UM College of Arts and Sciences Education 2nd Floor, DPT Building, Matina Campus, Davao City The University of Mindanao Phone No.: (082)300-5456/305-0647 Local 134 LET'S ANALYZE Activity #37. Balance the following equations and indicate the types of reaction. Reaction 1. BaCl2 + KIO: → Ba(IO )2 + KCI 2. H2SO4 + NaCN → HCN + NazSO 3. Sn + NaOH > NazSnO, + Hz 4. K+ Brz→ KBr 5 Sb + 12 → Sbla 6. Sr + O2 → SrO 7 KCIO > KCI + 0, 8. Reaction: Reaction Reaction: Reaction: Reaction Reaction Reaction: Reaction: Reaction H, + F2> HE 9 Fe + Oz→ Fe:O 10 Mg + HCI) M9CI, + H
UM College of Arts and Sciences Education 2nd Floor, DPT Building, Matina Campus, Davao City The University of Mindanao Phone No.: (082)300-5456/305-0647 Local 134 LET'S ANALYZE Activity #37. Balance the following equations and indicate the types of reaction. Reaction 1. BaCl2 + KIO: → Ba(IO )2 + KCI 2. H2SO4 + NaCN → HCN + NazSO 3. Sn + NaOH > NazSnO, + Hz 4. K+ Brz→ KBr 5 Sb + 12 → Sbla 6. Sr + O2 → SrO 7 KCIO > KCI + 0, 8. Reaction: Reaction Reaction: Reaction: Reaction Reaction Reaction: Reaction: Reaction H, + F2> HE 9 Fe + Oz→ Fe:O 10 Mg + HCI) M9CI, + H
Chapter7: Statistical Data Treatment And Evaluation
Section: Chapter Questions
Problem 7.10QAP
Related questions
Question
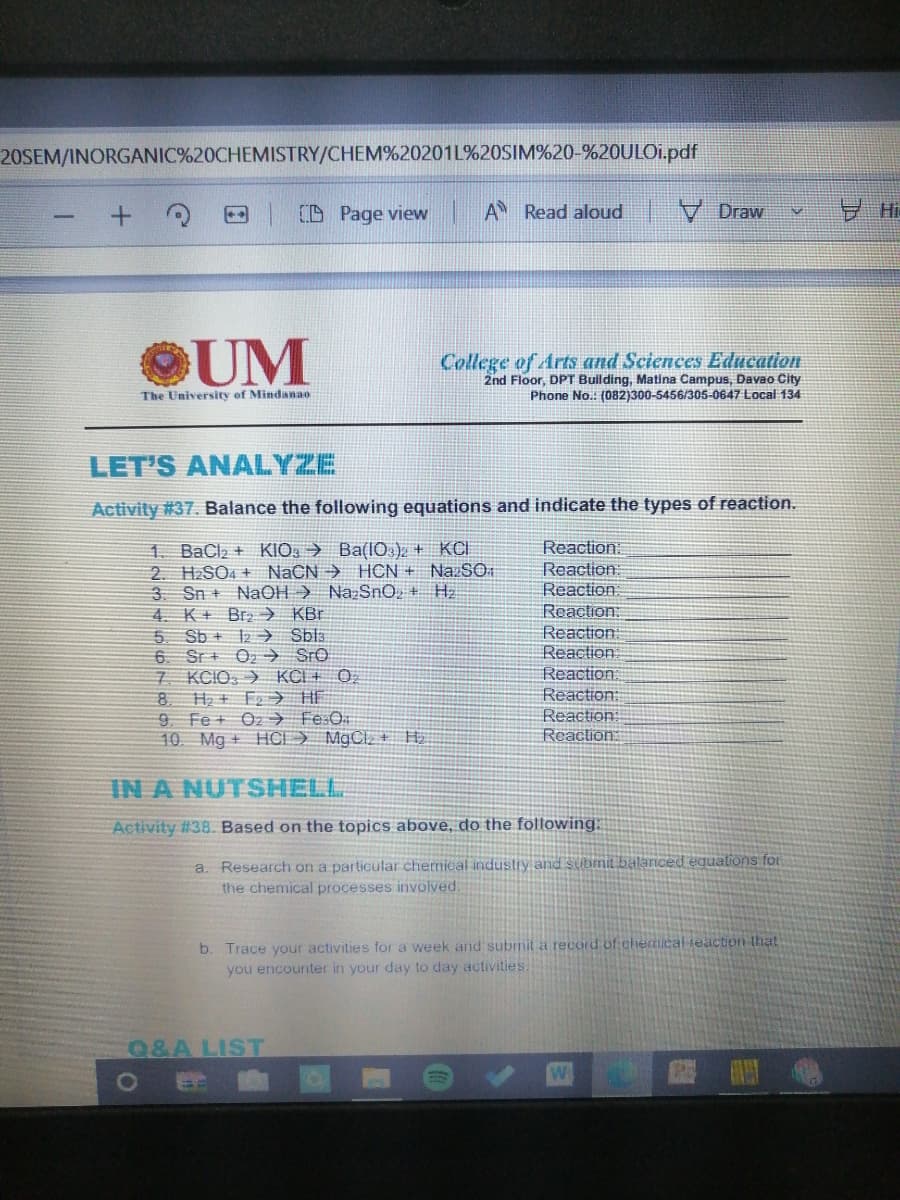
Transcribed Image Text:20SEM/INORGANIC%20CHEMISTRY/CHEM%20201L%20SIM%20-%20ULO1.pdf
(D Page view
A Read aloud
V Draw
UM
College of Arts and Sciences Education
2nd Floor, DPT Bullding, Matina Campus, Davao City
Phone No.: (082)300-5456/305-0647 Local 134
The University of Mindanao
LET'S ANALYZE
Activity #37. Balance the following equations and indicate the types of reaction.
1. BaCl2 + KIO: → Ba(l0.) + KCI
2. H2SO4 + NACN → HCN + NazSO
3. Sn + NaOH ) NazSnO, + H2
4. K+ Br > KBr
5 Sb + 2 → Sbla
6. Sr + Oz → Sro
7. KCIO, ) KCI + 0,
8 H + F2> HE
9 Fe + Oz → Fe:O.
10 Mg + HCI ) MgCl, + H.
Reaction:
Reaction:
Reaction:
Reaction:
Reaction:
Reaction:
Reaction
Reaction:
Reaction:
Reaction:
IN A NUTSHELL
Activity #38. Based on the topics above, do the following:
a. Research on a particular chemical industiy and submit balanced equalions for
the chemical processes involved.
b. Trace your activities for a week and submit a record of chemicalieaction that
you encounter in your day to day activities.
Q&A LIST
日
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Recommended textbooks for you

