urce Dose (mremlyear) turally Occurring Radiation emic rays rrestrial radiation (rocks, buildings) side human body (K-40 and Ra-266 in the bones) don in the air tal 27 28 39 200 294 mificial Radiation dical Xrays aclear medicine nsumer products aclear power plants I others tal 39 14 10 0.5 1.5 65.0 rand total 359 e the data in the table to decide which is more serious: an accident involving radiation exposure in which a person receives 4.23x10* Sv or the amount of radiation that the average person receives hually from terrestrial radiation (rocks, buildings) . (1 rem = 0.01 Sv) cident rrestrial radiation (rocks, buildings)
urce Dose (mremlyear) turally Occurring Radiation emic rays rrestrial radiation (rocks, buildings) side human body (K-40 and Ra-266 in the bones) don in the air tal 27 28 39 200 294 mificial Radiation dical Xrays aclear medicine nsumer products aclear power plants I others tal 39 14 10 0.5 1.5 65.0 rand total 359 e the data in the table to decide which is more serious: an accident involving radiation exposure in which a person receives 4.23x10* Sv or the amount of radiation that the average person receives hually from terrestrial radiation (rocks, buildings) . (1 rem = 0.01 Sv) cident rrestrial radiation (rocks, buildings)
Chemistry for Engineering Students
4th Edition
ISBN:9781337398909
Author:Lawrence S. Brown, Tom Holme
Publisher:Lawrence S. Brown, Tom Holme
Chapter14: Nuclear Chemistry
Section: Chapter Questions
Problem 14.73PAE
Related questions
Question
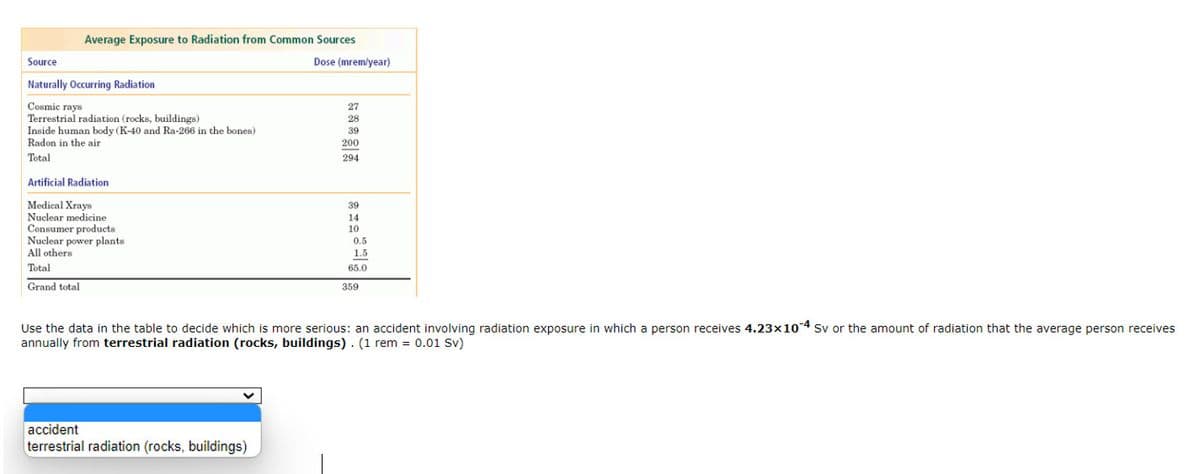
Transcribed Image Text:Average Exposure to Radiation from Common Sources
Source
Dose (mremlyear)
Naturally Occurring Radiation
Cosmic rays
Terrestrial radiation (rocks, buildings)
Inside human body (K-40 and Ra-266 in the bones)
Radon in the air
27
28
39
200
Total
294
Artificial Radiation
Medical Xrays
Nuclear medicine
Consumer products
Nuclear power plants
All others
39
14
10
0.5
1.5
Total
65.0
Grand total
359
Use the data in the table to decide which is more serious: an accident involving radiation exposure in which a person receives 4.23x104 Sv or the amount of radiation that the average person receives
annually from terrestrial radiation (rocks, buildings). (1 rem = 0.01 Sv)
accident
terrestrial radiation (rocks, buildings)
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

World of Chemistry, 3rd edition
Chemistry
ISBN:
9781133109655
Author:
Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:
Brooks / Cole / Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

World of Chemistry, 3rd edition
Chemistry
ISBN:
9781133109655
Author:
Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:
Brooks / Cole / Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning