Q: Aluminum will react with bromine to form aluminum bromide (used as an acid catalyst in organic…
A:
Q: Calculate the percent composition of H in NH4NO3, a compound used as a fertilizer. A. 27.8 B. 8.07…
A: Recall, Compound= NH4NO3 %H = ?
Q: Determine the concentration of all solute species present in a 0.50 solution of nitrous acid (HNO2)…
A: Given : Concentration of HNO2 = 0.50 mol/L Ka of acid = 7.1 x 10-4
Q: (6) After three half-lives, 100% (A) (B) 50% (C) 25% (D) 12.5% % of the initial isotope of a…
A:
Q: A 29mL sample of chromium-51 contains 1.06 mCimCi. If a patient is to receive a 50 μCiμCi dose to…
A:
Q: (1.8)The area of a paper is 758 cm². What is the area of the paper in m²? O 7.58 x 104 m² O 7.58 x…
A:
Q: 2 → HOCI Activity 2: Complete the reaction by drawing or writing the correct structure to match the…
A:
Q: you add all the oxidation numbers in a polyatomic ion, what should the sum be? A The sum should…
A: lets solve this problem by taking an example of SO42-
Q: 5. Given the following mechanism: EIOH Br . What is the type of this reaction mechanism (E1, E2, Sul…
A: According to the company guidelines, we have to answer only the first question. So, please kindly…
Q: Dilution: 1.How many mL of 6 M solution should be diluted to 200 mL to make a 1.5 M soln? 2. To…
A:
Q: REDUCED 3 Hg2+ + 2 Fe (s) → 3 Hg2 + 2 Fe3+
A: reduction ; 3 Hg2+ + 6e- ---> 3 Hg2 oxidation ; 2Fe ---> 2 Fe3+ + 6e-
Q: (3.10: Similar to Example 3.17 & 18)Find the empirical formula of a compound containing 36.86%…
A: Given, The mass percent of Nitrogen = 36.86% mass percent of Oxygen = 63.14 % The…
Q: A food contains 131 kcals, 24 grams carbohydrates. What % of kcals are from carbohydrates?
A: Given, Total energy in a food = 131 kcals mass of carbohydrate = 24 g % of kcals are from…
Q: Propose a chemical structure for the name below. Make sure to clearly define the stereochemistry…
A: 1) The chemical structure of (R)-4-methyl-4-phenylcyclohex-2-en-1-one is
Q: At a temperature of 525 K the Keq = 0.041 for the following reaction: PCl5(g) ⇄ PCl3(g) + Cl2(g) At…
A:
Q: Consider the reaction: AB(g) → B(g) +A(g) with the rate law Rate = K[AB]2 and the rate constant k =…
A: given, the rate law equation, Rate = k[AB]2 rate constant, k = 5.0×10-2 M-1.s-1 [AB] = 2.0 M
Q: The rate of attack on [Co(H 2 O) 6 ]^ 3+ by an entering group Y is nearly independent of Y, with the…
A:
Q: Name the compound (IUPAC NAME)
A:
Q: (1.3)(Question 42 on page 40) Which of the following represent(s)' homogeneous mixture'? (b) (c) O…
A: In a mixture in where the components of that particular mixture are uniformly distributed throughout…
Q: Calculate the freezing point (to the nearest 0.1° C) for a solution containing 17.3 g of SrBr and…
A: Given data : Mass of SrBr = 17.3 g Mass of lactose = 30.8 g Mass of water = 200 g = 0.200 kg Kf =…
Q: A chemist adds 35.0 mL of a 7.43 x 10μM zinc oxalate (ZnC₂04) solution to a reaction flask.…
A:
Q: Compounds containing these ions are Soluble NO₂™ Na, K, NH₂ CF, Br, I SO4² Compounds containing…
A: Given reaction: CaI2 (aq) + Na3PO4 (aq) → We have to write the balanced molecular, complete ionic,…
Q: 9. The water-soluble vitamins B₁ (thiamine hydrochloride), B₂ (riboflavin), B3 (niacinamide), and B…
A: According to the given pKa value and the following structure, the order of elution for the four B…
Q: Indicate which substance in the given pairs has the higher entropy value. Explain your choices. a.…
A:
Q: How many mL of a 15% (w/v) solution must be administered to deliver 8.0 g of solute?
A: 15% (w/v) solution = mass of solute in gvolume of solution in mL×100
Q: Griven 2 half cells Reduction half Reaction | Reducitur potentialu) Cu2+ caa) +26 C4 (5) Ni (aq) +…
A:
Q: A certain reaction has ∆?°= -19.5KJ and ∆S°= 42.7 J/K, does the reaction lead to an increase or…
A: Entropy is measure of randomness or disorderness in the system. More the entropy, more is…
Q: Which of the following would not affect the position of equilibrium of the chemical reaction system…
A:
Q: (3.8: Similar to For More Practice 3.13) Determine the mass of a sample of carbon monoxide…
A:
Q: Continue the synthesis by selecting the reagents necessary for the next transformation shown. ? Br…
A: The correct order of reagents used for this transformation is given in second step.
Q: Research and explain the equilibrium relationship the following system: Human blood A…
A: Since you have posted a question with multiple sub-parts, we will solve first three sub-parts for…
Q: (19) Consider the following redox reaction that occurs in a voltaic cell: Cr2+ (aq) + Cu²+ (aq) →…
A:
Q: 2.The initial rate of the reaction: BrO3(aq) + 5 Br(aq) + 8 H*(aq) → 3 Brz(1) + H₂O(1) Has been…
A: Here we are required to find the initial rate of reaction
Q: The molecules butane and 2-methylpropane both have the formula C4H10. Their space-filling models are…
A:
Q: 16. The decomposition of hydrogen bromide is 2 HBr(g) → H2(g) + Br2(g) The reaction has…
A:
Q: Determine the concentration of all species and the pH of the following solutions: H2CO3 0.10 mol/L…
A: Note - Dear student, since the given question is a multiple question, hence I solved first one…
Q: 8. The diagram below illustrates an osmotic pressure experiment involving two solutions, 0.10 M…
A:
Q: 4. a. What is the pH, and the equilibrium concentrations for [H*], [CH³COO-], [CH3COOH] and [OH-] in…
A:
Q: CH3 2 3 CH3 CH3
A: Chiral carbon is that carbon which posses four different groups attached to it.
Q: (7) A . B C D What is the major product of the following reaction? Br CH3O+Na CH3OH J (A) OCH 31 (B)…
A:
Q: Assign E or Z configuration (a) H3C CH₂OH C=C CH3CH₂ Cl (c) CH3 C=C CO₂H CH₂OH to the following…
A: Rules for assigning E or Z configuration
Q: For each problem, you must: 1) Calculate the degree of unsaturation. 2) Assign the principal IR…
A: The correct answer is given below
Q: 8. Which of the following is NOT produced during the electrolysis of water? Electricity a) b) Acid…
A: we have to select the option which is not produced in electrolysis of water
Q: Which of the following minerals can be leached using NaOH? a.Oxides b.Bauxites c.Sulfides d.Sulfates…
A: Bauxites Oxygen Physical leaching process Elemental metal is routinely converted to metal cations at…
Q: A particular beer is 6.50 % ethanol by volume (C₂H₆O). If a single bottle of beer contain 750.0 mL…
A:
Q: Consider the reaction shown below at equilibrium with solid BaSO4 present. BaSO4(s) Ba2+, + SO4²…
A:
Q: At equilibrium, the rate of the forward reaction is use pic a 0.00 mol/min b 0.25mol/min c 1.0…
A:
Q: 19. Calculate the number of moles oh hydrogen atoms in 17.4 g of (NH4)2SO4. A. 2.13 mol H atoms B.…
A:
Q: (13) A B C OD What is not an elementary step in the mechanism for the following reaction? H₂O НО. X…
A:
Q: CI The common name for metabolite 3 Metabolite 3 -OH
A: Metabolites: They are the intermediate merchandise produced all through metabolism, catalyzed with…
Trending now
This is a popular solution!
Step by step
Solved in 5 steps with 4 images

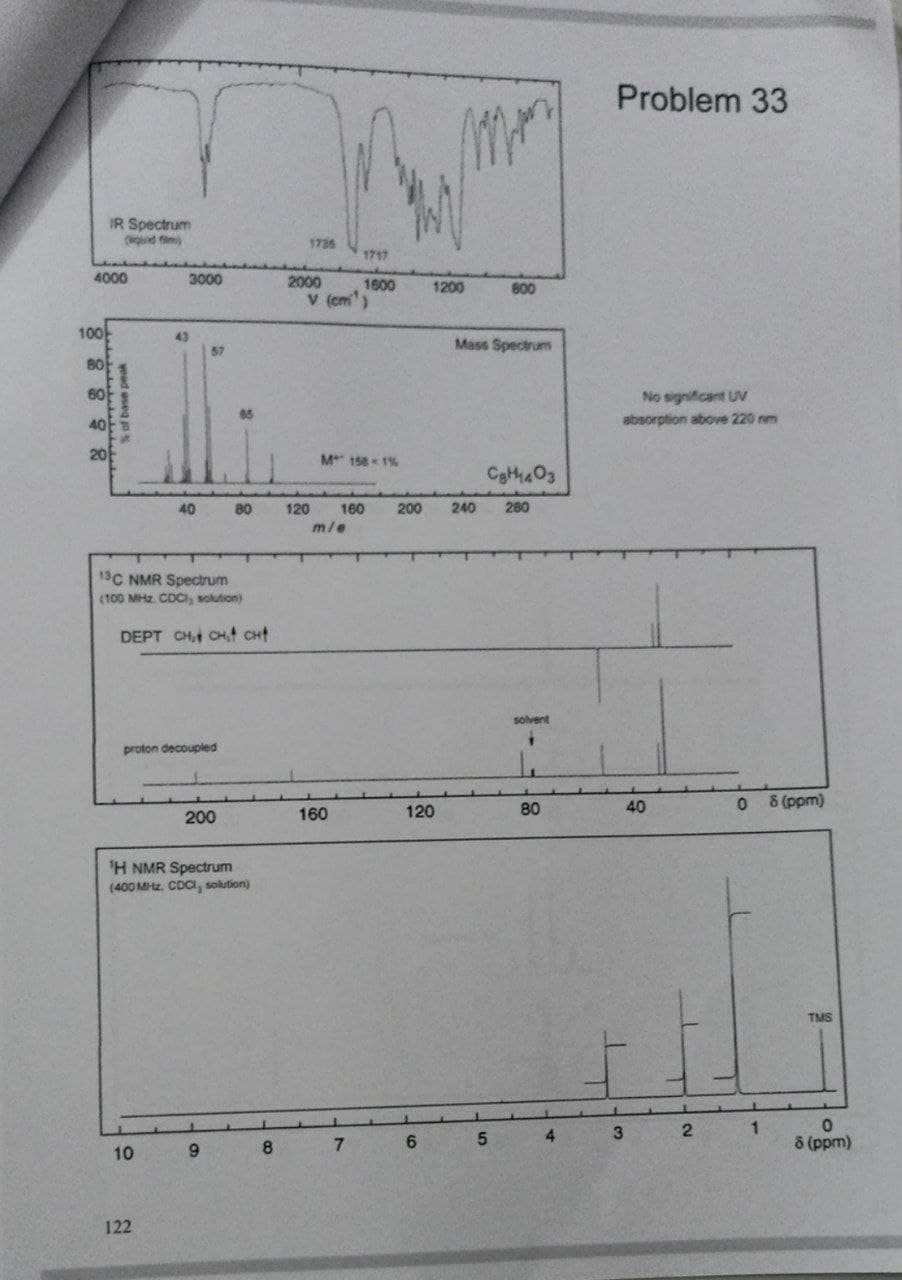
- plese don't handwritten solution... What MS resolving power is needed to fully resolve the C8H8NO2 and C7H6N2O2 ions (at the base peaks)?A H1 NMR signal is observed at 117.0 Hz (downfield of TMS) on a 1.20×102‑MHz instrument. a) What is the observed chemical shift in parts per million (ppm)? Chemical shift is ppm b) What is the chemical shift, in parts per million (ppm), if the sample is analyzed with a 60.0‑MHz instrument? Chemical shift is ppm c) Where will the proton signal appear, in hertz, if the sample is analyzed with a 60.0‑MHz instrument?1. For each set of spectral data, propose an acceptable structure that is consistent with the data. a. MS: M+ m/z=71, IR: 3340, 2960, 2870 cm-1 , 13C NMR: 47.20 (CH2), 25.64 (CH2) ppm b. MS: M+ m/z= 102, IR: 2960, 2870 cm-1, 13C NMR: 72.73 (t), 23.20 (t), 10.80 (q) ppm c. MS: M+ m/z=74, IR: 3310, 2960, 2870 cm^-1, 13C NMR: 62.44 (down), 34.86 (down), 18.98 (down), 13.88 (up) ppm (DEPT-direction) d. MS: M+ m/z=92 (M:M+2=3:1), IR: 2960, 2870 cm^-1, 13C NMR: 67.29 (C), 34.46 (CH3) ppm e. MS:M+ m/z=70, IR: 3070, 2960, 2870, 1666 cm^-1, 13C NMR: 138.92 (t), 114.26 (d), 35.96 (t), 22.17 (t), 13.59 (q) ppm f. MS: M+ m/z= 72, IR: 2960, 2870 cm^-1, 13C NMR: 31.68 (t), 29.80 (d), 22.20 (q), 11.69 (q) ppm g. MS: M+ m/z= 84, IR: 2960, 2870, 1734 cm^-1, 13C NMR: 220.16 (C), 38.30 (CH2), 23.24 (CH2) ppm h. MS:M+ m/z= 138, (M:M+2=1:1), IR: ~3010(broad), 1715 cm^-1, 13C NMR: 173.61(s), 25.21(t) ppm i. MS:M+ m/z=88, IR: 2960, 2870, 1736cm^-1, 13C NMR: 170.95 (C), 60.34 (CH2), 20.98 (CH3), 14.23 (CH3)…
- For each set of spectral data, propose an acceptable structure that is consistent with the data 1.. MS: M+ m/z=92 (M:M+2=3:1), IR: 2960, 2870 cm^-1, 13C NMR: 67.29 (C), 34.46 (CH3) ppm 2.. MS:M+ m/z=70, IR: 3070, 2960, 2870, 1666 cm^-1, 13C NMR: 138.92 (t), 114.26 (d), 35.96 (t), 22.17 (t), 13.59 (q) ppm 3.. MS: M+ m/z= 72, IR: 2960, 2870 cm^-1, 13C NMR: 31.68 (t), 29.80 (d), 22.20 (q), 11.69 (q) ppmThe NMR spectra for compound 1 were acquired in a 7.5 mg / 0.6 mL solution ofCDCl3 and are found in the accompanying file. The 1H are also listedbelow. Identify the name of compound 1 and Provide a full analysis of the NMR spectra for compound 1 with table showing peak name and assignment 1H NMR (400 MHz, CDCl3) δ 7.73 (d, J = 9.5 Hz, 1H), 7.56 (ddd, J = 8.5, 7.5, 1.6 Hz, 1H),7.51 (dd, J = 7.5, 1.6 Hz, 1H), 7.36 (d, J = 8.5 Hz, 1H), 7.30 (dd, J = 8.5, 7.5 Hz, 1H), 6.45(d, J = 9.5 Hz, 1H).Using a 300 MHz NMR instrument: a.How many Hz downfield from TMS is a signal at 2.5 ppm? b. If a signal comes at 1200 Hz downfield from TMS, at what ppm does it occur? c.If two signals are separated by 2 ppm, how many Hz does this correspond to?
- For each set of spectral data, propose an acceptable structure that is consistent with that data 1.. MS: M+ m/z= 84, IR: 2960, 2870, 1734 cm^-1, 13C NMR: 220.16 (C), 38.30 (CH2), 23.24 (CH2) ppm 2. MS:M+ m/z= 138, (M:M+2=1:1), IR: ~3010(broad), 1715 cm^-1, 13C NMR: 173.61(s), 25.21(t) ppm 3. MS:M+ m/z=88, IR: 2960, 2870, 1736cm^-1, 13C NMR: 170.95 (C), 60.34 (CH2), 20.98 (CH3), 14.23 (CH3) ppm 4. MS: M+ m/z=73, IR: 3410, 2960, 2870, 1668 cm^-1, 13C NMR: 171.27 (C), 26.26 (CH3), 22.87 (CH3) ppmUsing a 300 MHz NMR instrument:a. How many Hz downfield from TMS is a signal at 2.5 ppm?b. If a signal comes at 1200 Hz downeld from TMS, at what ppm does it occur?c. If two signals are separated by 2 ppm, how many Hz does this correspond to?Two isomers have NMR chemical shifts of 5.5 and 7.0. What would be the coalescence time in a 500 MHz NMR?
- 1. Using NMR integral ratios of peaks from sesamin and episesamin in the 1H NMR spectrum of the epimerized sample attached, calculate the actual ratio of sesamin to episesamin in the epimerized sample a. state the ppm values of the two NMR peaks you compared -sesamin 1H NMR peak __________ppm -episesamin 1H NMR peak _________ppm b. please show the calculation for the actual ratio of sesamin to episesaminCalculate the IHD and identify the important peaks in the following MS spectral data and draw the structure of the important peaks in the following MS spectral data.use software to make 1H-NMR and 13C-NMR from the data in the paper and make a table with the following data 1H: chemical shift , multiplet structure, integration, assignment 13C: chemical shift

