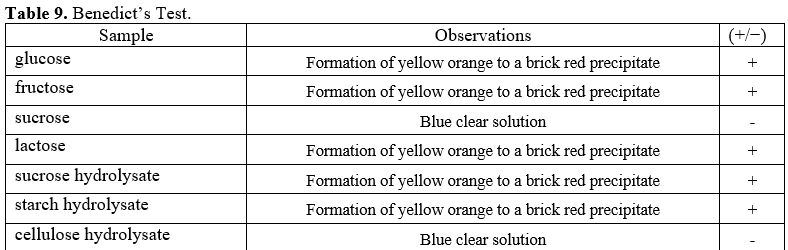
What inferences can be made from the Benedict’s test of the hydrolysates of the following? Was the acid treatment able to hydrolyze the sugars? a. sucrose b. starch c. cellulose
Q: Which of the following is not a saturated fatty acid? A. Linoleic acid B. Caproic acid C. Myristic…
A: Fatty acid can be classified into saturated fatty acid and unsaturated fatty acid.
Q: Can the iodine test distinguish between the following carbohydrates? Explain your answer a)…
A: Introduction- Carbohydrates are simple sugars that contain hydrogen, carbon, and oxygen atoms in…
Q: What is the difference between a carbohydrate and sugar? Give a simple explanation.
A: Carbohydrates are the essential macronutrients that contain carbon (C), hydrogen (H), and oxygen (O)…
Q: What happens to carbohydrates when reacted with strong HCl?
A: Carbohydrates are polyhdroxyaldehyde or polyhydroxyketone or componds which produce these products…
Q: Glucose and fructose are reducing sugars. Sucrose, or table sugar, is a disaccharide consisting of…
A: Carbohydrates are the energy-producing biomolecules commonly known as sugars that are can be…
Q: Hexoses are six-carbon sugars; examples include glucose, fructose, and galactose.
A: BASIC INFORMATION BIOMOLECULES These are the molecules that are made up of the biological elements…
Q: Which of the following is not a carbohydrate? А. С. надон новон ОН НО на на во B. D. НО НО ОН ОН ОН
A: INTRODUCTION Carbohydrates : These are biomolecules contains carbon, hydrogen and oxygen.
Q: An example of a polysaccharide is O A. fructose O B. glucose O C. galactose O D. cellulose
A: Introduction :- The most prevalent type of carbohydrates in food are polysaccharides, often known as…
Q: What difference can you observe in the iodine test for glycogen and starch? Explain the difference
A: Carbohydrates are one of the main organic molecules required by the body for proper functioning and…
Q: In the following reaction the product(s) would be: CH3CH₂COOH + CH3OH → O a) an aldehyde and water…
A: Here we can see there are two alcohols reacting together. This is a kind of dehydration reaction.
Q: Compare/contrast the structures of the following carbohydrate pairs: (a) D-ribose and D-ribulose…
A: Carbohydrates or carbs are macronutrient consisting of Carbon, hydrogen and oxygen atoms. In nature…
Q: Starch and glycogen are both composed of glucose monomers. What is/are the differences between the…
A: Starch and glycogen both are polysaccharides and are made up of glucose monomer, general Function of…
Q: Polyhydroxy aldehydes and ketones are
A: Polyhydroxy aldehydes and ketones are
Q: A. Biuret Test Mix 1 mL of sample with 1 mL of 5% NaOH. Add 2 – 3 drops of 1% CuSO4. Question…
A: Biuret test is done to check for the presence for proteins. CuSO4 and NaOH are used as reagents.…
Q: What is the role of fats as nonpolar and a source of enrgy?
A: Lipids are organic substances that are insoluble in water and soluble in organic solvents. Fats are…
Q: A. Is glucose an aldose or ketose sugar? B. Based on the number of carbons glucose has, what is…
A: Sugars or carbohydrates are the most abundant biopolymers in living organisms. Carbohydrates are…
Q: A solution of starch at room temperature does not readily decompose to form a solution of simple…
A: Starch is a form of carbohydrate that is basically tasteless and insoluble in cold water, alcohol,…
Q: Define the following terms briefly as they relate to the experiments Cite an example for each using…
A: Carbohydrates are large biomolecules composed of carbon, hydrogen and oxygen. Carbohydrates are…
Q: which describes the complex carbohydrate cellulose
A: Cellulose is a polysaccharide that incorporates a linear chain of numerous hundred to numerous…
Q: Ellman's reagent is used to quantify the (one word) content of proteins.
A: Proteins are large biological polymers (macromolecules) made up of monomer units of amino acid…
Q: DESCRIBE THE RESULTS OF THE REDUCTION TEST FOR SUGARS USING THE FOLLOWING TEST MOLISCH’S TEST SUGAR…
A: Carbohydrates or sugars are the most abundant biopolymers in living organisms. Carbohydrates are…
Q: Five carbohydrate samples were analyzed for their qualitative reactions and the results are…
A: Carbohydrates or carbs are macro molecules which are composed of carbon, hydrogen and oxygen. They…
Q: A dehydration reaction joins two glucose molecules toform maltose. The formula for glucose is…
A: Glucose is a simple sugar (monosaccharide). Its formula is C6H12O6. It is called hexose sugar as it…
Q: The question is : Examples of polysaccharides that have 1,6 bonding are ___ and ____
A: Polysaccharide as the name suggests that “poly” means many and “saccharides” mean carbohydrates or…
Q: What are the advantages and disadvantages of using the Lieberman-Burchard Test for Lipids
A: Lieberman-Burchard Test : this is the specific test used for detection of cholesterol in which…
Q: Numerous drugs are obtained from Glycosides. If you were to conduct a research using glycoside as…
A: Glycosides are described as chemicals in which one or more sugars are linked to non-sugar molecules…
Q: Describe the property of potassium iodide solution in its affects on the results of detection of…
A: Iodine test is used in detection of starch using iodine and potassium iodide solution.
Q: Given Ribose, Briefly explain its expected reaction (based on their structural formula) to the…
A: Ribose is a simple sugar and carbohydrate. Ribose, also called D-ribose is a five-carbon sugar found…
Q: Define the following terms: a. sugar b. glucose c. monosaccharide d. polysaccharide e. cellulose
A: Carbohydrates or sugar are the main biomolecules nutrient in food. Carbohydrates are divided into 5…
Q: In what way are sugar and salt similar
A: Salt is an ionically linked mixture of sodium and chloride. Sugar, on the other hand, is made up of…
Q: Hoe Cellulose acetate is made of ?
A: Cellulose is a complex polysaccharide that consists of 3000 units of glucose. The basic structural…
Q: Monosaccharides are:
A: Carbohydrates are the polyhydroxy of aldehyde or ketone bodies. The carbohydrates are of three types…
Q: What carbohydrate(s) would give the following test results? a. Produces 9 raddich
A: Answer 5- Benedict's Test- This test is done for reducing sugar. It is a semi-quantitative test.…
Q: Answer the following question about monosaccharide A. What two aldoses yield A in a Wohl…
A: Wohl's degradation is a carbohydrate shortening reaction. In this reaction, there is a cleavage…
Q: Is it somehow possible to generate carbohydrate from non-carbohydrate? If yes, how and if no, why?
A: Carbohydrates are organic molecules, which act as the primary source of energy. Glucose is the…
Q: In a laboratory experiment, a student was given an unknown compound. After performing the test for…
A: Carbohydrates are the polyhydroxy aldehyde or ketone. Aldehyde containing monosaccharides are called…
Q: glucose and galactose
A: Glucose: Glucose is a simple sugar composed of C, H, and O atoms and tastes sweet. Galactose:…
Q: Will all carbohydrates gives a positive result with Molisch test? Explain your answer:
A: Molisch test is named so after the name of its discoverer, Hans Molisch. It is a kind of chemical…
Q: - What inferences can be made from the Benedict's test of the hydrolysates of the following? Was the…
A: Benedict's test is a biochemical test used to detect the presence of reducing sugars in a given…
Q: A topping for ice cream contains fructose, hydrogenated soybean oil, salt, and cellulose. What types…
A: Atoms are small substances that together make molecules and compounds.
Q: of the following are monosaccharides, Exce a. Galactose b. Fructose C. Sucrose
A: Monosaccharides are the simplest type of carbohydrate. Monosaccharides can be joined together by…
Q: The hydrolysis of starch was stopped when the iodine test no longer gave a blue color. Does this…
A: Starch is a complex carbohydrate, which is made up of amylose and amylopectin subunits. Since…
Q: If you are performing the carbohydrate analysis with sugar maltose, what are all the tests that…
A: Maltose is a disaccharide shaped from two units of glucose that got together with an α bond. In the…
Q: Does a positive result will occur on a Molisch's Test when trying disaccharides or polysaccharides?
A: The Molisch test is a colorimetric method for determining whether or not an analyte contains…
Q: What is cellulose
A: Cellulose is a carbohydrate, It is a polysaccharide, which means it is made of a linear chain of…
Q: Although both sugar and salt are soluble in water, the way in which they dissolve is different.…
A: Salt is an ionic compound. Sugar is a covalent compound.
Q: Why does the number of chiral carbon atoms in glucose increase when it mutarotates? You may include…
A: Introduction: The term “mutarotation” refers to the observed change in the optical rotation of the…
Q: What are at least three things which can happen to carbohydrates after they get absorbed into the…
A: The metabolism of carbohydrates:It is the process of receiving the carbohydrates to provide energy…
Q: Which of the following is not a use for cellulose?(a) lumber for building(b) fodder for cattle(c)…
A: Cellulose is a polysaccharide of glucose that is present in the plant's cell wall and is…
What inferences can be made from the Benedict’s test of the hydrolysates of the following? Was the acid treatment able to hydrolyze the sugars?
a. sucrose
b. starch
c. cellulose
Trending now
This is a popular solution!
Step by step
Solved in 5 steps

- Which of the following statements best explains the positive result of the acid hydrolysate of sucrose for benedict's test? A. The acid hydrolysate of sucrose contains glucose only B. The acid hydrolysate of sucrose contains fructose only C. Both glucose and fructose are present in the acid hydrolysate of sucrose D. Neither glucose nor fructose is present in the acid hydrolysate of sucroseFill in the volumes required to make the standard glucose solutions of various conentrations provided. provide calculations as well.Explain the role of potassium thiocyanate, KSCN as a component of the Benedict’s reagent for the Estimation of Glucose
- dont mind the questions from a-e. please help me know the answers for f. How much is the molar mass of the fatty acid sample? g How many moles of ATP per gram of fatty acid is formed from the complete catabolism of the sample? basing from the questions above. thank you!In glucose estimation using Benedict's Reagent, what is/are the chemical reaction/s that resulted in the white precipitate observed.How do I calculate the final concentration? I just need one solution to walk through the steps. Prepare eight standards of glucose with your 1 mM glucose solution, each with a final volume of 10 mL. Complete the table on the next page, indicating what your final concentration of glucose will be for each standard, in mM. Volume of Potassium Ferrocyanide Solution Volume of Enzyme Solution Volume of 1 mM glucose solution Final volume of solution Final Concentration of glucose in final solution (mM) 2.50 mL 1.00 mL 0.00 mL 10.00 mL 2.50 mL 1.00 mL 0.50 mL 10.00 mL 2.50 mL 1.00 mL 1.00 mL 10.00 mL 2.50 mL 1.00 mL 2.00 mL 10.00 mL 2.50 mL 1.00 mL 3.00 mL 10.00 mL 2.50 mL 1.00 mL 4.00 mL 10.00 mL 2.50 mL 1.00 mL 5.00 mL 10.00 mL 2.50 mL 1.00 mL 6.00 mL 10.00 mL
- In healthy adults, the concentration of glucose in blood is approximately80 to 110 milligrams per deciliter (mg/dl). After a carbohydrate-richmeal, however, the concentration may spike to 140 mg/dl. Describe thehormonal action that returns blood glucose to normal.An individual goes in to get their blood work results for their enzyme deficiency in glycolysis. Based on the results, provide an explanation of which enzymes are deficient in the individual? Blood Glucose levels are normal *Glucose-6 phosphate, *fructose-6 phosphate, *fructose-1,6 biphosphate, *glyceraldehyde-3 phosphate, *1,3-biphosphoglycerate, and *phosphoenolpyruvate levels are ALL increased Pyruvate, ATP, and RBC concentration are all at a decreased levelAs soon as lysis occurs, proteolysis, dephosphorylation and denaturation begin. These events can be slowed down considerably if samples are kept on ice or at 4°C at all times and appropriate inhibitors (protease and phosphatase) are added fresh to the lysis buffer. -Why inhibitors are necessary in this stage?-

