Chapter26: Biomolecules: Amino Acids, Peptides, And Proteins
Section26.SE: Something Extra
Problem 25MP: The chloromethylated polystyrene resin used for Merrifield solidphase peptide synthesis is prepared...
Related questions
Question
This is the 8 step synthesis of Ibuprofen. Please check if correct while keeping only 8 steps
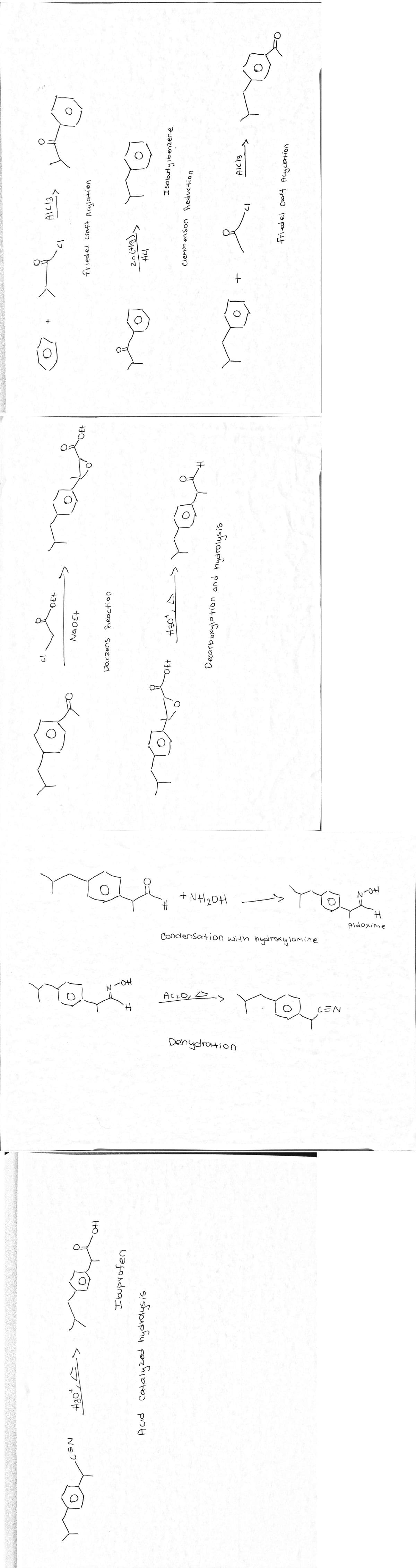
Transcribed Image Text:NH20H
N-OH
Aldoxime
Condensation with hydroxylamine
-0H
AczO, E>
CEN
Denydration
friedel craft Acylation
DE+
Isobutyibenzene
duazuaqıh
Na OEt
clemmenson Reductio.
H.
+30.
2 =
HO
NE?
H30°
I buprofen
Acid Catalyzed hydrdysis
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning


Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning