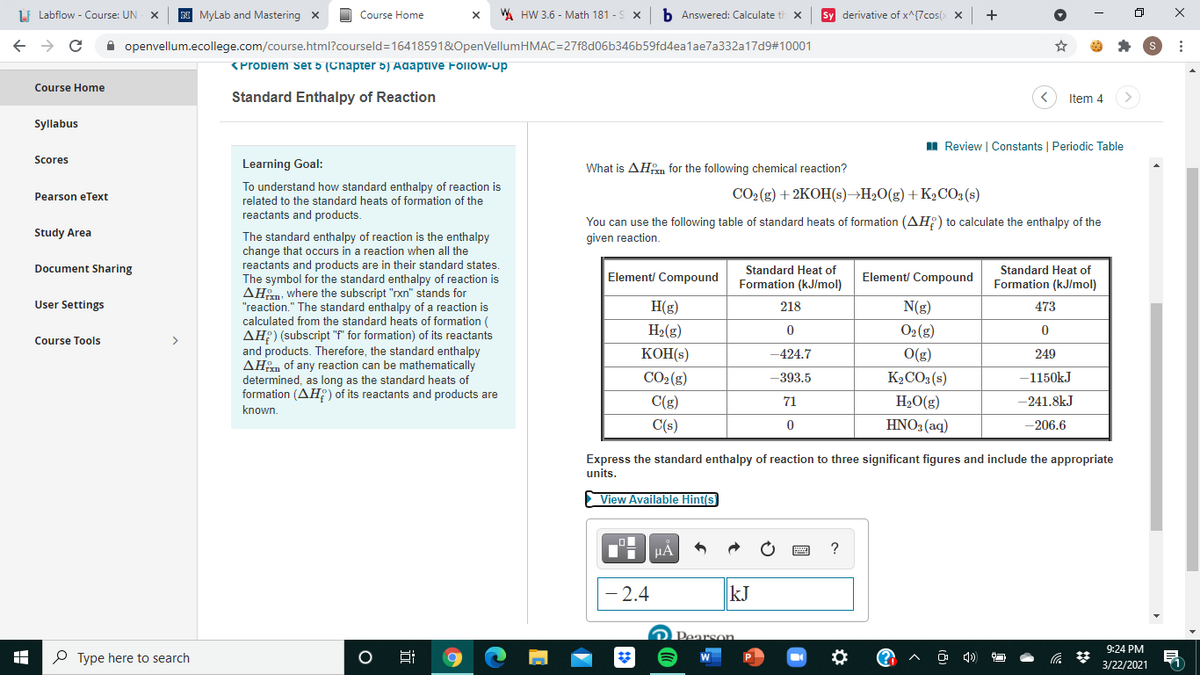
What is AHfm for the following chemical reaction? CO2(g) + 2KOH(s)¬H20(g) + K½CO3(s) You can use the following table of standard heats of formation (AH; ) to calculate the enthalpy of the given reaction. Standard Heat of Formation (kJ/mol) Standard Heat of Element/ Compound Element/ Compound Formation (kJ/mol) H(g) 218 N(g) 473 H2(g) O2(g) KOH(s) -424.7 O(g) 249 CO2(g) -393.5 K½CO3(s) -1150kJ C(g) 71 H2O(g) -241.8kJ C(s) HNO3 (aq) -206.6 Express the standard enthalpy of reaction to three significant figures and include the appropriate units.
What is AHfm for the following chemical reaction? CO2(g) + 2KOH(s)¬H20(g) + K½CO3(s) You can use the following table of standard heats of formation (AH; ) to calculate the enthalpy of the given reaction. Standard Heat of Formation (kJ/mol) Standard Heat of Element/ Compound Element/ Compound Formation (kJ/mol) H(g) 218 N(g) 473 H2(g) O2(g) KOH(s) -424.7 O(g) 249 CO2(g) -393.5 K½CO3(s) -1150kJ C(g) 71 H2O(g) -241.8kJ C(s) HNO3 (aq) -206.6 Express the standard enthalpy of reaction to three significant figures and include the appropriate units.
Chapter7: Statistical Data Treatment And Evaluation
Section: Chapter Questions
Problem 7.16QAP
Related questions
Question
Transcribed Image Text:LJ Labflow - Course: UN
N MyLab and Mastering x
Course Home
WA HW 3.6 - Math 181 - S X
b Answered: Calculate th x
Sy derivative of x^{7cos( X
A openvellum.ecollege.com/course.html?courseld=16418591&OpenVellumHMAC=27f8d06b346b59fd4ealae7a332a17d9#10001
KPropiem set 5 (Chapter 5) Adaptive Foilow-up
Course Home
Standard Enthalpy of Reaction
Item 4
Syllabus
II Review | Constants | Periodic Table
Scores
Learning Goal:
What is AHm for the following chemical reaction?
To understand how standard enthalpy of reaction is
related to the standard heats of formation of the
CO2 (g) + 2KOH(s)→H2O(g) +K2CO3 (s)
Pearson eText
reactants and products.
You can use the following table of standard heats of formation (AH:) to calculate the enthalpy of the
Study Area
The standard enthalpy of reaction is the enthalpy
change that occurs in a reaction when all the
reactants and products are in their standard states.
The symbol for the standard enthalpy of reaction is
AHn. where the subscript "rxn" stands for
"reaction." The standard enthalpy
calculated from the standard heats of formation (
AH:) (subscript "f" for formation) of its reactants
and products. Therefore, the standard enthalpy
AHm of any reaction can be mathematically
determined, as long as the standard heats of
formation (AH;) of its reactants and products are
given reaction.
Document Sharing
Standard Heat of
Standard Heat of
Element/ Compound
Element/ Compound
Formation (kJ/mol)
Formation (kJ/mol)
User Settings
of a reaction is
H(g)
218
N(g)
473
H2(g)
O2 (g)
Course Tools
>
KOH(s)
-424.7
O(g)
249
CO2 (g)
-393.5
K2CO3 (s)
-1150kJ
C(g)
71
H2O(g)
-241.8kJ
known
C(s)
HNO3 (aq)
-206.6
Express the standard enthalpy of reaction to three significant figures and include the appropriate
units.
View Available Hint(s)
?
2.4
kJ
D Pearson
9:24 PM
P Type here to search
3/22/2021
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you



Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning



Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning