Q: Write a reaction scheme and draw the reaction mechanism for the williamson ether synthesis.
A: The Williamson ether synthesis is an organic reaction in which the forming an ether from an…
Q: What is the major organic product obtained from the following reaction? A, B, C, or D?
A: Given that : We have to predict the major product of the following reaction :
Q: The triphenylmethyl cation is so stable that a salt such as triphenylmethyl chloride can be isolated…
A: The species in which carbon carries a positive charge is called as carbocation. Carbocations are…
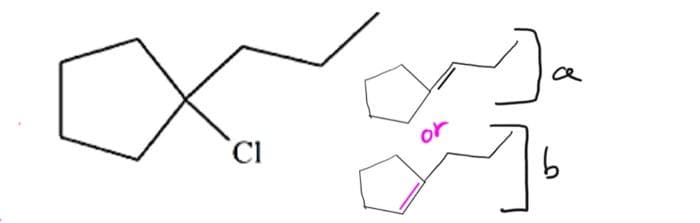
Q: Draw the structure (including stereochemistry) of an alkyl chloride thatforms attached alkene as the…
A: The alkyl chloride should be such chosen that it forms the product as the one and only major…
Q: Select the compound(s) that will give only a single monochlorination product in its free radical…
A:
Q: Select the member of each pair that undergoes SN1 solvolysis in aqueous ethanol more rapidly
A: Given pair is,
Q: Why is there a difference in racemization for the two reactions even though they both include the…
A: Bromine is an excellent leaving group compared to hydroxyl group. Hence, in case of 2-bromobutane,…
Q: Where will a nucleophile initially bond to the structure of carvone, provide a reason
A:
Q: Give the product and the mechenism of the following reaction
A: The reaction given is,
Q: Draw the structure (including stereochemistry) of an alkyl chloride that forms each alkene as the…
A: Both ‘H’ and ‘Br’ group should be trans to each other. Thus the alkyl halides needed for the…
Q: Select the member of each pair that shows the greater rate of SN2 reaction with KI in acetone.
A:
Q: When styrene is protonated as shown, on which carbon will the carbocation initially form?
A: Protonation occurs in such a way that a stable carbocation intermediate is formed. The stability of…
Q: Rank the following in terms of reactivity towards a nucleophile (1 = least reactive and 4 = most…
A: Electrophiles are reagents that are electron deficient. These attack reagents that are rich in…
Q: A Moving to another question will save this response. Question 5 Which one of the following…
A: In the given compounds toluene,benzene, chlorobenzene,t-butyl benzene undergo a Friedel-crafts…
Q: Show, in increasing order, the relative stability of the following carbocations
A: Stability of carbocation intermediate can be explained in terms of number of hyperconjugative…
Q: Which compounds are more nucleophilic, ethers or sulfides? Explain your answer and provide an…
A: The special group in an organic compound which decides the chemical properties is known as the…
Q: Provide the appropriate reagent(s) for the following formation
A:
Q: What is the appropriate electrophile nucleophile and solvent to achieve this final product
A:
Q: Which carbon will most likely undergo radical halogenation when reacted with Br, in the presence of…
A: Note: As per our guidelines, we are supposed to answer only one question. Please repost the other…
Q: Explain why a nucleophilic reagent such as ethoxide adds to an alkyne more easily than it adds to an…
A: The unsaturated hydrocarbons are of two types namely alkenes and alkynes. The alkenes consist of at…
Q: a. What alkene would give only a ketone with three carbons as a product of oxidative cleavage? b.…
A: b.
Q: What reaction sequence would lead to the following transformation? ?
A: here we are required to predict the reagent needed to carry out the following conversion.
Q: Draw the alkyl iodide starting material that would give the following alkene as the major product of…
A:
Q: Q) Write the structure of the Dieckmann cyclization product formed on treatment of each of the…
A:
Q: 4) What happens to the stereochemistry during and SN2 reaction? Why? Provide a reaction to…
A: SN2 reaction is bimolecular nucleophilic substitution reaction. This is single step reaction that…
Q: Why does bromination give exclusively anti products while chlorination gives a mixture of syn and…
A: Halogenation is the addition of halogens to alkenes. Halogenation usually gives anti products. If…
Q: 6 Draw the structure (including stereochemistry) of an alkyl chloride that forms each alkene as the…
A: In presence of strong base conditions alkylhalides undergo elimination reactions by eliminating…
Q: 3. Rank the following in order by the rate in which they could participate in an SN1 reaction (“1"…
A:
Q: For the following pair of carbocations (A and B), select the one that is more stable and explain…
A:
Q: b. A third alcohol product is formed under these conditions that is not one of the two possible…
A: Given is hydration reaction.
Q: Explain why alkylation of an α-carbon works best if the alkyl halide used in the reaction is a…
A: The alkylation reaction of an α-carbon depends on the R-X (alkyl halide) used in the reaction. The…
Q: From the Hammet equation data, it was observed that the nucleophilic substitution reaction of benzyl…
A:
Q: a) What is (are) the major product(s) of the nitration reaction. Explain this selectivity, and…
A:
Q: Select the member of each pair that shows the greater rate of SN2 reaction with KN3 in acetone.
A: SN2 reaction rate depends upon the steric effect. Higher the steric hindrance, lower would be the…
Q: Explain why compound A will not undergo a ring-opening reaction under thermal conditions, but…
A: Ring opening reactions for A and B are given below :
Q: (a) Why cyclohexene and cyclohexa-1,3-diene are expected to undergo addition reaction but benzene…
A: Organic compound undergoes electrophilic substitution reaction in presence of electrophile. Electron…
Q: Consider the four ethers below. Three of them can be made using one (or more) of the methods we…
A:
Q: Determine the product of the reaction
A: In the hydro-chlorination reaction, first proton is added in the least substituted carbon then the…
Q: Which mononitro product is the result of the nitration reaction of the compound whose open structure…
A: In amide group, -CO-NH- group lone pair of Nitrogen atom are in resonance with Carbonyl group. The…
Q: Give the major organic product for each step of the following reaction
A:
Q: Part A: Describe the mechanism for dihydroxylation of an alkene through the use of a peroxide…
A:
Q: B D Write the letters that correspond to the starting material and the product for the mechanistic…
A: In this question, you give the letters for starting material and Product ( on the right side energy…
Q: Please help. What other combination of ylide and aldehyde or ketone will give methylenecyclohexane…
A:
Q: In the most efficient Wittig synthesis, the halide. Which of these compounds would the alkene in the…
A: Wittig reaction involves the formation of alkenes from aldehydes or ketones using…
Q: b) In a polar solvent such as water, the SN1 and E1 reactions of a tertiary alkyl halide have the…
A: SN1 is a unimolecular nucleophilic substitution reaction. It is a reaction between an alkyl halide…
Q: a) place asterisks(*) at the allylic position of compound A. b) Draw the resonance forms of the…
A:
Q: What happens to the stereochemistry during and SN2 reaction? Why? Provide a reaction to illustrate…
A:
what is the
Step by step
Solved in 2 steps with 2 images


