Use the following information to answer question 35. An Equilibrium System N₂O4(g) + energy = 2 NO₂(g) Row A. B. C. D. Concentration (mol/L) N₂O4(8) NO₂(g) Time (min) 35. The equilibrium system shown in the graph above responds to a change caused by the addition of i and shifts toward the ii The statement above is completed by the information in row i ii NO₂(g) di products NO₂(g) reactants N₂O4(g) products N₂04(g) OOOOH reactants alevik o
Use the following information to answer question 35. An Equilibrium System N₂O4(g) + energy = 2 NO₂(g) Row A. B. C. D. Concentration (mol/L) N₂O4(8) NO₂(g) Time (min) 35. The equilibrium system shown in the graph above responds to a change caused by the addition of i and shifts toward the ii The statement above is completed by the information in row i ii NO₂(g) di products NO₂(g) reactants N₂O4(g) products N₂04(g) OOOOH reactants alevik o
Chapter4: Calculations Used In Analytical Chemistry
Section: Chapter Questions
Problem 4.26QAP
Related questions
Question
Need these two!!
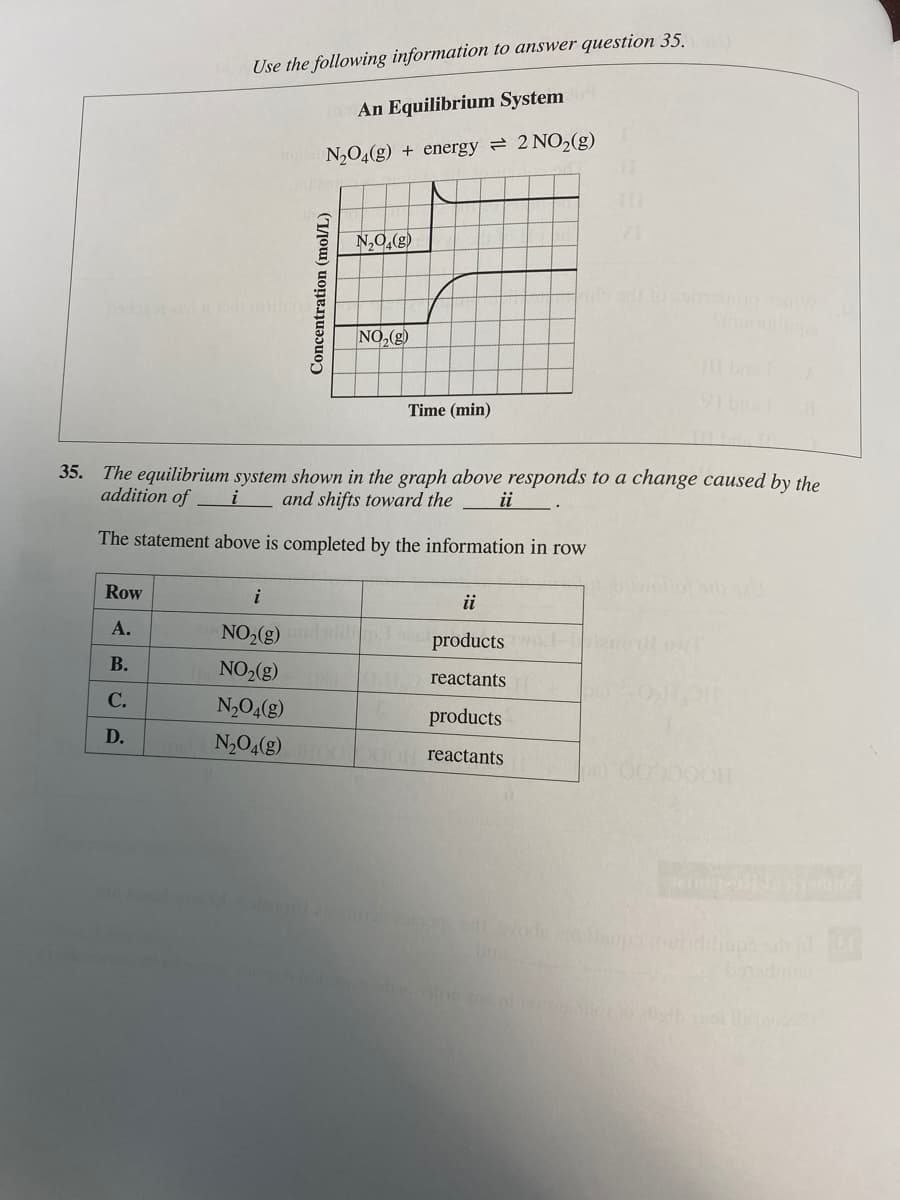
Transcribed Image Text:Use the following information to answer question 35.
An Equilibrium System
N₂O4(g) + energy = 2 NO₂(g)
Row
A.
B.
C.
D.
Concentration (mol/L)
N₂O4(8)
NO₂(g)
Time (min)
35. The equilibrium system shown in the graph above responds to a change caused by the
addition of
i and shifts toward the
ii
The statement above is completed by the information in row
i
ii
NO₂(g) di products wo
NO₂(g)
reactants
N₂O4(g)
products
N₂O4(g) OOOOH reactants
Digib suot Us Lande?)

Transcribed Image Text:A.
34. Which properties of the chromate/dichromate system would confirm that it has reached
equilibrium?
B.
C.
Use the following additional information to answer question 34.
Possible Properties of a System
The pressure of the system is constant.
The colour of the system is constant.
The mass of the system is constant.
The pH of the system is constant.
D.
I
II
III
IV
I and III
I and IV
II and III
II and IV
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

World of Chemistry, 3rd edition
Chemistry
ISBN:
9781133109655
Author:
Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:
Brooks / Cole / Cengage Learning


Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

World of Chemistry, 3rd edition
Chemistry
ISBN:
9781133109655
Author:
Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:
Brooks / Cole / Cengage Learning

Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning

Chemistry for Today: General, Organic, and Bioche…
Chemistry
ISBN:
9781305960060
Author:
Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:
Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:
9780078746376
Author:
Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:
Glencoe/McGraw-Hill School Pub Co