
Chemistry & Chemical Reactivity
10th Edition
ISBN: 9781337399074
Author: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 17, Problem 35PS
Using Figure 17.11, suggest an indicator to use in each of the following titrations:
- (a) The weak base pyridine is titrated with HCl.
- (b) Formic acid is titrated with NaOH.
- (c) Ethylenediamine, a weak diprotic base, is titrated with HCl.
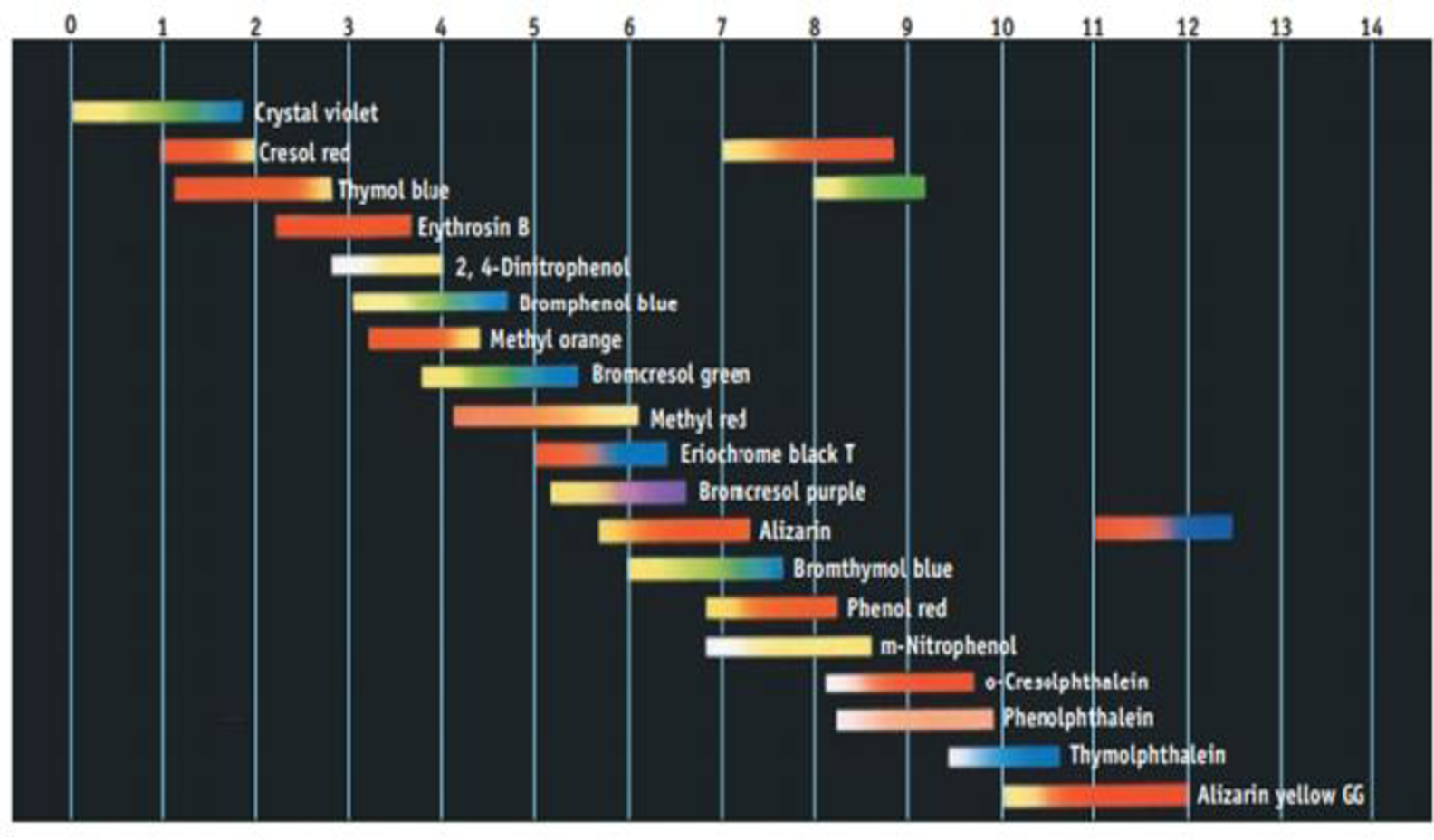
Figure 17.11 Common acid-base indicators. The color changes occur over a range of pH values. Notice that o few indicators hove color changes over two different pH ranges.
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
An approximately 0.1-molar solution of NaOH is to be standardized by titration. Assume that the following materials are available.
Clean, dry 50 mL buret •Analytical balance
250 mL Erlenmeyer flask •Wash bottle filled with distilled water
Phenolphthalein indicator solution
Potassium hydrogen phthalate, KHP, a solid monoprotic acid (to be used as the primary standard)
Briefly describe the steps you would take, using the materials listed above, to standardize the NaOH solution.
Describe (the set up) the calculations necessary to determine the concentration of the NaOH solution.
The potentiometric titration of a weak acid against a stong base is shown below. For A) calculate the pH at different stages of the titration using the appropriate chemical equation & formula and then select the appropriate answer. Example of answer: Between pH 1 and 2. A.) - Between what is the pH of the solution before any strong base has been added? - Between what is the pH of the solution after 10.00mL of strong base has been added? - Between what is the pH of the solution after 25.00mL of strong base has been added? - Between what is the pH of the solution after 40.00mL of strong base has been added? - Between what is the pH of the solution after 50.00mL of strong base has been added? - Between what is the pH of the solution after 60.00mL of strong base has been added? B.) Give the name of another weak acid that will have a similar titration curve. Briefly explain the reason for the similarity.
Define the term selective precipitation?
Chapter 17 Solutions
Chemistry & Chemical Reactivity
Ch. 17.1 - You have a 0.30 M solution of formic acid (HCO2H)...Ch. 17.2 - What is the pH of a buffer solution composed of...Ch. 17.2 - Use the Henderson-Hasselbalch equation to...Ch. 17.2 - Using an acetic acid/sodium acetate buffer...Ch. 17.2 - Calculate the pH of 0.500 L of a buffer solution...Ch. 17.3 - The titration of 0.100 M acetic acid with 0.100 M...Ch. 17.3 - Calculate the pH after 75.0 mL of 0.100 M HO has...Ch. 17.4 - The barium ion concentration, [Ba2+], in a...Ch. 17.4 - Calculate the solubility of AgCN in moles per...Ch. 17.4 - Calculate the solubility of Ca(OH)2 in moles per...
Ch. 17.4 - Calculate the solubility of BaSO4 (a) in pure...Ch. 17.5 - Solid Pbl2 (Ksp = 9.8 109) is placed in a beaker...Ch. 17.5 - Prob. 17.13CYUCh. 17.5 - Prob. 17.14CYUCh. 17.6 - Silver nitrate (0.0050 mol) is added to 1.00 L of...Ch. 17.6 - Calculate the value of the equilibrium constant,...Ch. 17.6 - Prob. 1.1ACPCh. 17.6 - What is the minimum volume of 0.0071 M NaCN(aq)...Ch. 17.6 - Use the formation constant of [Au(CN)2] in...Ch. 17.6 - Silver undergoes similar reactions as those shown...Ch. 17.6 - Write a balanced chemical equation for the...Ch. 17.6 - Phosphate ions are abundant in cells, both as the...Ch. 17.6 - A typical total phosphate concentration in a cell,...Ch. 17 - Does the pH of the solution increase, decrease or...Ch. 17 - Does the pH of the solution increase, decrease, or...Ch. 17 - What is the pH of a solution that consists of 0.20...Ch. 17 - What is the pH of 0.15 M acetic acid to which 1.56...Ch. 17 - What is the pH of the solution that results from...Ch. 17 - What is the pH of the solution that results from...Ch. 17 - What is the pH of the buffer solution that...Ch. 17 - Lactic acid (CH3CHOHCO2H) is found in sour milk,...Ch. 17 - What mass of sodium acetate, NaCH3CO2, must he...Ch. 17 - What mass of ammonium chloride, NH4Cl, must be...Ch. 17 - Calculate the pH of a solution that has an acetic...Ch. 17 - Calculate the pH of a solution that has an...Ch. 17 - What must the ratio of acetic acid to acetate ion...Ch. 17 - What must the ratio of H2PO4 to HPO42 be to have a...Ch. 17 - A buffer is composed of formic acid and its...Ch. 17 - A buffer solution is composed of 1.360 g of KH2PO4...Ch. 17 - Which of the following combinations would be the...Ch. 17 - Which of the following combinations would be the...Ch. 17 - Describe how to prepare a buffer solution from...Ch. 17 - Describe how to prepare a buffer solution from NH3...Ch. 17 - Determine the volume (in mL) of 1.00 M NaOH that...Ch. 17 - Determine the volume (in mL) of 1.00 M HC1 that...Ch. 17 - A buffer solution was prepared by adding 4.95 g of...Ch. 17 - You dissolve 0.425 g of NaOH in 2.00 L of a buffer...Ch. 17 - A buffer solution is prepared by adding 0.125 mol...Ch. 17 - What is the pH change when 20.0 mL of 0.100 M NaOH...Ch. 17 - Phenol, C6H5OH, is a weak organic acid. Suppose...Ch. 17 - Assume you dissolve 0.235 g of the weak acid...Ch. 17 - You require 36.78 mL of 0.0105 M HCl to reach the...Ch. 17 - A titration of 25.0 mL of a solution of the weak...Ch. 17 - Without doing detailed calculations, sketch the...Ch. 17 - Without doing detailed calculations, sketch the...Ch. 17 - You titrate 25.0 mL of 0.10 M NH3 with 0.10 M HCl....Ch. 17 - Using Figure 17.11, suggest an indicator to use in...Ch. 17 - Using Figure 17.11, suggest an indicator to use in...Ch. 17 - Name two insoluble salts of each of the following...Ch. 17 - Prob. 38PSCh. 17 - Using the solubility guidelines (Figure 3.10),...Ch. 17 - Predict whether each of the fallowing is insoluble...Ch. 17 - For each of the following insoluble salts, (1)...Ch. 17 - Prob. 42PSCh. 17 - When 1.55 g of solid thallium(I) bromide is added...Ch. 17 - At 20 C, a saturated aqueous solution of silver...Ch. 17 - When 250 mg of SrF2, strontium fluoride, is added...Ch. 17 - Calcium hydroxide, Ca(OH)2, dissolves in water to...Ch. 17 - You add 0.979 g of Pb(OH)2 to 1.00 L of pure water...Ch. 17 - You place 1.234 g of solid Ca(OH)2 in 1.00 L of...Ch. 17 - Estimate the solubility of silver iodide in pure...Ch. 17 - What is the molar concentration of Au+(aq) in a...Ch. 17 - Prob. 51PSCh. 17 - Estimate the solubility of lead(II) bromide (a) in...Ch. 17 - The Ksp value for radium sulfate, RaSO4, is 4.2 ...Ch. 17 - If 55 mg of lead(II) sulfate is placed in 250 mL...Ch. 17 - Prob. 55PSCh. 17 - Prob. 56PSCh. 17 - Calculate the molar solubility of silver...Ch. 17 - Calculate the solubility of silver bromide, AgBr,...Ch. 17 - Compare the solubility, in milligrams per...Ch. 17 - What is the solubility, in milligrams per...Ch. 17 - Calculate the solubility, in moles per liter, of...Ch. 17 - Calculate the solubility, in moles per liter, of...Ch. 17 - Which insoluble compound in each pair should be...Ch. 17 - Which compound in each pair is more soluble in...Ch. 17 - You have a solution that has a lead(II) ion...Ch. 17 - Sodium carbonate is added to a solution in which...Ch. 17 - If the concentration of Zn2+ in 10.0 mL of water...Ch. 17 - You have 95 mL of a solution that has a lead(II)...Ch. 17 - Prob. 69PSCh. 17 - Will a precipitate of Mg(OH)2 form when 25.0 mL of...Ch. 17 - Zinc hydroxide is amphoteric (Section 16.10). Use...Ch. 17 - Solid silver iodide, AgI, can be dissolved by...Ch. 17 - What amount of ammonia (moles) must be added to...Ch. 17 - Can you dissolve 15.0 mg of AuCl in 100.0 mL of...Ch. 17 - What is the solubility of AgCl (a) in pure water...Ch. 17 - Prob. 76PSCh. 17 - Prob. 77GQCh. 17 - Prob. 78GQCh. 17 - Prob. 79GQCh. 17 - Calculate the hydronium ion concentration and the...Ch. 17 - Calculate the hydronium ion concentration and the...Ch. 17 - For each of the following cases, decide whether...Ch. 17 - Prob. 83GQCh. 17 - A sample of hard water contains about 2.0 103 M...Ch. 17 - What is the pH of a buffer solution prepared from...Ch. 17 - Prob. 86GQCh. 17 - Describe the effect on the pH of the following...Ch. 17 - What volume of 0.120 M NaOH must be added to 100....Ch. 17 - A buffer solution is prepared by dissolving 1.50 g...Ch. 17 - What volume of 0.200 M HCl must be added to 500.0...Ch. 17 - What is the equilibrium constant for the following...Ch. 17 - Calculate the equilibrium constant for the...Ch. 17 - Prob. 93GQCh. 17 - The solubility product constant for calcium...Ch. 17 - In principle, the ions Ba2+ and Ca2+ can be...Ch. 17 - A solution contains 0.10 M iodide ion, I, and 0.10...Ch. 17 - A solution contains Ca2+ and Pb2+ ions, both at a...Ch. 17 - Prob. 98GQCh. 17 - Prob. 99GQCh. 17 - Prob. 100GQCh. 17 - Each pair of ions below is found together in...Ch. 17 - Each pair of ions below is found together in...Ch. 17 - The cations Ba2+ and Sr2+ can be precipitated as...Ch. 17 - You will often work with salts of Fe3+, Pb2+, and...Ch. 17 - Aniline hydrochloride, (C6H5NH3)Cl, is a weak...Ch. 17 - The weak base ethanolamine. HOCH2CH2NH2, can be...Ch. 17 - For the titration of 50.0 mL of 0.150 M...Ch. 17 - A buffer solution with it pH of 12.00 consists of...Ch. 17 - To have a buffer with a pH of 2.50, what volume of...Ch. 17 - What mass of Na3PO4 must be added to 80.0 mL of...Ch. 17 - You have a solution that contains AgNO3, Pb(NO3)2,...Ch. 17 - Prob. 112ILCh. 17 - Suggest a method for separating a precipitate...Ch. 17 - Prob. 114SCQCh. 17 - Prob. 115SCQCh. 17 - Two acids, each approximately 0.01 M in...Ch. 17 - Composition diagrams, commonly known as alpha...Ch. 17 - The composition diagram, or alpha plot, for the...Ch. 17 - The chemical name for aspirin is acetylsalicylic...Ch. 17 - Prob. 120SCQ
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Consider the titration of HF (K a=6.7104) with NaOH. What is the pH when a third of the acid has been neutralized?arrow_forwardSketch the general appearance of the curve for the titration of a weak acid with a strong base. Explain (in words) what chemistry governs the pH in each of the four distinct regions of the curve.arrow_forwardFor a titration to be effective, the reaction must be rapid and the yield of the reaction must essentially be 100%. Is Kc > 1, < 1, or ≈ 1 for a titration reaction? Kc is the product of all reaction products (choose from: Multiplied by, added to, divided by, subtracted from) the product of the reactants, with all concentrations of reactants and products raised to their respective stoichiometric powers. As the titration proceeds, the amount of reactants (choose from: Increases, decreases, remains the same) and the amount of products (choose from: Increases, decreases, remains the same). Because the value of the numerator is (Choose from much greater than, much less than, approximately equal to) the value of the denominator, the value of Kc will be (Choose from much greater than, much less than, approximately equal to)1 for an effective titration reaction.arrow_forward
- Explain the pH response of pure water as a buffer to additions of strong acid or basesarrow_forwardAt what point in the titration of a weak base with a strong acid is the maximum buffer capacity reached? This is the point at which a given small addition of acid causes the least pH change.arrow_forwardDescribe the changes in pH that take place during the titration of: (a) a weak acid with a strong base, (b) a weak base with a strong acid .arrow_forward
- For each of the statements, identify the appropriate key point (I, II, III, IV, or V) in the titration.arrow_forwardA student carried out an acid-base titration by adding NaOH solution from a buret to an Erlenmeyer flask containing HCl solution and using phenolphthalein as an indicator. At the equivalence point, she observed a faint reddish-pink color. However, after a few minutes, the solution gradually turned colorless. What do you suppose happened? Explain.arrow_forwardDefine the following terms: titration equivalence point buffer point molarity What instrument are you going to use in this experiment to measure the pH? 3 Give the color of phenolphthalein indicator at the end point for this study Calculate the pH of a 0.10M weak acid, HA, whose Ka is 1.8 X 10arrow_forward
- Which of the following acid-base titrations initially forms a buffer? lactic acid titrated with potassium hydroxide sodium hydroxide titrated with ascorbic acid sulfuric acid titrated with ammonia all these initially form a bufferarrow_forwardA student decided not to filter the saturated calcium hydroxide solution prior to titration with HCl. How does this affect the calculation of the concentration of calcium hydroxide?arrow_forwardWhat is the difference between endpoint and equivalence point in acid-base titration?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemical Principles in the LaboratoryChemistryISBN:9781305264434Author:Emil Slowinski, Wayne C. Wolsey, Robert RossiPublisher:Brooks Cole
Chemical Principles in the LaboratoryChemistryISBN:9781305264434Author:Emil Slowinski, Wayne C. Wolsey, Robert RossiPublisher:Brooks Cole Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:9781305079113
Author:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:9780534420123
Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:Cengage Learning

Chemical Principles in the Laboratory
Chemistry
ISBN:9781305264434
Author:Emil Slowinski, Wayne C. Wolsey, Robert Rossi
Publisher:Brooks Cole

Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning
General Chemistry | Acids & Bases; Author: Ninja Nerd;https://www.youtube.com/watch?v=AOr_5tbgfQ0;License: Standard YouTube License, CC-BY