Use the molar bond enthalpy data in the table to estimate the Average molar bond enthalpies. (Hbond) value of AHKn for the equation Bond -1 kJ.mol -1 Bond kJ mol CCI, (g)2 F2(g) CF, (g) 2 Cl2 (g) О-Н 464 C=N 890 О-О The bonding in the molecules is shown. 142 N-H 390 С-О 351 N-N 159 O=O 502 N=N 418 CI F F-F C-CI C=O + 730 N=N 945 C CCI F-CF + C CI F-F С-С 347 F-F 155 Cl-C C=C 243 615 C C 192 Br-Br 811 С -Н 435 Н-Н 414 kJ AHxn 565 Н-F 439 С -F 431 Н-СI 331 С-СI 368 Н-Вг 276 С-Вг 364 Н-S 293 С-N 225 S-S 615 C=N
Use the molar bond enthalpy data in the table to estimate the Average molar bond enthalpies. (Hbond) value of AHKn for the equation Bond -1 kJ.mol -1 Bond kJ mol CCI, (g)2 F2(g) CF, (g) 2 Cl2 (g) О-Н 464 C=N 890 О-О The bonding in the molecules is shown. 142 N-H 390 С-О 351 N-N 159 O=O 502 N=N 418 CI F F-F C-CI C=O + 730 N=N 945 C CCI F-CF + C CI F-F С-С 347 F-F 155 Cl-C C=C 243 615 C C 192 Br-Br 811 С -Н 435 Н-Н 414 kJ AHxn 565 Н-F 439 С -F 431 Н-СI 331 С-СI 368 Н-Вг 276 С-Вг 364 Н-S 293 С-N 225 S-S 615 C=N
Principles of Modern Chemistry
8th Edition
ISBN:9781305079113
Author:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Chapter12: Thermodynamic Processes And Thermochemistry
Section: Chapter Questions
Problem 51P
Related questions
Question
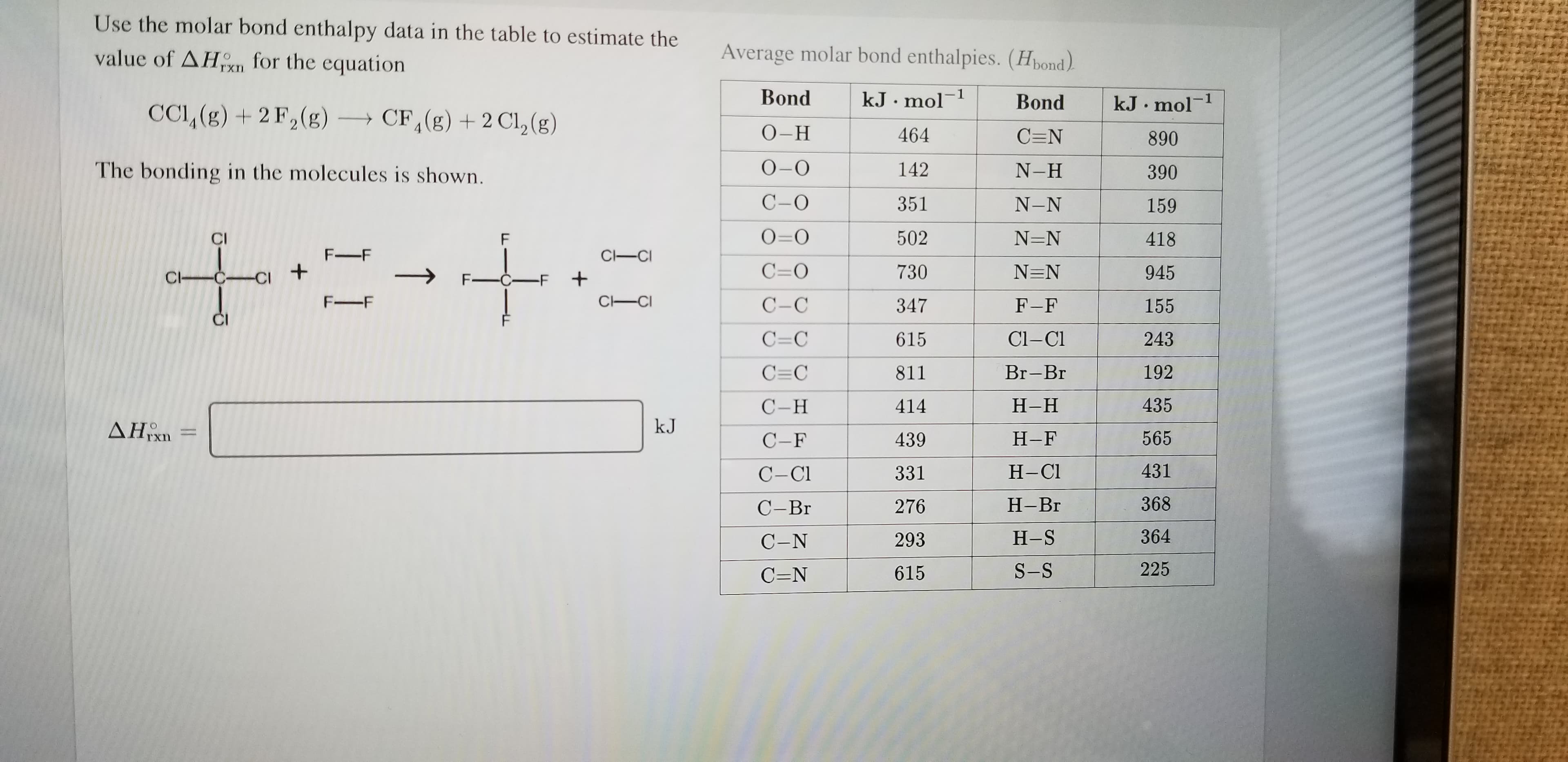
Transcribed Image Text:Use the molar bond enthalpy data in the table to estimate the
Average molar bond enthalpies. (Hbond)
value of AHKn for the
equation
Bond
-1
kJ.mol
-1
Bond
kJ mol
CCI, (g)2 F2(g)
CF, (g) 2 Cl2 (g)
О-Н
464
C=N
890
О-О
The bonding in the molecules is shown.
142
N-H
390
С-О
351
N-N
159
O=O
502
N=N
418
CI
F
F-F
C-CI
C=O
+
730
N=N
945
C CCI
F-CF
+
C CI
F-F
С-С
347
F-F
155
Cl-C
C=C
243
615
C C
192
Br-Br
811
С -Н
435
Н-Н
414
kJ
AHxn
565
Н-F
439
С -F
431
Н-СI
331
С-СI
368
Н-Вг
276
С-Вг
364
Н-S
293
С-N
225
S-S
615
C=N
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Recommended textbooks for you

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:
9780078746376
Author:
Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:
Glencoe/McGraw-Hill School Pub Co

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:
9780078746376
Author:
Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:
Glencoe/McGraw-Hill School Pub Co

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning