Q: Which equation is used to determine the amount of time required for the initial concentration rate…
A:
Q: When solid ammonium chloride is added to water, all of the solid dissolves and the solution becomes…
A: According to the answering guidelines, I'm posting the solution for the first question. Kindly post…
Q: 10. то ОН HCI
A: Introduction Tertiary alcohols are organic compounds in which a hydroxy group is attached to a…
Q: (c) Explain in chemical terms why treatment of ethanol with H₂SO4 and heat is not the best way to…
A: The Williamson ether synthesis is a chemical reaction used to synthesize ethers from alcohols and…
Q: What is the energy change when the temperature of 11.2 grams of solid diamond is decreased from 39.3…
A: Amount of heat energy ,Q required to change the temperature of a substance of mass ,m and specific…
Q: See Figure 3-1. Match the letter to the correct functional group. A B D ✓ [Choose ] alkene aromatic…
A: When compound contains C=C bond then functional group is alkene. When compound contains benzene ring…
Q: AlP Express answer as an ion.
A: Since, Ionic compound are those compound which on dissociation produced cation and anion. Thus,
Q: Consider solutions of Cr(NO3)3-9H₂O in water. The solubility of Cr(NO3)3-9H2O in water at 15 °C is…
A: Introduction Solubility is an important physical property of matter that is related to a variety of…
Q: molar mass of solute B is 101.103 g mol−1 A 100.5 g sample of solute B dissolved in 100 g of…
A: Given that, A 100.5 g sample of solute B dissolved in 100 g of water is decreased from 85 °C to 35…
Q: The following Lewis diagram represents the valence electron configuration of a main-group element.…
A: Given : main group element which has 1 valence electron.
Q: Which of the following molecules exhibit hydrogen bonding? Write "yes" if it does or "no" otherwise.…
A:
Q: 11. H Br CI-
A: An organic reaction in which an electron-rich species attacks the electron-deficient carbon and…
Q: Question 1 The reaction below is third order in A and second order in B, what is the rate law? The…
A: Given: Third order in A Second order in B To determine: The rate law The units of rate constant must…
Q: + d. What is the rate of this reaction between 20.0 and 50.0 seconds? Use the balanced equation…
A: The rate of a chemical reaction is the change in concentration of a reactant or product over time.…
Q: What is the average kinetic energy (in kJ mol-¹) of a hypothetical gas with molecular weight 52.6 g…
A: Please find your solution below : The average kinetic energy of a gas is directly proportional to…
Q: The mass of the average boysenberry is 0.28 ounces. The average Tesla weighs 4900 lbs. If one were…
A: Given: mass of boysenberry = 0.28 ounces = 0.28 x 28.3495 g = 7.938 g mass of Tesla = 4900 lbs =…
Q: Which compound would have the largest wavelength for its Xmax? A B
A: Homo-annular dienes have a λmax value of 253 nm. On addition of a conjugated double bond to this…
Q: Using the information in the table, the value of the rate constant for the reaction A(g) + 3 B(g) →…
A:
Q: Suppose you performed a similar experiment using cyclohexane as your solvent. Given the data in the…
A: Answer: When a non-volatile solute is added in a solvent, it causes a depression in its freezing…
Q: Consider the reaction: 2 C₂H₂(g) +5 O₂(g) →4 CO₂(g)+2 H₂O(1) Hrxn=-2600. kJ/mol pos90 31000d food 0…
A: Given : reaction and value of ∆Hrxn Tip : energy is given for 1 mole
Q: Indicate wether each of the following samples of matter is a heterogenous mixture, homogenous…
A:
Q: Vapor pressure (mm Hg) 900 800 700 600 500 400 300 200 100 0. Carbon disulfide 10 20 30 °C Methano…
A: Answer: variation of vapor pressure with temperature is given in the mentioned diagrams and we have…
Q: Provide a name or a Structure
A: Given: name and structure of molecule Tip : identify parent group
Q: 49. Which one of the following is NOT a Maxwell thermodynamic relation? (a) (), = (ar), ƏV (b) (c)…
A: Maxwell relations are thermodynamic relationships between the partial derivatives of thermodynamic…
Q: A solution is made up by dissolving 15.0 g MgSO4.7H₂O in 60.0 g of water. What is the molality of…
A: The given data is as follows: The mass of MgSO4.7H2O = 15.0 g The mass of water = 60.0 g = 0.060…
Q: could I have help wit this 3-part question about partial gasses of two substances, how would i go…
A: Given that - Volume of helium = 260 mL = 0.260 L Pressure of helium = 759 torr = 0.998 atm Volume…
Q: 7) from 2.50 s¹ at a temperature of 22.0°C to 12.5 s¹ at a temperature of 35.0°C. Calculate the…
A: Since you have asked multiple question, we will solve the first question for you. If you want any…
Q: Determine which of the following alkyl halides will react faster in an E2 reaction with NaiPr and…
A: The E2 reaction is the elimination reaction and it involves the conversion of an alkyl halide into…
Q: Consider two statements and state whether they are true or false: (1) If the energetics of…
A: The enthalpy of solution, also known as the heat of solution, is the amount of heat absorbed or…
Q: Curved arrows are used to illustrate the flow of electrons. Follow the arrows to predict the…
A:
Q: OH CH₂OH H OH Н OH OH In the box below, draw the open-chain structure (as a Fischer projection)…
A: We have to convert this compound from Haworth to fisher projection.
Q: Complete the following cooling and heating curves based on the information provided: Kryptonite A…
A: Answer: Two diagrams for phase transition when heat is added or removed are given for kryptonite and…
Q: Bromine has two stable isotopes: 79Br and 81Br with masses of 78.9183371 amu and 80.9162906 amu. If…
A: In this question we have to tell the the Mass of 79Br in 10 gram of Br which has 79.904 amu mass.
Q: Ⓒ Diaw structural formulas for the major organic products for XOH SOCI₂ CH b) 2) HO Hcl -OH HBr…
A:
Q: The arrangement of electrons around the nucleus of an atom is known as the aufbau principle the…
A: Answer:- The arrangement of electrons around the nucleus of an atom is known as electronic…
Q: If a solute is polar, then the solvent that theoretically should work for recrystallization should…
A: In the recrystallization experiment, the impure compound is purified by dissolving it in a minimum…
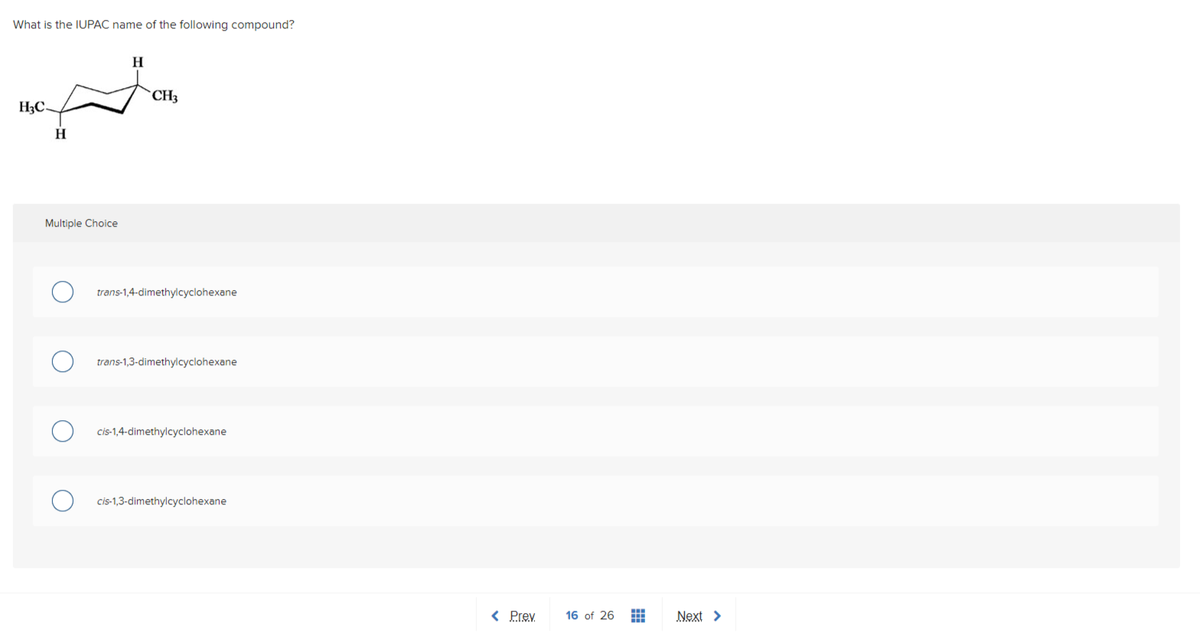
Q: Of the five names listed below, four contain deliberate errors. Which is the only name that is…
A: Answer:- This question is answered by using the simple concept of naming the organic compounds in…
Q: < A(g) + 3 B(g) → C(g) + 2 D(g) is [A]. (M) 1.25 1.25 3.75 [B]。 (M) 0.100 0.300 0.200 Tap here or…
A: Given : reaction and data
Q: Current Attempt in Progress A certain liquid has a vapor pressure of 6.91 mmHg at 0.0 °C. If this…
A: According to the question, The vapor pressure of the liquid is given by = P1 = 6.91mm Hg P2 = 1 atm…
Q: 3. Mechanisms and Rate Laws: What is the rate law for the overall reaction based on the proposed…
A: Answer: In the chemical reaction that takes place in more than one step, slowest step of the…
Q: A reaction mechanism has the following proposed elementary steps: Step 1: A ⇌ B + C Step 2: A + B →…
A: The given steps of the reaction are: Step 1: A⇌B+CStep 2: A+B→DStep 3: 2A+D→C+E In chemical…
Q: a. The density of sorbitol 70% w/w solution is 1.285g/ml. What is the concentration of sorbitol in…
A: 70 %w/w solution means that 70 g of sorbitol is present in 100 g of solution.
Q: Nitrogen monoxide, NO, is formed in automobile exhaust by the reaction of N₂ and O2 (from air).…
A: Given that, a equilibrium reaction is N2g + O2g ⇌ 2NOg. The equilibrium constant (Kc) is 0.0025 at…
Q: In the table, T₁ = 47.7 °C, E₁ = 153 J, E₂ = 836 J, E3 = 1410 J, and E4 = 2850 J. Other experiments…
A: Answer: When a solid substance is heated its temperature rises up to its fusion temperature and it…
Q: 5. Helium on the Moon was found to be 0.520% 2He, 8.25% ³He, and 91.23% 4He. What is the average…
A: average atomic mass = ∑(mass of its isotope× natural abundance )
Q: What is the overall reaction order for the following rate law: Rate = K[A] [B][C]²
A:
Q: what is the solution?
A: Introduction A solution can be anything that can be made by mixing two or more substances. Soda…
Q: What is the molar mass (in g mol-1) of a gas which has a density of 0.88 g/L at a pressure of 0.84…
A: Given P = 0.84 bar T = 22.6°C Density = 0.88 g/L
Q: [Pt¹V (NCS)6]
A: The given complex should not have zero charge but its have -2 charge.
Q: What is the molar mass (in g mol-1) of a gas which has a density of 0.48 g/L at a pressure of 0.98…
A:
Step by step
Solved in 2 steps with 1 images

- Show how to convert cyclopentene into cis-1,2-CyclopentanediolcompoundSolution:- 3. Determine the amount, in of a (2.20 M s olution of dichloromethane needed to completely react with 15.72g cyclohexene to give 1,2-dibromocuclohexanw. Assume 12% excess is needed in order to react completely. a . How much 1,2 -dibromocyclohexane would theoretically be produced ? c. How many ML of the 2.20 M Br2 solution are required?A chemist allows some pure (2S,3R)-3-bromo-2,3-diphenylpentane to react with a solution of sodium ethoxide(NaOCH2CH3) in ethanol. The products are two alkenes: A (cis-trans mixture) and B, a single pure isomer. Under thesame conditions, the reaction of (2S,3S)-3-bromo-2,3-diphenylpentane gives two alkenes, A (cis-trans mixture) and C.Upon catalytic hydrogenation, all three of these alkenes (A, B, and C) give 2,3-diphenylpentane. Determine the structuresof A, B, and C; give equations for their formation; and explain the stereospecificity of these reactions
- please help me with also using correct sig figs and labelling units. #2reactions and products for C9H8O4 C9H8O4 + O2 --> C9H8O4 + H2O --> C9H8O4 + HCl --> C9H8O4 + OH- --> C9H8O4 + Na --> C9H8O4 + F -->Alcohols can be converted to alkyl chlorides using SOCl2 with complete inversion of stereochemistry. Using curved arrows draw the stepwise mechanism for chlorination of an alcohol. Be sure to include lone pairs necessary to the mechanism steps and any non-zero formal charges.
- b. In a strongly basic solution, the starting material again converts into a molecule with the molecular formula C6H12OC6H12O. but with a completely different structure.Compound X is insoluble in cold KMnO4, Br2 in CCl4, and conc. H2SO4. Compound X is most likely: a. an alkane b. none of these c. an alkene d. an alcohol e. an alkyl halide Indicate which of the ff. statements regarding nucleophilicity is incorrect. F- is more nucleophilic, hence, more reactive towards methyl iodide than Cl-. Second row elements are more nucleophilic than first row elements of comparable basicity. The rate of SN2 reaction may be markedly affected by the nucleophilicity of the attacking atom. Nucleophilicity is the affinity of a nucleophile to an electrophilic carbon Which of the following alkynes can be deprotonated by NaNH2 in liquid NH3? 3-Methylhex-2-yne Pent-2-yne 3-Methylbutyne none of these Hex-3-yneKMnO4, warm, conc'd reacts with hept-1-ene to yield __________. CO2, hex-1-ene CO2, hexanoic acid Formic acid, pentanoic acid Ethanoic acid, pentanal Formic acid, hexanone
- Complete the reaction map by providing the answer from A-E. Write the IUPAC name of the products. conc HNO B conc H,SO, B, D FeBrs conc HNO, E AIC conc H2SO. conc H2SO4Build a model of methylcyclohexane, and use the model to complete the following Newmanprojections of methylcyclohexane in the chair conformation: a. When the methyl group is in an axial or equatorial (circle one) position, the molecule is inits lowest potential energy conformation. b. Label one Newman projection above anti and the other gauche to describe the relationshipbetween the methyl group and C3 of the ring. c. In general, which is a lower PE conformation, anti or gauche? d. Explain how your answer to b and c provide an explanation for why it is more favorable fora large group to be in an equatorial than an axial position.Rank the following groups in order of decreasing priority. −CH=CH2, −CH3, −C≡CH, −H







