What amino acids will be positive at a pH of 2? Use the pKa table
Human Heredity: Principles and Issues (MindTap Course List)
11th Edition
ISBN:9781305251052
Author:Michael Cummings
Publisher:Michael Cummings
Chapter9: Gene Expression And Gene Regulation
Section: Chapter Questions
Problem 15QP
Related questions
Question
What amino acids will be positive at a pH of 2? Use the pKa table
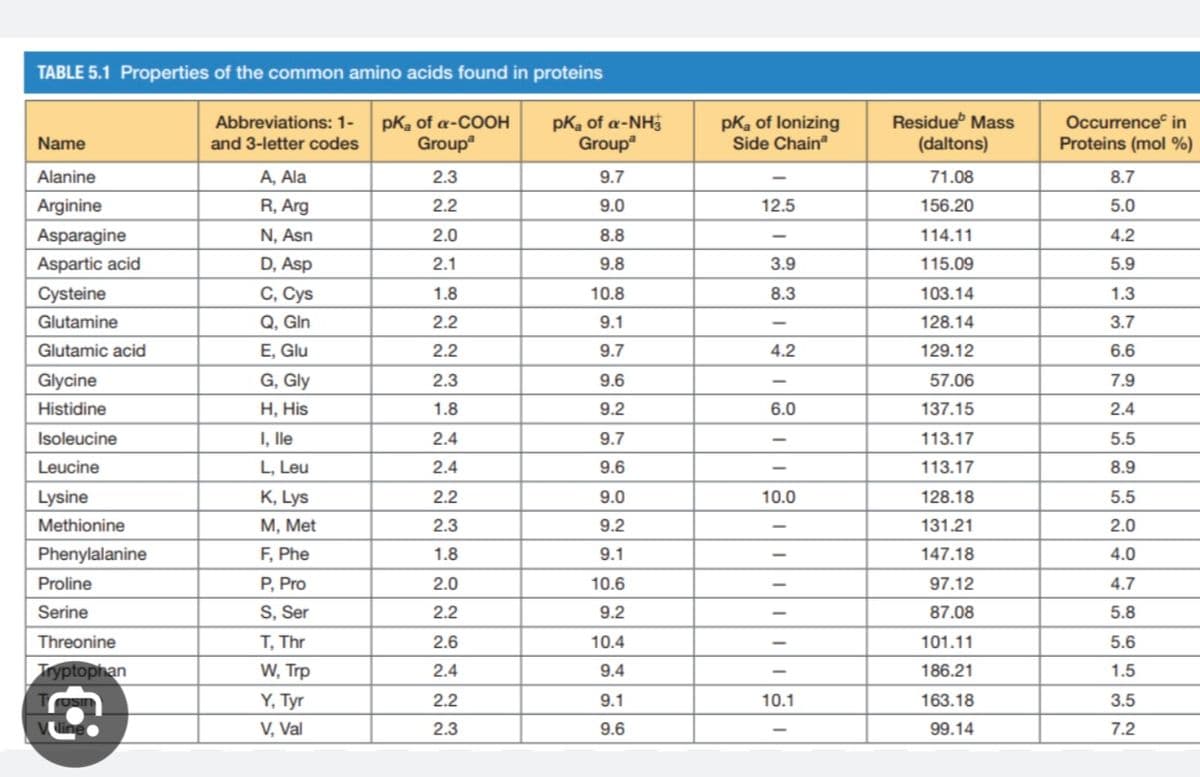
Transcribed Image Text:TABLE 5.1 Properties of the common amino acids found in proteins
pK₂ of a-COOH
Groupª
Name
Alanine
Arginine
Asparagine
Aspartic acid
Cysteine
Glutamine
Glutamic acid
Glycine
Histidine
Isoleucine
Leucine
Lysine
Methionine
Phenylalanine
Proline
Serine
Threonine
Tryptophan
Abbreviations: 1-
and 3-letter codes
A, Ala
R, Arg
N, Asn
D, Asp
C, Cys
Q, Gin
E, Glu
G, Gly
H, His
I, lle
L, Leu
K, Lys
M, Met
F, Phe
P, Pro
S, Ser
T, Thr
W, Trp
Y, Tyr
V, Val
2.3
2.2
2.0
2.1
1.8
2.2
2.2
2.3
1.8
2.4
2.4
2.2
2.3
1.8
2.0
2.2
2.6
2.4
2.2
2.3
pK₂ of a-NH3
Groupª
9.7
9.0
8.8
9.8
10.8
9.1
9.7
9.6
9.2
9.7
9.6
9.0
9.2
9.1
10.6
9.2
10.4
9.4
9.1
9.6
pK₂ of lonizing
Side Chainª
12.5
3.9
8.3
4.2
6.0
10.0
10.1
Residue Mass
(daltons)
71.08
156.20
114.11
115.09
103.14
128.14
129.12
57.06
137.15
113.17
113.17
128.18
131.21
147.18
97.12
87.08
101.11
186.21
163.18
99.14
Occurrence in
Proteins (mol %)
8.7
5.0
4.2
5.9
1.3
3.7
6.6
7.9
2.4
5.5
8.9
5.5
2.0
4.0
4.7
5.8
5.6
1.5
3.5
7.2
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 4 steps

Recommended textbooks for you

Human Heredity: Principles and Issues (MindTap Co…
Biology
ISBN:
9781305251052
Author:
Michael Cummings
Publisher:
Cengage Learning

Human Heredity: Principles and Issues (MindTap Co…
Biology
ISBN:
9781305251052
Author:
Michael Cummings
Publisher:
Cengage Learning