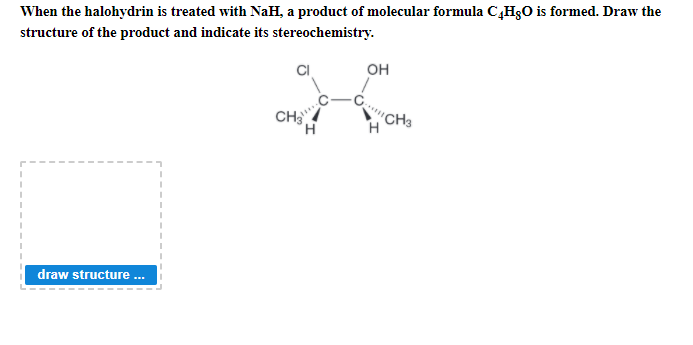
Vhen the halohydrin is treated with NaH, a product of molecular formula C¸H§O is formed. Draw the ructure of the product and indicate its stereochemistry. он CH3 CH draw structure .
Q: A в
A: When A is treated with only C6H5Li (excess), When A is treated with H2O,
Q: Draw the products formed when each alkyne is treated with O3 followed by H2O.
A: Since in the reaction of alkyne with O3 followed by H2O, the triple bond is broken and the carbon is…
Q: What reagents are needed to convert each compound to acetophenone (C6H5COCH3)?
A: Reagent use to convert acetophenone (C6H5COCH3) to Benzene (C6H6) is CH3COCl in presence of AlCl3.
Q: Br .S.
A: SOLUTIONS: Step 1: The above compound is a secondary alkyl halide. The secondary alkyl halide when…
Q: lejimalide B, an anticancer agent with a 24-membered ring, is isolated from a tunicate found off le…
A: For E and Z compound: If the groups attached with the C=C double bond having the high priority…
Q: Iejimalide B, an anticancer agent with a 24-membered ring, is isolated from a tunicate found off Ie…
A: How to find E and Z-configuration? First of all look out for the double bonds in the structure and…
Q: Draw the products of each reaction, and include the stereochemistry at any stereogenic centers in…
A: p-toluenesulfonic acid is abbreviated as OTs OTs is a better leaving group Alkyl tosylate (R-OTs )…
Q: Draw the product formed when the α,β-unsaturated ketone A is treated with each reagent.a. NaBH4,…
A: NOTE: Since, we only answer upto three sub-parts, we'll answer the first 3. Please resubmit the…
Q: Draw the products formed when each compound is treated with HNO3 and H2SO4.
A: EXPLANATION: a) part -OCH3 is ortho, para director whereas -COOCH3 is a meta director.Because -OCH3…
Q: Draw the products of each reaction and indicate the stereochemistry at any stereogenic centers.
A: (a)
Q: OMe OH он 1. DIBAL-H, THF then H3O* 2. H20 EtO он бМе 3. AcO, Py, CH,Cą 4. Nai, acetone draw…
A: DIBAL-H is a reducing agent for the reduction of esters, nitriles, lactones, etc. NaI is a reagent…
Q: Draw the product formed when each starting material is treated with LDA in THF solution at −78 °C.
A:
Q: 10.46 Draw the products formed when (CH3)2C=CH2 is treated with each reagent. f. NBS (aqueous DMSO)…
A: Organic reactions involve addition, substitution, elimination and rearrangement of functional…
Q: Draw a stepwise mechanism for the following reaction and explain the stereochemistry of the product.
A: The protonation step is shown below.
Q: One step in the synthesis of the nonsteroidal anti-inammatory drug rofecoxib (trade name Vioxx)…
A: Suzuki reaction is a type of coupling reaction. The product formed by the reaction is R-R. It…
Q: Draw the products formed (including stereoisomers) when attached compound is reduced with NaBH4 in…
A: Reduction of aldehyde: NaBH4 reduces the aldehyde into a primary alcohol. It acts as a source of…
Q: Ethanal is treated with CyclohexylMgCl/H2O and resulting product is dehydrated by Conc.H2SO4.
A: Answer:
Q: c. Assign stereochemistry to the product. d. Draw the mechanism. NaOEt E2
A: E2 is one step reaction like SN2. Rate of E2 reaction = [alkyl halide] [base] Overall order of a E2…
Q: 20. Draw the major substitution product with the correct stereochemistry. HO (1) PB 3, Pyridine (2)…
A: Applying concept of substitution and elimination reaction.
Q: 10.29 When ethylbenzene is treated with one equivalent of NBS and irradiated with UV light, two…
A: Racemic (1:1) The stereoisomers I and II forms in equal amounts
Q: Draw a stepwise mechanism for the following reaction, the last step in afive-step industrial…
A:
Q: (a) Label the carbon–carbon double bond in A as E or Z. (b) Draw the products (including…
A: a) The black color ball in the given figure represents C and the white ball indicates H atom. E-Z…
Q: Draw the products of each reaction and indicate the stereochemistry at any stereogenic centers.…
A: The product with stereochemistry at stereo genic centers are given below a) b) c)
Q: What product is formed when each carbonyl compound is treated with K2Cr207?
A: a. The given carbonyl compound contains a -CHO group. On the oxidation of aldehydes in the presence…
Q: Draw the products formed when each compound is treated with HNO3 and H2SO4.State whether the…
A: a) Electron withdrawing groups deactivates the ring towards the electrophilic substitution reaction.…
Q: When attached halohydrin is treated with NaH, a product of molecular formula C4H8O is formed. Draw…
A: In the given reaction, halohydrin is treated with NaH. When a halohydrin is treated with a base…
Q: Draw the products of each reaction, and include the stereochemistry at any stereogenic center in the…
A: Hey since you have posted a question with multiple sub-parts, we will solve first three sub-parts…
Q: ndicate the products obtained in the reaction of propan-2-one with p-chlorobenzaldehyde with aqueou…
A:
Q: When attached halohydrin is treated with NaH, a product of molecularformula C4H8O is formed. Draw…
A: In the given reaction, halohydrin is treated with sodium hydride (NaH). When a halohydrin is treated…
Q: Draw the products of each reaction and indicate the stereochemistry atany stereogenic centers.
A: (a)
Q: Draw the product formed when each compound is treated with two equivalents of CH3CH2CH2CH2MgBr…
A: The given compounds contain the carbonyl groups. The reagent given is an organometallic compound.…
Q: What product is formed when each compound is treated with either Ag2O, NH4OH or Na2Cr2O7, H2SO4,…
A: Since among the above reagents , Na2Cr2O7, H2SO4, H2O is a strong oxidising agent Hence they will…
Q: Draw the product formed when the α,β-unsaturated ketone A is treatedwith following reagent. H2 (1…
A:
Q: Identify the reagents from a to d HO HO CH3 H3CO CH3 a C CH-CH3
A: Each reagent along with its name is given in step 2. please proceed to step 2.
Q: 5.66 A mixture contains equal amounts of compounds A-D. A B C D a. Which compounds alone are…
A: A,B,C,D are four compounds.The asteric mark(*) indicated for chiral centre in the below structures.
Q: When attached halohydrin is treated with NaH, a product of molecular formula C4H8O is formed. Draw…
A: When attached halohydrin is treated with NaH, a product of molecular formula C4H8O is formed. The…
Q: (a) Label the carbon–carbon double bond in A as E or Z. (b) Draw the products (including…
A: a) The black color ball in the given figure represents C and the white ball indicates H atom. E-Z…
Q: Even though B contains three ester groups, a single Dieckmann product results when B is treated with…
A: Given compound:
Q: What results when (Z)-3-methylpent-2-en-1-ol is treated with tBuOOH, Ti(OiPr)4, and (-)-DET? HO an…
A: Given reaction is known as Sharpless Asymmetric epoxidation reaction. In this reaction ,…
Q: Dimethyl cyclopropanes can be prepared by the reaction of an α, β-unsaturated carbonyl compound X…
A:
Q: (a) Draw the structures of A to Ein the following reactions. 1. LIAH, 2. H0 PCC 00 H Lindias…
A: This is an opeing of epoxide by nucleophiles such as LiAlH4. The opening of epoxide is always at…
Q: Draw the products formed (including stereoisomers) in each reaction.
A: Lithium diisopropylamide is a strong base and is widely used because of its good solubility in…
Q: он о a) Draw the enantiomer of K. b) Draw an enol tautomer of K. c) Draw the product that forms when…
A:
Q: Draw a stepwise mechanism for the Carroll rearrangement, a reaction that prepares a γ,δ-unsaturated…
A: Carroll rearrangement is a reaction in which β — keto ester reacts with allylic alcohol in the…
Q: 7. Draw the products formed when each compound is trea ted with HNO3 and H2SO4. OCH, OCH, CH3 Br NO2…
A: a) In this methoxy group is ortho para directing hence nitration takes place at ortho position to…
Q: 5.66 A mixture contains equal amounts of compounds A-D. a D B A a. Which compounds alone are…
A:
Q: Draw the product formed when the α,β-unsaturated ketone A is treatedwith following reagent. [1]…
A:
Q: Draw the products formed when each dihalide is treated with excessNaNH2.
A: Given reaction,
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

- When attached halohydrin is treated with NaH, a product of molecular formula C4H8O is formed. Draw the structure of the product and indicateits stereochemistry.When each halohydrin is treated with NaH, a product of molecular formula C4H8O is formed. Draw the structure of the product and indicate its stereochemistry.Draw the products formed when cembrene A is treated with O3 followed by CH3SCH3. Label each product as chiral or achiral
- Draw the products formed (including stereoisomers) when attached compound is reduced with NaBH4 in CH3OH.Draw the products formed when attached alkene is treated with BH3 followed by H2O2, HO−. Include the stereochemistry at all stereogenic centersAn acid–base reaction of (R)-sec-butylamine with a racemic mixture of 2-phenylpropanoic acid forms two products having different melting pointsand somewhat different solubilities. Draw the structure of these twoproducts. Assign R and S to any stereogenic centers in the products.How are the two products related? Choose from enantiomers,diastereomers, constitutional isomers, or not isomers of each other.
- Clopidogrel is the generic name for Plavix, a drug used to prevent the formation of blood clots in patients that have a history of heart attacks or strokes. A single enantiomer of clopidogrel can be prepared in three steps from the chiral αhydroxy acid A. Identify B and C in the following reaction sequence, and designate the conguration of the enantiomer formed by this route as R or S.Draw the products formed when p-methylaniline (p-CH3C6H4NH2) istreated with following reagent. Part (b), then CH3COCl, AlCl3Draw the products formed when CH3COCH2CH2CH = CH2 is treated with each reagent:(a) LiAlH4, then H2O;(b) NaBH4 in CH3OH;(c) H2 (1 equiv), Pd-C;(d) H2 (excess), Pd-C;(e) NaBH4 (excess) in CH3OH;(f) NaBD4 in CH3OH.
- Draw the products obtained (including stereochemistry) when each compound is treated with LDA, followed by CH3I.Draw the product formed when phenylacetic acid (C6H5CH2COOH) istreated with following reagent. With some reagents, no reaction occurs. CH3NH2, DCCName the structure and give it's stereogenic centers as well as it's stereochemistry (R or S)

