V'18, Lavonda Deale ( 4766/ S) A student collected the data shown in the table below during an experiment. Liquid Characteristics Liquid Characteristics Boiling Point Freezing Point Color Toxicity Mercury 357 °C -39 °C shiny silver gray hazardous if tube is broken Alcohol 78 °C -115 °C colorless relatively non-hazardous Based on the data, which of the following conclusions can be made about the use of mercury and alcohol thermometers? O An alcohol thermometer can easily measure the boiling point of a colorless liquid that boils at 100 °C. O A mercury thermometer can easily measure the boiling point of a colorless liquid that boils at 82 °C. O A mercury thermometer can easily measure the freezing point of a colorless liquid that freezes at -50 °C. O An alcohol thermometer can easily measure the freezing point of a colorless liquid that freezes at -110 °C.
V'18, Lavonda Deale ( 4766/ S) A student collected the data shown in the table below during an experiment. Liquid Characteristics Liquid Characteristics Boiling Point Freezing Point Color Toxicity Mercury 357 °C -39 °C shiny silver gray hazardous if tube is broken Alcohol 78 °C -115 °C colorless relatively non-hazardous Based on the data, which of the following conclusions can be made about the use of mercury and alcohol thermometers? O An alcohol thermometer can easily measure the boiling point of a colorless liquid that boils at 100 °C. O A mercury thermometer can easily measure the boiling point of a colorless liquid that boils at 82 °C. O A mercury thermometer can easily measure the freezing point of a colorless liquid that freezes at -50 °C. O An alcohol thermometer can easily measure the freezing point of a colorless liquid that freezes at -110 °C.
Chapter82: Physical Constants Of Liquids: The Boiling Point And Density
Section: Chapter Questions
Problem 5P
Related questions
Question
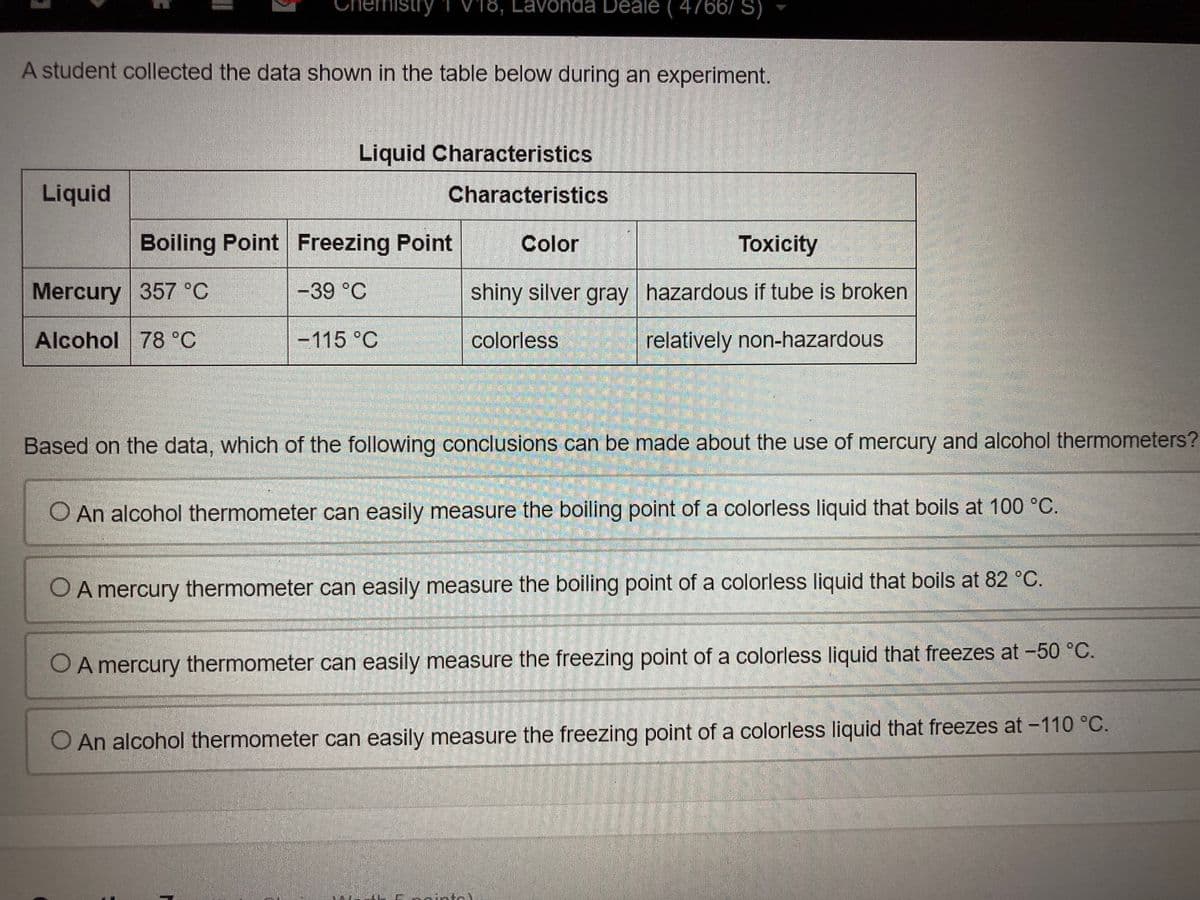
Transcribed Image Text:emistry
V18, Lavonda Deale ( 4766/ S)
A student collected the data shown in the table below during an experiment.
Liquid Characteristics
Liquid
Characteristics
Boiling Point Freezing Point
Color
Toxicity
Mercury 357 °C
-39 °C
shiny silver gray hazardous if tube is broken
Alcohol 78 °C
-115 °C
colorless
relatively non-hazardous
Based on the data, which of the following conclusions can be made about the use of mercury and alcohol thermometers?
O An alcohol thermometer can easily measure the boiling point of a colorless liquid that boils at 100 °C.
O A mercury thermometer can easily measure the boiling point of a colorless liquid that boils at 82 °C.
OA mercury thermometer can easily measure the freezing point of a colorless liquid that freezes at -50 °C.
O An alcohol thermometer can easily measure the freezing point of a colorless liquid that freezes at -110 °C.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:
9781305446021
Author:
Lampman
Publisher:
CENGAGE LEARNING - CONSIGNMENT

Macroscale and Microscale Organic Experiments
Chemistry
ISBN:
9781305577190
Author:
Kenneth L. Williamson, Katherine M. Masters
Publisher:
Brooks Cole

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:
9781305446021
Author:
Lampman
Publisher:
CENGAGE LEARNING - CONSIGNMENT

Macroscale and Microscale Organic Experiments
Chemistry
ISBN:
9781305577190
Author:
Kenneth L. Williamson, Katherine M. Masters
Publisher:
Brooks Cole

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax
