Use standard reduction potentials to determine the approximate magnitude of the equilibrium constant for the following redox reaction a 2 Cr3+ (aq) + 3 Fe(s) 3 Fe2+ (aq) + 2 Cr(s) - Fe2+ (aq) + 2 e→→ Fe(s) E = -0.44 V Cr3+ (aq) + 3 e Cr(s) E = -0.74 V CHOOSE THE CLOSEST ANSWER IF YOUR EXACT ANSWER IS NOT PRESENT. OA 10-31 OB. 1 OC-1 OD. 1015 OE 10-11 OF. 10-16 OG. 1010 OH. 1030 QU A Moving to another question will save this response.
Use standard reduction potentials to determine the approximate magnitude of the equilibrium constant for the following redox reaction a 2 Cr3+ (aq) + 3 Fe(s) 3 Fe2+ (aq) + 2 Cr(s) - Fe2+ (aq) + 2 e→→ Fe(s) E = -0.44 V Cr3+ (aq) + 3 e Cr(s) E = -0.74 V CHOOSE THE CLOSEST ANSWER IF YOUR EXACT ANSWER IS NOT PRESENT. OA 10-31 OB. 1 OC-1 OD. 1015 OE 10-11 OF. 10-16 OG. 1010 OH. 1030 QU A Moving to another question will save this response.
Chapter18: Electrochemistry
Section: Chapter Questions
Problem 150CP: Given the following two standard reduction potentials, solve for the standard reduction potential of...
Related questions
Question
3
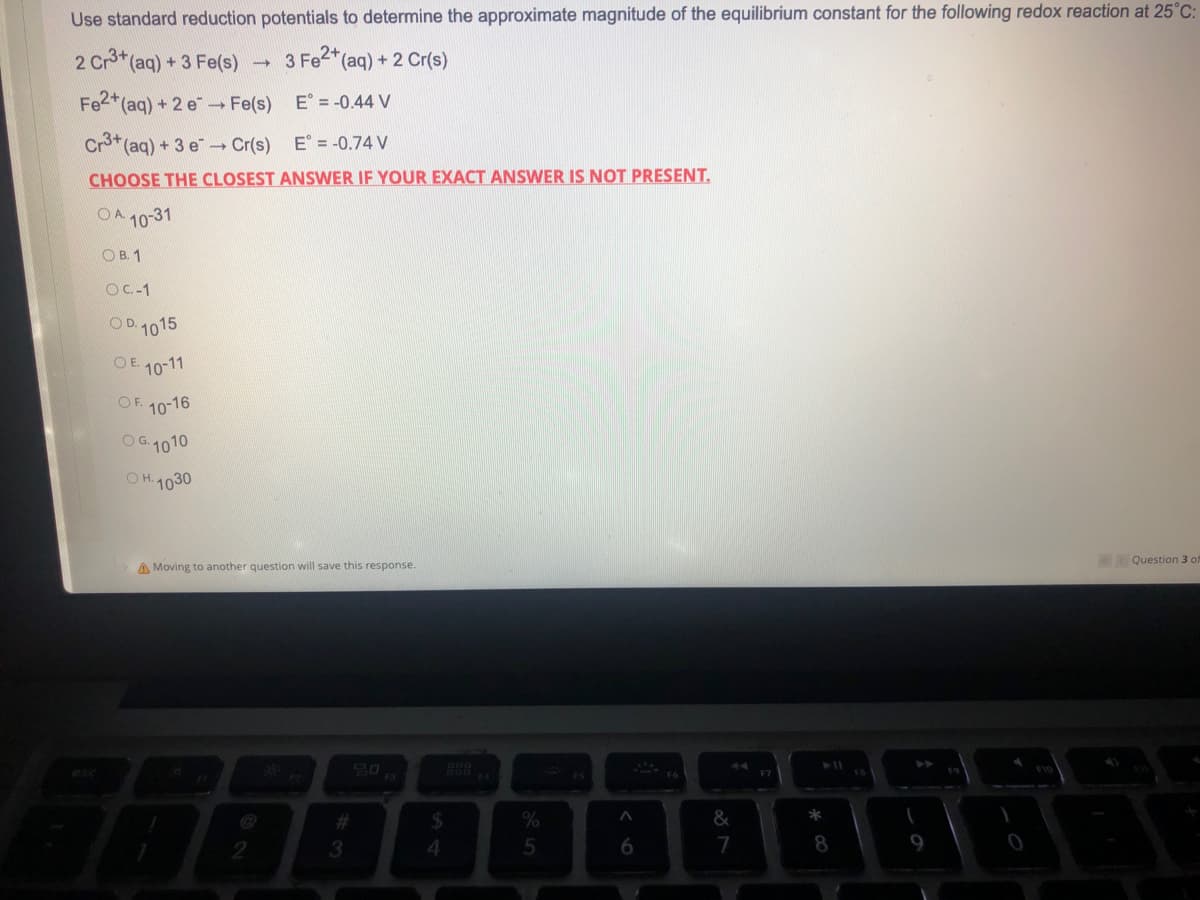
Transcribed Image Text:Use standard reduction potentials to determine the approximate magnitude of the equilibrium constant for the following redox reaction at 25°C:
2 Cr3+ (aq) + 3 Fe(s)
3 Fe2+ (aq) + 2 Cr(s)
-
Fe2+ (aq) + 2 e→→ Fe(s)
E = -0.44 V
Cr3+ (aq) + 3 e Cr(s)
E = -0.74 V
CHOOSE THE CLOSEST ANSWER IF YOUR EXACT ANSWER IS NOT PRESENT.
OA 10-31
OB. 1
OC.-1
OD. 1015
OE 10-11
OF 10-16
OG. 1010
OH. 1030
<< Question 3 of
A Moving to another question will save this response.
F7
#
F
2
3
$
4
%
5
6
&
7
*
8
9
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 3 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning