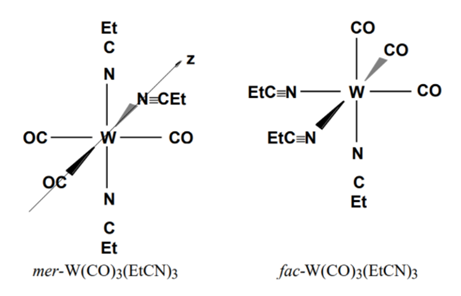
Tungsten hexacarbonyl (W(CO)6) is reacted with (EtC≡N) and the product is W(CO)3(N≡CEt)3. However, the product can be a mixture of fac and mer isomers. But a Chemist can determine from analysis of the infrared spectrum if the product is indeed a mixture of isomers or not. In the synthesis experiment, the product shows 3 carbonyl stretching frequencies in the infrared spectrum, at 2021, 1898 and 1840 cm-1. Determine the irreducible representations of the stretches expected for each of the two species in their respective point group. Can you confirm what isomer was isolated?
Tungsten hexacarbonyl (W(CO)6) is reacted with (EtC≡N) and the product is W(CO)3(N≡CEt)3. However, the product can be a mixture of fac and mer isomers. But a Chemist can determine from analysis of the infrared spectrum if the product is indeed a mixture of isomers or not. In the synthesis experiment, the product shows 3 carbonyl stretching frequencies in the infrared spectrum, at 2021, 1898 and 1840 cm-1. Determine the irreducible representations of the stretches expected for each of the two species in their respective point group. Can you confirm what isomer was isolated?
Chemistry by OpenStax (2015-05-04)
1st Edition
ISBN:9781938168390
Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Chapter19: Transition Metals And Coordination Chemistry
Section: Chapter Questions
Problem 24E: Predict which will be more stable, [CrO4]2- or [WO4]2-, and explain.
Related questions
Question
Tungsten hexacarbonyl (W(CO)6) is reacted with (EtC≡N) and the product is W(CO)3(N≡CEt)3.
However, the product can be a mixture of fac and mer isomers. But a Chemist can determine from
analysis of the infrared spectrum if the product is indeed a mixture of isomers or not. In the
synthesis experiment, the product shows 3 carbonyl stretching frequencies in the infrared
spectrum, at 2021, 1898 and 1840 cm-1.
Determine the irreducible representations of the stretches
expected for each of the two species in their respective point group. Can you confirm what isomer
was isolated?
Transcribed Image Text:Et
co
co
N
EtC=N -
W — со
co
N CEt
W-
co
EtC=N
N
N
Et
Et
mer-W(CO)3(EtCN)3
fac-W(CO)3(EtCN)3
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning