The temperature of 0.38 kg of water increases by 2.3 °C after transferring some heat. Calculate the heat required in kJ, where a kilojoule is one thousand joules. Approximate the specific heat of water as 4.2 kJ kg1 °C-1| Do not include units in your answer. Write your answer as a number in normal form.
The temperature of 0.38 kg of water increases by 2.3 °C after transferring some heat. Calculate the heat required in kJ, where a kilojoule is one thousand joules. Approximate the specific heat of water as 4.2 kJ kg1 °C-1| Do not include units in your answer. Write your answer as a number in normal form.
College Physics
10th Edition
ISBN:9781285737027
Author:Raymond A. Serway, Chris Vuille
Publisher:Raymond A. Serway, Chris Vuille
Chapter11: Energy In Thermal Processes
Section: Chapter Questions
Problem 6WUE
Related questions
Question
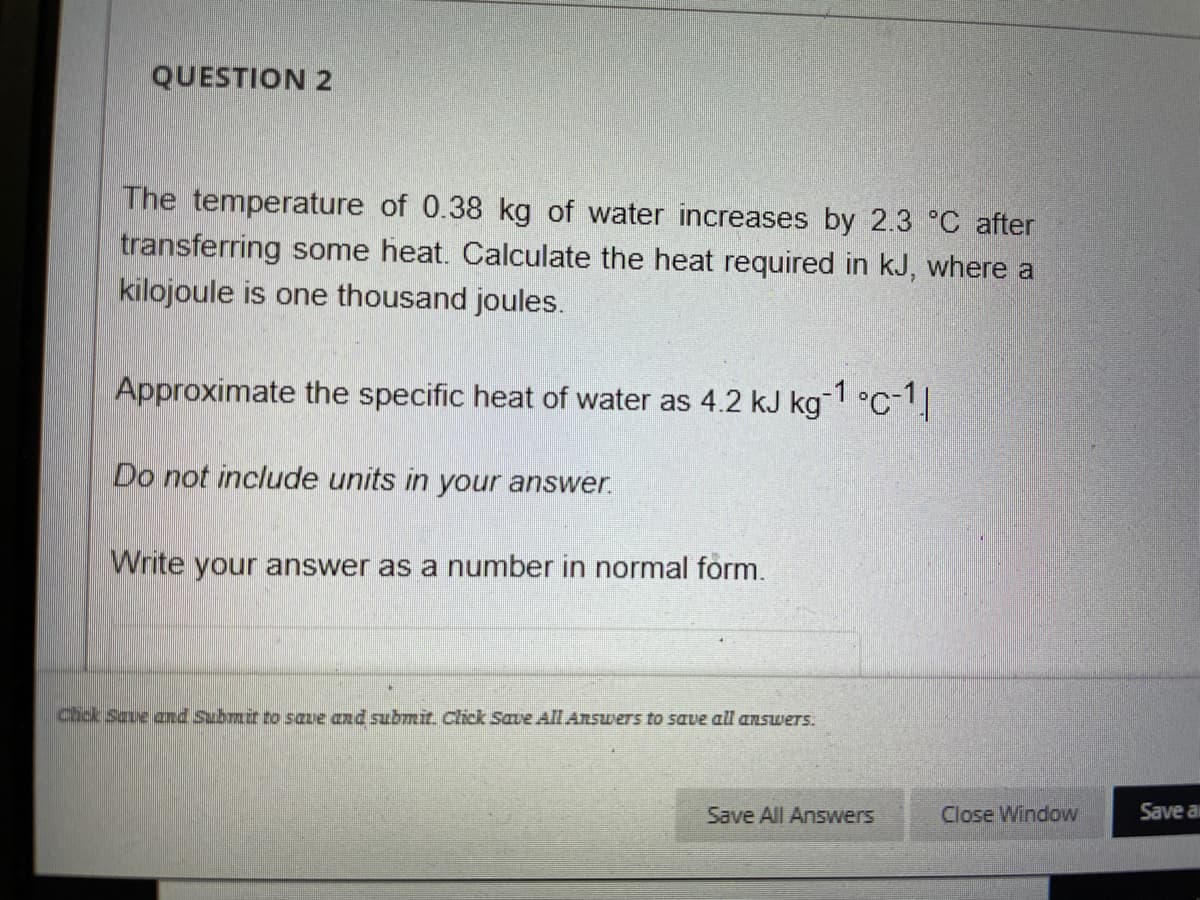
Transcribed Image Text:QUESTION 2
The temperature of 0.38 kg of water increases by 2.3 °C after
transferring some heat. Calculate the heat required in kJ, where a
kilojoule is one thousand joules.
Approximate the specific heat of water as 4.2 kJ kg 1 °C-1
Do not include units in your answer.
Write your answer as a number in normal form.
Chck Save and Submit to save and submit. Click Save All Answers to save all answers.
Save All Answers
Close Window
Save al
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Recommended textbooks for you

College Physics
Physics
ISBN:
9781285737027
Author:
Raymond A. Serway, Chris Vuille
Publisher:
Cengage Learning

College Physics
Physics
ISBN:
9781938168000
Author:
Paul Peter Urone, Roger Hinrichs
Publisher:
OpenStax College


College Physics
Physics
ISBN:
9781285737027
Author:
Raymond A. Serway, Chris Vuille
Publisher:
Cengage Learning

College Physics
Physics
ISBN:
9781938168000
Author:
Paul Peter Urone, Roger Hinrichs
Publisher:
OpenStax College


Physics for Scientists and Engineers, Technology …
Physics
ISBN:
9781305116399
Author:
Raymond A. Serway, John W. Jewett
Publisher:
Cengage Learning

An Introduction to Physical Science
Physics
ISBN:
9781305079137
Author:
James Shipman, Jerry D. Wilson, Charles A. Higgins, Omar Torres
Publisher:
Cengage Learning
