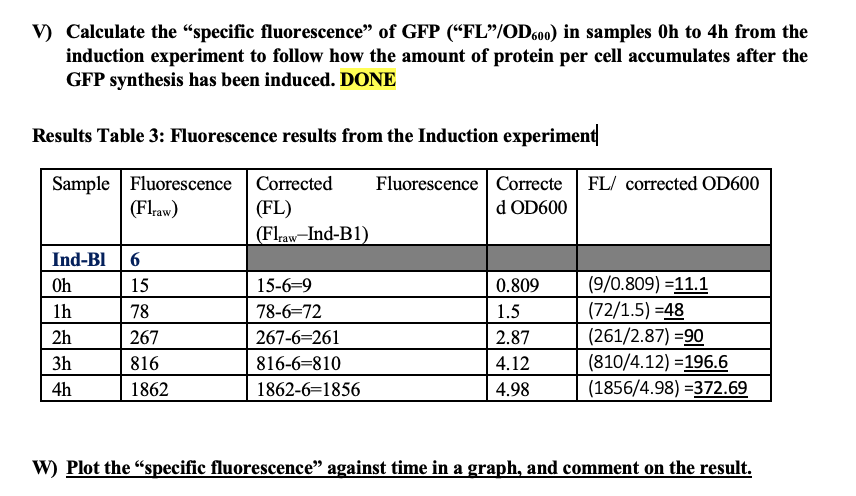
W) Plot the "specific fluorescence” against time in a graph, and comment on the result.
Q: State the steps of beta-oxidation pathway. Please make sure to state all the enzymes and co-factors…
A: Both prokaryotic cells' cytoplasm and the mitochondria of eukaryotic cells use the metabolic pathway…
Q: How many net ATP would be produced in these liver cells from the complete oxidation of 1 molecule of…
A: The ATP synthase is an enzyme present in the inner membrane and cristae of mitochondria. It is a…
Q: What are 5 regulation of carbohydrate metabolism in the liver
A: The liver controls carbohydrate metabolism and glucose homeostasis. Understanding liver carbohydrate…
Q: What type of signal transduction is involved in signaling from hormones such as estrogen and…
A: Hormones are signalling biomolecules that are released from endocrine cells into the bloodstream and…
Q: b. Please draw the structure of the polypeptide: threonine-lysine-glutamine-valine at pH 7.0, and…
A: Amino acid sequences are written with N-terminal amino acid on the left and C-terminal amino acid on…
Q: Can the fatty acid B be used to generate glucose in animals and is this possible for an unbranched…
A: Beta oxidation: The enzyme fatty acyl-CoA synthase (FACS) adds a CoA group to the fatty acid chain…
Q: Draw the structure and titration curve of Cysteine by hand. Make sure to label the x- and y-axes,…
A: Amino acid has a typical structure. It has a central Carbon atom called (Cα). To it 4 groups are…
Q: Explain the difference in the results obtained regaarding the action of amylase on starch? Explain…
A: When amylase acts on starch, it initiates a series of chemical reactions that lead to the breakdown…
Q: What is the indication of violet blue color in biuret test?
A: Proteins, essential macromolecules in living organisms, play crucial roles in various biological…
Q: Is Raffinose tri-saccharide a reducing sugar? Explain the chemistry of the reducing sugar test and…
A: Raffinose is a trisaccharide consisting of galactose, glucose, and fructose as its constituent sugar…
Q: In which of the following metabolic conversions is ATP “generated” during glycolysis?…
A:
Q: A 32-year-old woman was admitted to the hospital following 2½ days of severe vomiting. Before this…
A: The maintenance of the body's numerous physiological processes depends heavily on minerals. They are…
Q: (a) H* transport from the outside of the IMM into the matrix drives this process. Assume the pH…
A: pH of inside the matrix=8.0 Concentration of H+ ion inside the matrix=1.0 A-10-8.0 pH outside the…
Q: Why is this compound achiral?
A: A plane of symmetry in the molecular framework of 1-chloro-3-methylcyclobutane causes it to be…
Q: Based on this information and patient history you have concerns of alcohol use disorder and…
A: In order to understand how alcohol consumption along with irregular intake of food leads to alcohol…
Q: In glycogen breakdown: i.Phosphorylase cleaves alpha(1-->4) glycosidic bonds yielding glucose…
A: Glycogen breakdown, also known as glycogenolysis, is the process by which glycogen is broken down…
Q: Oxidative decarboxylations— involve loss of CO2 and the production of NADH. do not occur…
A: INTRODUCTION: Oxidation response where the carboxyl molecule is eliminated as carbon dioxide.…
Q: Give typed full explanation not a single word hand written otherwise leave it
A: The basic metabolic route known as the tricarboxylic acid (TCA) cycle, sometimes referred to as the…
Q: 15.18) Determine whether the following statements describe insulin, glucagon, or both insulin and…
A: Glycogenolysis is the breakdown of glycogen, a complex carbohydrate that is stored in the liver and…
Q: Over time (a lot of it!) mutations in metabolic enzymes can occur to create versions with different…
A: Glycolysis is a process wherein glucose molecules are metabolized to form pyruvate in a series of…
Q: Show how to bring about the following chemical transformation. This transformation might require one…
A:
Q: Which of the following fatty acids is most likely to be a solid at room temperature? OA) B) C) D) OH…
A: Fatty acids are carboxylic acids with a hydrocarbon chain ranging from 4 carbon to 36 carbons. The…
Q: Describe the oxidative and non oxidative phases of Pentose phosphate pathways discusss how the…
A: The pentose phosphate pathway (PPP) is a metabolic pathway that occurs in the cytoplasm of cells. It…
Q: Atp bookkeeping. Explain where the number of 38 and 32 atp per glucose molecules comes from
A: Glucose oxidation is the process of breaking down of glucose completely through the process…
Q: What phosphate buffer conditions can be used to isolate protein (isoelectric point of 5.2) using…
A: Anion Exchange Chromatography is a type of ion exchange chromatography wherein the resin present…
Q: Draw the schematic diagram of the protein purification through hydrophobic column chromatography and…
A: Hydrophobic Column Chromatography or Hydrophobic Interaction Chromatography (HIC) is a technique…
Q: rate of transport Vmax 1/2Vmax transporter-me ted diffusion Km simple diffusion concentration of…
A: Diffusion across a membrane is the process by which a substance moves from a region of higher…
Q: Consider analysis of a mixture of saturated and unsaturated triacylglycerols (fish oil) and/or fatty…
A: Fatty acids are carboxylic acids with a hydrocarbon chain ranging from 4 carbon to 36 carbons. They…
Q: C₂ 200 Ketone R-C=O Aldehydes H-C=O Ester RO-C=O Carboxylic Acid HO-C=O Choose the Carbons HH I…
A: The graph is that of 13C NMR. The X-axis gives the chemical shift value in ppm. Every functional…
Q: 9. In sugars that contain many chiral centers, only the chiral center that is most distant from the…
A: Carbohydrates are polyhydroxy aldehydes or ketones. They can be classified as monosaccharides,…
Q: Question 1 How many net ATP are made per glucose during glycolysis? O a.0 O b. 1 OC. 2 O d.5 O e. 8…
A: Glycolysis is a catabolic pathway and first step in cellular respiration in which a glucose molecule…
Q: Hey could someone please help me with this human biochem questions 1. Describe the process of…
A: Dietary lipid metabolism is a complex process that breaks complex lipid molecules into smaller…
Q: 2. How are single, double and triple covalent bonds different?
A: Chemical bonds stabilize and form molecules. Covalent chemical bonds are crucial. Atoms share…
Q: Objective: Becoming familiar with the purification and kinetic analysis of an enzyme that produces a…
A: Electrophoresis means the migration of a charged particle under the influence of an applied electric…
Q: Mutations can be classified as small-scale or large-scale, depending on the number of nucleotides…
A: a) change from CCGATG to CCGGATGThe sequence is altered from CCGATG to CCGGATG by the insertion of…
Q: 4. Consider the following sequence of nine amino acid residues taken from a larger polypeptide chain…
A: We know that the polypeptide chain has a N-terminal and a C-terminal. The N-terminal is a free…
Q: 20. Under a normal metabolic rate which of the following compounds is the most energy rich?…
A: The basal metabolic rate (BMR) is the amount of energy needed while resting in a temperate…
Q: What are the 3 chemical buffer systems ? in which part of the body is each buffer the most important
A: Buffer systems in the body are essential for maintaining a constant pH in the blood and other body…
Q: From the graphs given, can you find the Km from the Michaelis-Menten curve and the Lineweaver-Burke…
A: For a one-substrate enzyme-catalyzed reaction, the Michaelis-Menton equation shows the quantitative…
Q: In the pathway for degradation of BCAAs, which reaction takes place prior to the action of the BCKDC…
A: There are three branched chain amino acids. They are valine, leucine and isoleucine. They are called…
Q: Identify the following electron transport intermediate. H : O : O : H A.…
A: Electron transport refers to the movement of electrons through a series of membrane-bound protein…
Q: a. Use three-letter amino acid abbreviations to write the representation for the tripeptide shown…
A: The four types of biological macromolecules are proteins, nucleic acids, lipids and carbohydrates.…
Q: write the reactions using structural formulas by which a 7-carbon fatty acid is activated and…
A: The 7 carbon fatty acid is first activated to its acyl-CoA form. Then it undergoes beta-oxidation to…
Q: A fragment of a protein commonly found in food was sequenced and found to have the sequence: RDCEHKY…
A: The four types of biological macromolecules are proteins, nucleic acids, lipids and carbohydrates.…
Q: Select the correct statement. Upon digestion, fatty O amino acids O acetone O acetyl COA O…
A: The four types of biological macromolecules are proteins, nucleic acids, carbohydrates and lipids.…
Q: You are fresh recruits to a molecular biology laboratory, and your new boss has tasked you to study…
A: Polymerase chain reaction is abbreviated PCR. PCR is a laboratory technique for rapidly producing…
Q: -fatty acid synthesis RNA -> DNA Necessary to make RNA nucleotide G3P F6P &
A: Glucose-6-phosphate is a sugar molecule that is an important intermediate in several metabolic…
Q: What is a Polyglycolic acid
A: Polyglycolic acid: Polyglycolic acid (PGA) is a synthetic biomaterial that belongs to the family of…
Q: A set of biomolecules listed in the table at right are in solution at pH 6.8, when they are passed…
A: Ultrafiltration is a technique similar to dialysis where the sample containing a mixture of proteins…
Q: Explain in one statement/phrase why the base sequence ATC could not be a codon.
A: As per the central dogma of molecular biology, genetic information flows from DNA →RNA →Protein.…
HEEELP -ME with this exersice please.
I dont understand which values I should use for plotting the graph
Step by step
Solved in 3 steps with 1 images

- pls explain Increasing the saturation of the ammonium sulfate is a prerequisite in isolating a target protein that is rich in Cys and Tyr residues. Which of the following techniques should be considered in accurately quantifying the isolated protein?I. Running the isolated protein in a dialysis or GFC set up.II. Using Biuret or BCA assay as the colorimetric quantitation method.III. Using Bradford or Lowry assay as the colorimetric quantitation method.A. I onlyB. II onlyC. I and IIID. I, II and III. Bradford Assay is most suitable to use when the extraction buffer is below the target protein’s pI. This is so because the protein would be morea. Positively charged allowing the CBB G-250 dye to bind via its sulfonate groups.b. Negatively charged allowing the CBB G-250 dye to bind via its sulfonate groups.c. Neutrally charged allowing the CBB G-250 dye to bind via its sulfonate groups.d. Zwitterionic allowing the CBB G-250 dye to bind via its sulfonate groups.The movements of single motor-protein moleculescan be analyzed directly. Using polarized laser light, it ispossible to create interference patterns that exert a cen-trally directed force, ranging from zero at the center to afew piconewtons at the periphery (about 200 nm from thecenter). Individual molecules that enter the interferencepattern are rapidly pushed to the center, allowing them tobe captured and moved at the experimenter’s discretion.Using such “optical tweezers,” single kinesin mol-ecules can be positioned on a microtubule that is fixed toa coverslip. Although a single kinesin molecule cannotbe seen optically, it can be tagged with a silica bead andtracked indirectly by following the bead (Figure Q16–3A).In the absence of ATP, the kinesin molecule remains at thecenter of the interference pattern, but with ATP it movestoward the plus end of the microtubule. As kinesin movesalong the microtubule, it encounters the force of the inter-ference pattern, which simulates the load…Beer's Law to determine Protein Concentration You have purified a recombinant form of the p53 protein from E. coli and determined the A280 to be1.35. Calculate the molar and mass concentration of the purified protein if the extinction coefficientand molecular weight of p53 is 35,410 M-1 cm-1 and 43,653 Da, respectively (l = 1 cm).
- In Multi-Column Purification of rGFP. What happens to the protein amount, protein purity, and/or specific activity of a purification fraction if one of the three is changed? (i.e. understand the relationship between the three.)Pls explain If the voltage were NOT increased at the start of the resolving gel run, the overall experiment time will ______, while the calculated electrophoretic mobilities will ______.a. increase, increaseb. increase, decreasec. decrease, increased. decrease, decreasePick all that are TRUE regarding analysis of quaternary structures of proteins using polyacrylamide electrophoresis:I. The added β-mercaptoethanol disrupts S--S bonds bridging the polypeptide chains causing the appearance of higher Rf bands compared to the native protein run. II. Heating up any protein before subjecting to SDS-PAGE will always result in the formation of more than one band.III. A good asymmetrical gel layout would be : (Lane 1) MW ladder, (2) native protein, (3) protein + β-ME, (4) protein + HCL, (5) protein + β-ME + HCl.IV. Formation of a single band in the protein + β-ME + HCl run, whose Rf is lower than the native run, could be indicative that the protein is a homodimer.A. I onlyB. I and IIC. II and…Fluorescence-activated cell sorting (FACS) is a powerful technique for separating cells according to their content of particular molecules. For example, a fluorescence-labeled antibody specific for a cell-surface protein can be used to detect cells containing such a molecule. Suppose that you want to isolate cells that possess a receptor enabling them to detect bacterial degradation products. However, you do not yet have an antibody directed against this receptor. Which fluorescence-labeled molecule would you prepare to identify such cells?
- Using the pKa data in as shown and the Henderson-Hasselbalch equation,calculate the approximate net charge on each of the four common ribonucleoside 5′-monophosphates (rNMPs) at pH 3.8. If a mixture of these rNMPs was placed in an electrophoresis apparatus, as shown, draw four bands to predict the direction and relative migration rate of each.b. Compounds A, B, C, and D are known to be intermediates in the pathway for production of protein E. To determine where the block in protein-E production occurred in each individual, the various intermediates were given to each individuals cel Is in culture. After a few weeks of growth with the intermediate, the cells were assayed for the production of protein E. The results for each individuals cells are given in the following table. A plus sign means that protein E was produced after the cells were given the intermediate listed at the top of the column. A minus sign means that the cells still could not produce protein E even after being exposed to the intermediate at the top of the column. Denote the point in the pathway in which each individual is blocked.a. Compounds A, B, C, and D are known to be intermediates in the pathway for production of protein E. To determine where the block in protein-E production occurred in each individual, the various intermediates were given to each individuals cel Is in culture. After a few weeks of growth with the intermediate, the cells were assayed for the production of protein E. The results for each individuals cells are given in the following table. A plus sign means that protein E was produced after the cells were given the intermediate listed at the top of the column. A minus sign means that the cells still could not produce protein E even after being exposed to the intermediate at the top of the column. Draw the pathway leading to the production of protein E.
- A chain of biochemical events is responsible for Aequorea victoria turning green. Firstly, the protein aequorin converts chemical energy into blue light. A second protein, known as GFP (green fluorescent protein), absorbs the light. Aequorin is a 21 kDa protein with a pI of 4.5 that generates blue light from an attached non-protein chromophore. GFP is a 27 kDa protein with a pI of 6.2. In order for it to fluoresce, 3 amino acids undergo a remarkable reaction when they are folded in the right way. Aequorin does not absorb light in the visible range, and GFP absorbs light at 395 nm and 475 nm. 1. Sketch what you would see if you ran a mixture of aequorin and GFP over a size-exclusion column. In your sketch, include absorbance data at 280, 395, and 475 nm.Make a table with a scale of absorbance and the concentration of protein in Chromatin sample from the following data for excel graph Absorbance=660nm following data are of tubes with concern A =0 B=0.036 C=0.011 D=0.001 E=0.027 F=0.020 G=0.032 H1=0.176 H2=0.183 I1=0.150 I2=0.171 also plot the graph??In a pUC19 digest for 1 ug of pUC19 (DNA conc. 282ng/ul) using 10X Cutsmart buffer, pure water and BamHI enzyme in a total volume of 40 ul, how much of each solution is added to the total volume?

