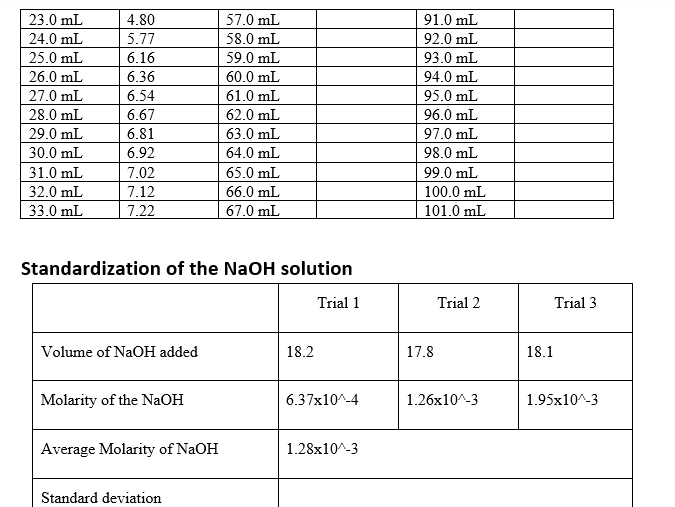
23.0 mL 4.80 57.0 mL 91.0 mL 24.0 mL 5.77 58.0 mL 92.0 mL 25.0 mL 6.16 59.0 mL 93.0 mL 26.0 mL 6.36 60.0 mL 94.0 mL 27.0 mL 6.54 61.0 mL 95.0 mL 28.0 mL 6.67 62.0 mL 96.0 mL 29.0 mL 6.81 63.0 mL 97.0 mL 30.0 mL 6.92 64.0 mL 98.0 mL 31.0 mL 7.02 65.0 mL 99.0 mL 32.0 mL 7.12 66.0 mL 100.0 mL 33.0 mL 7.22 67.0 mL 101.0 mL Standardization of the NaOH solution Trial 1 Trial 2 Trial 3 Volume of NaOH added 18.2 17.8 18.1 Molarity of the NaOH 6.37x10^-4 1.26x10^-3 1.95x10^-3 Average Molarity of NaOH 1.28x10^-3 Standard deviation Volume of weak acid titrated (mL): 25 (mL) Volume of NaOH added pH measured Volume of NaOH added pH measured Volume of pH measured NaOH added 0.0 mL 1.52 34.0 mL 7.31 68.0 mL 1.0 mL 1.48 35.0 mL 7.42 69.0 mL 2.0 mL 1.45 36.0 mL 7.51 70.0 mL 3.0 mL 1.51 37.0 mL 7.61 71.0 mL 4.0 mL 1.56 38.0 mL 7.72 72.0 mL 5.0 mL 1.61 39.0 mL 7.84 73.0 mL 6.0 mL 1.68 40.0 mL 7.99 74.0 mL 7.0 mL 1.73 41.0 mL 8.14 75.0 mL 8.0 mL 1.79 42.0 mL 8.38 76.0 mL 9.0 mL 1.83 43.0 mL 8.85 77.0 mL 10.0 mL 1.88 44.0 mL 10.14 78.0 mL 11.0 mL 1.94 45.0 mL 11.08 79.0 mL 12.0 mL 2.01 46.0 mL 11.59 80.0 mL 13.0 mL 2.07 47.0 mL 11.83 81.0 mL 14.0 mL 2.15 48.0 mL 12.02 82.0 mL 15.0 mL 2.25 49.0 mL 83.0 mL 16.0 mL 2.32 50.0 mL 84.0 mL 17.0 mL 2.42 51.0 mL 85.0 mL 18.0 mL 2.52 52.0 mL 86.0 mL 19.0 mL 2.66 53.0 mL 87.0 mL 20.0 mL 2.82 54.0 mL 88.0 mL 21.0 mL 3.05 55.0 mL 89.0 mL 22.0 mL 3.42 56.0 mL 90.0 mL
23.0 mL 4.80 57.0 mL 91.0 mL 24.0 mL 5.77 58.0 mL 92.0 mL 25.0 mL 6.16 59.0 mL 93.0 mL 26.0 mL 6.36 60.0 mL 94.0 mL 27.0 mL 6.54 61.0 mL 95.0 mL 28.0 mL 6.67 62.0 mL 96.0 mL 29.0 mL 6.81 63.0 mL 97.0 mL 30.0 mL 6.92 64.0 mL 98.0 mL 31.0 mL 7.02 65.0 mL 99.0 mL 32.0 mL 7.12 66.0 mL 100.0 mL 33.0 mL 7.22 67.0 mL 101.0 mL Standardization of the NaOH solution Trial 1 Trial 2 Trial 3 Volume of NaOH added 18.2 17.8 18.1 Molarity of the NaOH 6.37x10^-4 1.26x10^-3 1.95x10^-3 Average Molarity of NaOH 1.28x10^-3 Standard deviation Volume of weak acid titrated (mL): 25 (mL) Volume of NaOH added pH measured Volume of NaOH added pH measured Volume of pH measured NaOH added 0.0 mL 1.52 34.0 mL 7.31 68.0 mL 1.0 mL 1.48 35.0 mL 7.42 69.0 mL 2.0 mL 1.45 36.0 mL 7.51 70.0 mL 3.0 mL 1.51 37.0 mL 7.61 71.0 mL 4.0 mL 1.56 38.0 mL 7.72 72.0 mL 5.0 mL 1.61 39.0 mL 7.84 73.0 mL 6.0 mL 1.68 40.0 mL 7.99 74.0 mL 7.0 mL 1.73 41.0 mL 8.14 75.0 mL 8.0 mL 1.79 42.0 mL 8.38 76.0 mL 9.0 mL 1.83 43.0 mL 8.85 77.0 mL 10.0 mL 1.88 44.0 mL 10.14 78.0 mL 11.0 mL 1.94 45.0 mL 11.08 79.0 mL 12.0 mL 2.01 46.0 mL 11.59 80.0 mL 13.0 mL 2.07 47.0 mL 11.83 81.0 mL 14.0 mL 2.15 48.0 mL 12.02 82.0 mL 15.0 mL 2.25 49.0 mL 83.0 mL 16.0 mL 2.32 50.0 mL 84.0 mL 17.0 mL 2.42 51.0 mL 85.0 mL 18.0 mL 2.52 52.0 mL 86.0 mL 19.0 mL 2.66 53.0 mL 87.0 mL 20.0 mL 2.82 54.0 mL 88.0 mL 21.0 mL 3.05 55.0 mL 89.0 mL 22.0 mL 3.42 56.0 mL 90.0 mL
Chemistry: Principles and Practice
3rd Edition
ISBN:9780534420123
Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Chapter16: Reactions Between Acids And Bases
Section: Chapter Questions
Problem 16.54QE
Related questions
Question
Weak Acid titration
1st Equivalence point volume of NaOH added: _______________
1st Mid-point volume of NaOH added: _______________
2nd Equivalence point volume of NaOH added: _______________
2nd Mid-point volume of NaOH added: _______________
pKa1 of the unknown weak acid: _______________
Ka1 of the unknown weak acid: _______________
pKa2 of the unknown weak acid: _______________
Ka2 of the unknown weak acid: _______________
Concentration of the unknown weak acid, [H2A]: _______________
(show your work)
Transcribed Image Text:23.0 mL
4.80
57.0 mL
91.0 mL
24.0 mL
5.77
58.0 mL
92.0 mL
25.0 mL
6.16
59.0 mL
93.0 mL
26.0 mL
6.36
60.0 mL
94.0 mL
27.0 mL
6.54
61.0 mL
95.0 mL
28.0 mL
6.67
62.0 mL
96.0 mL
29.0 mL
6.81
63.0 mL
97.0 mL
30.0 mL
6.92
64.0 mL
98.0 mL
31.0 mL
7.02
65.0 mL
99.0 mL
32.0 mL
7.12
66.0 mL
100.0 mL
33.0 mL
7.22
67.0 mL
101.0 mL
Standardization of the NaOH solution
Trial 1
Trial 2
Trial 3
Volume of NaOH added
18.2
17.8
18.1
Molarity of the NaOH
6.37x10^-4
1.26x10^-3
1.95x10^-3
Average Molarity of NaOH
1.28x10^-3
Standard deviation
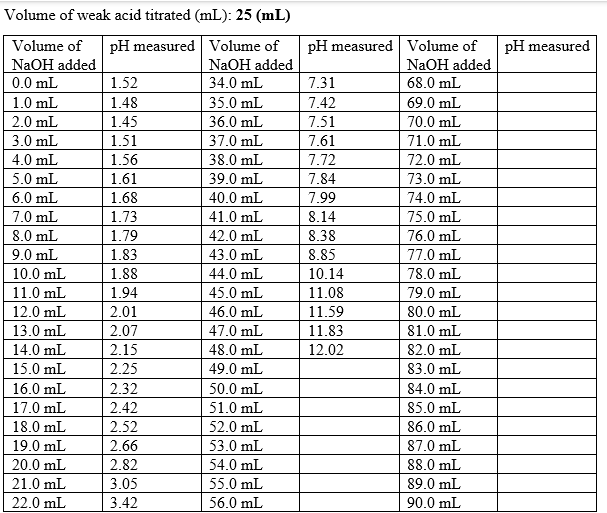
Transcribed Image Text:Volume of weak acid titrated (mL): 25 (mL)
Volume of
NaOH added
pH measured
Volume of
NaOH added
pH measured Volume of
pH measured
NaOH added
0.0 mL
1.52
34.0 mL
7.31
68.0 mL
1.0 mL
1.48
35.0 mL
7.42
69.0 mL
2.0 mL
1.45
36.0 mL
7.51
70.0 mL
3.0 mL
1.51
37.0 mL
7.61
71.0 mL
4.0 mL
1.56
38.0 mL
7.72
72.0 mL
5.0
mL
1.61
39.0 mL
7.84
73.0 mL
6.0
mL
1.68
40.0 mL
7.99
74.0 mL
7.0 mL
1.73
41.0 mL
8.14
75.0 mL
8.0 mL
1.79
42.0 mL
8.38
76.0 mL
9.0 mL
1.83
43.0 mL
8.85
77.0 mL
10.0 mL
1.88
44.0 mL
10.14
78.0 mL
11.0 mL
1.94
45.0 mL
11.08
79.0 mL
12.0 mL
2.01
46.0 mL
11.59
80.0 mL
13.0 mL
2.07
47.0 mL
11.83
81.0 mL
14.0 mL
2.15
48.0 mL
12.02
82.0 mL
15.0 mL
2.25
49.0 mL
83.0 mL
16.0 mL
2.32
50.0 mL
84.0 mL
17.0 mL
2.42
51.0 mL
85.0 mL
18.0 mL
2.52
52.0 mL
86.0 mL
19.0 mL
2.66
53.0 mL
87.0 mL
20.0 mL
2.82
54.0 mL
88.0 mL
21.0 mL
3.05
55.0 mL
89.0 mL
22.0 mL
3.42
56.0 mL
90.0 mL
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 3 images

Recommended textbooks for you

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry for Today: General, Organic, and Bioche…
Chemistry
ISBN:
9781305960060
Author:
Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry for Today: General, Organic, and Bioche…
Chemistry
ISBN:
9781305960060
Author:
Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:
Cengage Learning

Chemical Principles in the Laboratory
Chemistry
ISBN:
9781305264434
Author:
Emil Slowinski, Wayne C. Wolsey, Robert Rossi
Publisher:
Brooks Cole


Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning