A:
Q: Using the following equilibria and Kc values: SO3(3) + NO(g) 5 SO2(g) + NO2(g) 2 SO3(g) + N2(g) S2 S...
A: To find out the Kc value for resultant reaction, do the appropriate changes in the reaction and solv...
Q: A student ran the following reaction in the laboratory at 284 K: 2NO(g) + Br2(g) = 2NOBr(g) When she...
A: Given reaction: 2NO(g) + Br2(g) ⇌ 2NOBr(g) The initial number of moles of NO(g) = 0.184 moles The in...
Q: 1.)If 5.00g of iron (III) chloride hexahydrate us completely dehydrated, he many grams of anhydrous ...
A:
Step by step
Solved in 2 steps with 1 images

- i. Fill in the missing starting materials, products, or reagents as necessary. ii. Label each transformation as SN1, SN2, or acid/base. iii. Indicate if the product is racemic or a single enantiomerEthers: Reaction of 1-methoxy-2- methylpropane and HI, heat. Draw the organic product or products formed when the structure shown below is heated with HI. If no reaction occurs, draw the starting material. Interactive 3D display modeDraw mechanism arrows and then predict the major products and stereochemistry. Also include the labels of thermodynamic and kinetic no stick structure don't understand 3,4-dimethylhex-3-ene with BH3. After reaction is complete, add H2O2, –OH, and H2O
- These are synthesis questions. You need to show how the starting material can be converted into the product(s) shown. You may use any reactions we have learned. Show all the reagents you need. Show each molecule synthesized along the way and be sure to pay attention to the regiochemistry and stereochemistry preferences for each reaction. If a racemic molecule is made along the way, you need to draw both enantiomers and label the mixture as "racemic".Show arrows, mechanism and product of TiCl4 + CH3-O-CH3.Fill in the missing starting material, reaction conditions, or products for each reaction shown below. Take care to indicate if two sets of reagents have been added sequentially (using 1. ___ and 2.___ above the arrow), and also to show all MAJOR products and indicate stereochemistry. In cases where a pair of enantiomers are produced or stereochemistry is not controlled you can write “+ enantiomer” or use squiggly lines, as appropriate.
- Determine the product of the reactions below. Show stereochemistry- For the dehydration shown, use curved arrows to show the formation of the carbocation intermediate in the presence of sulfuric acid H2SO4H2SO4, then draw the structure of the major product of the elimination. (Picture 1): Step 1: Use curved arrows to complete the protonation mechanism of the alcohol. Step 2: Use a curved arrow to show the formation of the carbocation intermediate. Note: HSO−4HSO4− is formed from step 1, but not shown. (Picture 2): Step 3: Use a curved arrow to show the carbocation rearrangement. Step 4: Draw curved arrows to show the elimination to form the major product. Water is added as a base. - Draw the major product of the elimination. H3O+H3O+ has been pre‑drawn for your convenience.The synthesis above can be performed with some combination of the reagents listed below. Give the necessary reagents in the correct order. Be sure to consider stereochemistry and regiochemistry.A. NaOHB. mCPBAC. OsO4, tBuOOHD. NaHE. H2, LindlarF. CH3CH2CH2BrG. CH3CH2ClH. NaNH2I. H3O+J. Na, NH3(l)
- Name the alkene below.Use only E/Z designators to indicate stereochemistry.Draw eugenol as a line structure. The double bonds of the benzene ring are fairly unreactive unless you use special catalysts. But the double bond on the side group is reactive. Draw the mechanism of eugenol reacting with HBr to form the two possible carbocation intermediates. Put your intermediates into square brackets. Label each carbocation as a methyl carbon or 1°, 2°, or 3°. Then circle a more stable carbocation if one is more stable. Can you guess what the final product is?How many stereoisomers of 2-methyl-heptan-4-ol could form? How many would you expect to form? In what ratios? (All equal?). Explain your answer Please explain clearly why the ratio is 1:1. Thank you!

