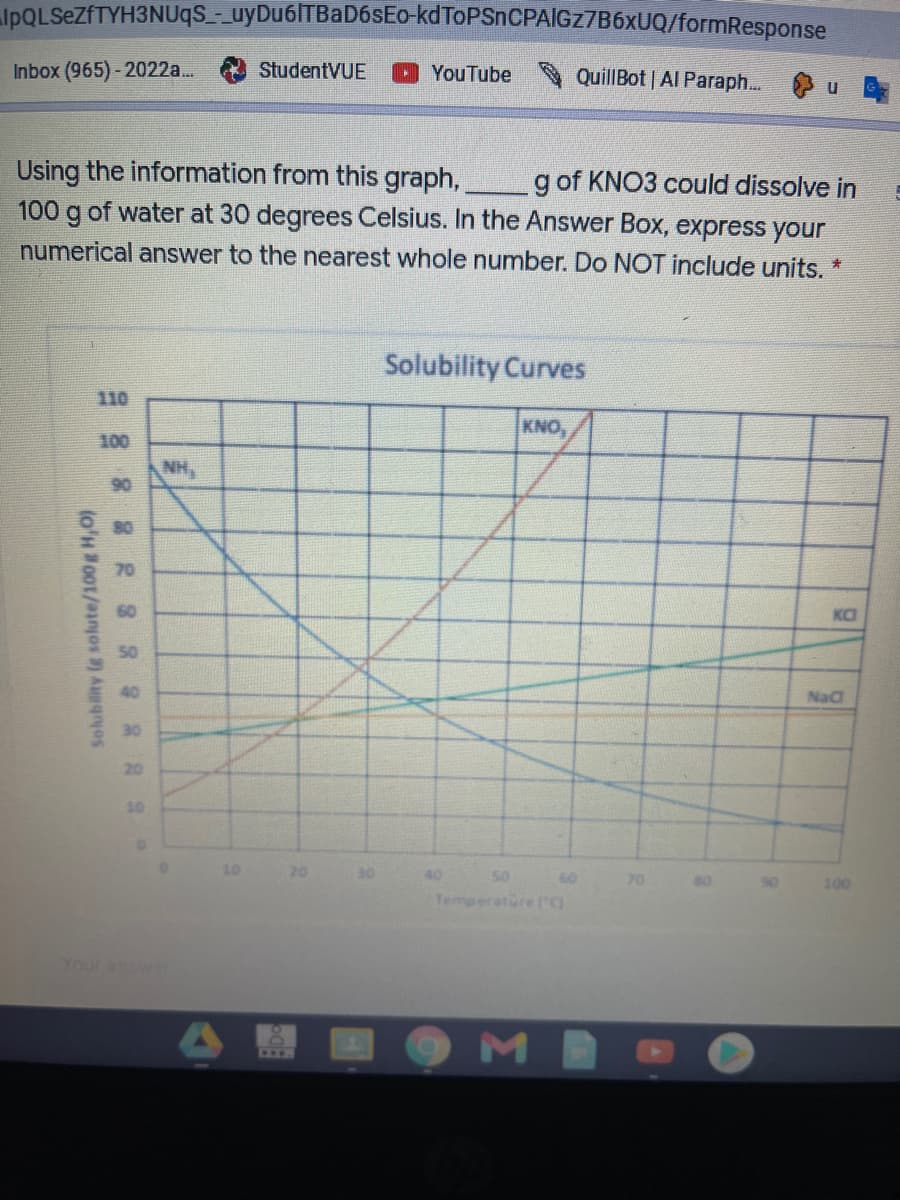
Using the information from this graph, 100 g of water at 30 degrees Celsius. In the Answer Box, express your g of KNO3 could dissolve in numerical answer to the nearest whole number. Do NOT include units. * Solubility Curves 110 KNO, 100 NH, 90 80 70 60 Ka 50 40 Nad 30 20 10 10 20 40 50 60 70 100 Tempe Gre lC Solubility (g solute/100g H,0)
Using the information from this graph, 100 g of water at 30 degrees Celsius. In the Answer Box, express your g of KNO3 could dissolve in numerical answer to the nearest whole number. Do NOT include units. * Solubility Curves 110 KNO, 100 NH, 90 80 70 60 Ka 50 40 Nad 30 20 10 10 20 40 50 60 70 100 Tempe Gre lC Solubility (g solute/100g H,0)
Chapter3: Statistical Tests With Excel
Section: Chapter Questions
Problem 1P
Related questions
Question
100%
Transcribed Image Text:IpQLSeZfTYH3NUqS_- uyDu6ITBaD6sEo-kdToPSnCPAlGZ7B6xUQ/formResponse
Inbox (965)- 2022a...
StudentVUE
QuillBot | Al Paraph...
YouTube
Using the information from this graph,,
100 g of water at 30 degrees Celsius. In the Answer Box, express your
g of KNO3 could dissolve in
numerical answer to the nearest whole number. Do NOT include units. *
Solubility Curves
110
KNO,
100
NH,
90
70
60
KO
50
40
NaC
30
20
10
10
20
30
40
50
60
70
100
Temperatore rg
Your answer
OMB
CEFO
Solubility (g solute/100g H,0)
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you



