Unknown Code Sample A Sample D Sample E Sample G Sample F Calculated Density using the Ruler (g/cm3) Calculated Density using Water Displacement (g/mL) Average Experimental Density of Both Methods (g/cm3) Identity of Unknown (compare the average experimental density with density from the table given above). ////////// ////////// ////////// ////////// ////////// ////////// "True" Identity of Unknown from the Key Table Copper PVC Aluminum Acrylic Iron "True " Density of Unknown from the Key Table (g/cm3) 8.96 1.39 2.70 1.15-1.20 7.87 % Error Iron - Density(g/cm^3) = 7.87
Unknown Code Sample A Sample D Sample E Sample G Sample F Calculated Density using the Ruler (g/cm3) Calculated Density using Water Displacement (g/mL) Average Experimental Density of Both Methods (g/cm3) Identity of Unknown (compare the average experimental density with density from the table given above). ////////// ////////// ////////// ////////// ////////// ////////// "True" Identity of Unknown from the Key Table Copper PVC Aluminum Acrylic Iron "True " Density of Unknown from the Key Table (g/cm3) 8.96 1.39 2.70 1.15-1.20 7.87 % Error Iron - Density(g/cm^3) = 7.87
Introductory Chemistry: A Foundation
9th Edition
ISBN:9781337399425
Author:Steven S. Zumdahl, Donald J. DeCoste
Publisher:Steven S. Zumdahl, Donald J. DeCoste
Chapter2: Measurements And Calculations
Section: Chapter Questions
Problem 32QAP: Why can the length of the pin shown in Fig. 2.5 not be recorded as 2.850 cm?
Related questions
Question
| Unknown Code | Sample A | Sample D | Sample E | Sample G | Sample F |
| Calculated Density using the Ruler (g/cm3) | |||||
| Calculated Density using Water Displacement (g/mL) | |||||
| Average Experimental Density of Both Methods (g/cm3) | |||||
| Identity of Unknown (compare the average experimental density with density from the table given above). | |||||
| ////////// | ////////// | ////////// | ////////// | ////////// | ////////// |
| "True" Identity of Unknown from the Key Table | Copper | PVC | Aluminum | Acrylic | Iron |
| "True " Density of Unknown from the Key Table (g/cm3) | 8.96 | 1.39 | 2.70 | 1.15-1.20 | 7.87 |
| % Error | |||||
Iron - Density(g/cm^3) = 7.87
Copper-Density(g/cm^3) = 8.96
Aluminum-Density(g/cm^3) =2.70
Acrylic-Density(g/cm^3) = 1.15-1.20
PVC-Density(g/cm^3) =1.39
Please complete the rest of the table and show work. Information about this is on the attached photos.Thank you
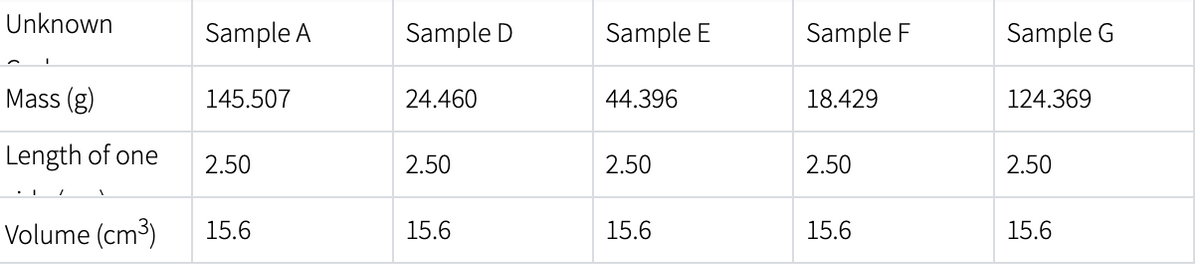
Transcribed Image Text:Unknown
Mass (g)
Length of one
Volume (cm³)
Sample A
145.507
2.50
15.6
Sample D
24.460
2.50
15.6
Sample E
44.396
2.50
15.6
Sample F
18.429
2.50
15.6
Sample G
124.369
2.50
15.6
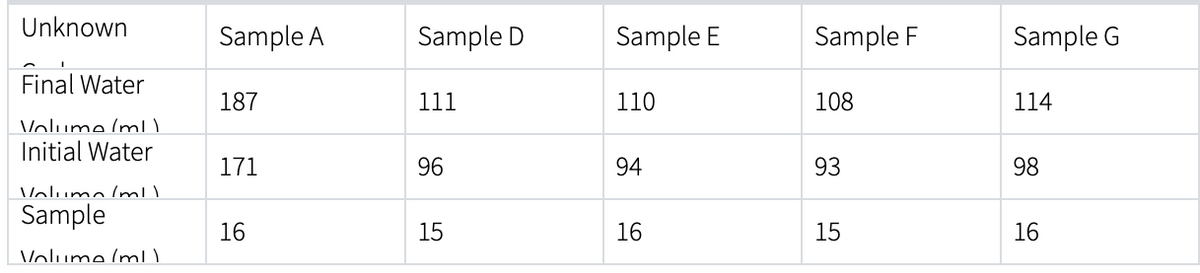
Transcribed Image Text:Unknown
Final Water
Volume (ml)
Initial Water
Volumo/ml)
Sample
Volume (ml)
Sample A
187
171
16
Sample D
111
96
15
Sample E
110
94
16
Sample F
108
93
15
Sample G
114
98
16
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Chemistry: Matter and Change
Chemistry
ISBN:
9780078746376
Author:
Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:
Glencoe/McGraw-Hill School Pub Co

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Chemistry: Matter and Change
Chemistry
ISBN:
9780078746376
Author:
Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:
Glencoe/McGraw-Hill School Pub Co

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning