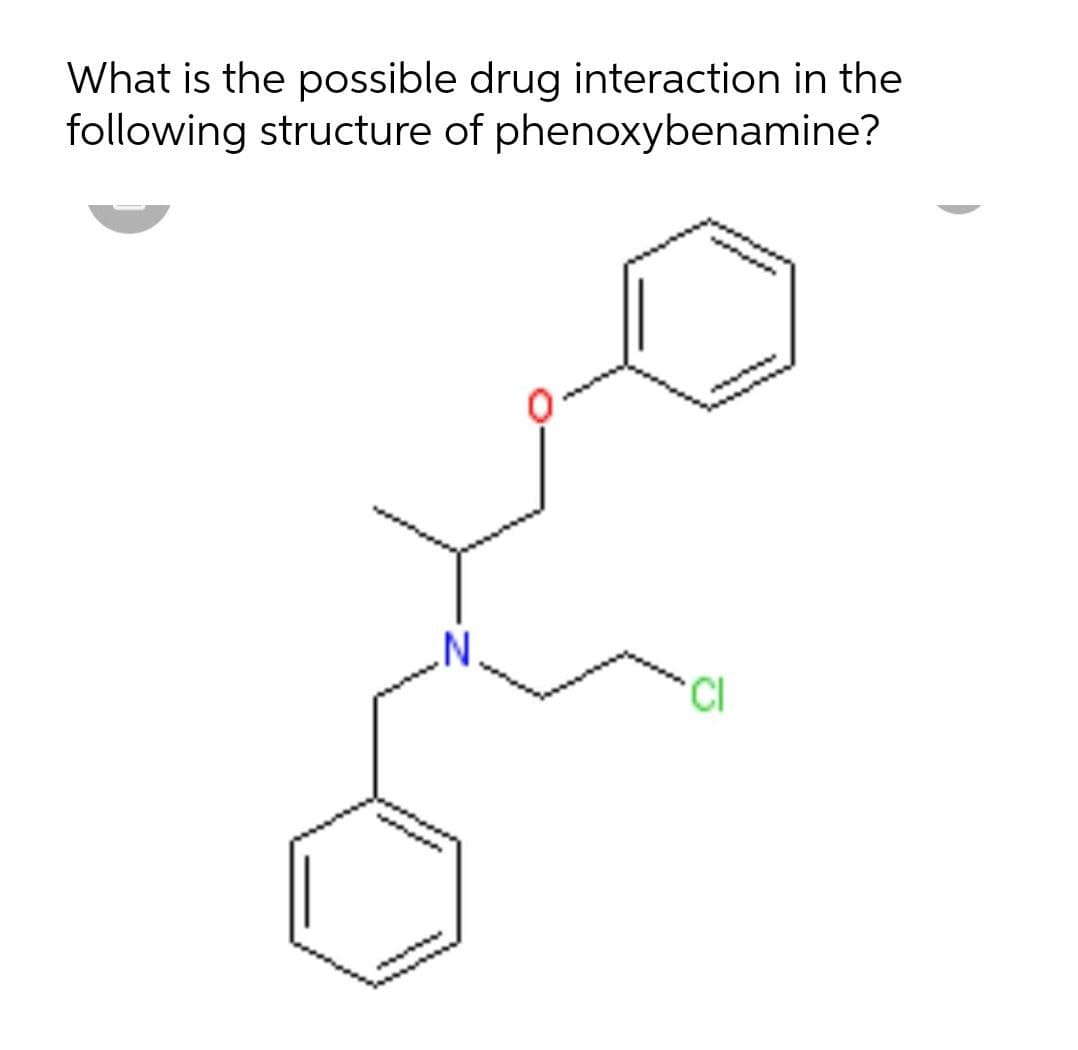
What is the possible drug interaction in the following structure of phenoxybenamine?
Q: Question 2 You are using molecular modelling software to examine the X-ray crystal structure of the…
A: Beta-Adrenoreceptor: They are GPCR and contain seven transmembrane helices. It is widely distributed…
Q: What type of monomers make up proteins? What are protein polymers called?
A: Note - Hi ! Thank you for the question. We are authorized to answer one question at a time. Since…
Q: 1. A student, halfan hour after the dinner, containing about 150 g of carbohydrates, 20 g of fat,…
A: Hi! Thank you for the question. We are authorized to answer one question at a time, since you have…
Q: Which of the following is a product of the first stage of the pentose phosphate pathway? Group of…
A: NADPH : Phosphorylated version of the coenzyme NADH Pentose Phosphate Pathway : PPP 1st stage of PPP…
Q: Explain which of the following substances ATP, CoA-SH, FAD and NAD+ have the subunits in their…
A: The nucleic acid polymer has nucleotide as its monomeric unit. synthesis of nucleotides is an…
Q: if they are true or false? Kindly leave a short explanation. Many thanks! 1. Unlike carbohydrates…
A: A biomolecule, also known as a biological molecule, is one of the many compounds created by cells…
Q: 4. How does compromised pyruvate kinase activity lead to anemia ?
A: Anemia is a disorder in which the body does not have enough healthy red blood cells to transport…
Q: Why is it difficult to accurately estimate Km and Vmax values from a Michaelis-Menten plot…
A: The rate of enzyme catalyzed reaction that is the enzyme kinetics can be expressed by the…
Q: 5. Compare and constrast the energy content of fats, carbohydrates and proteins.
A: Carbohydrates, proteins and fats are three major form of biomolecules found in living beings. These…
Q: 4
A: The general mechanism is the reversible inhibition that takes place due to the formation of…
Q: 4. During a lunch at a MeDonald's outlet, an office employee received about 350 g of carbohydrates…
A: "Since you have asked multiple questions, we will solve the first three subparts of the question for…
Q: Explain the mechanism of Warburg effect and how it benefits cancer cells
A: Cancer means uncontrolled cell growth. This uncontrolled cell growth may cause a lump of cell or…
Q: . ) In one (1) sentence point out a key structural similarity and difference in each of the…
A: Nucleic acids are constituted of nucleotides linked via phosphodiester linkages while proteins are…
Q: BACKGROUND A 2-year-old black girl is being seen by the hematologist after her pediatrician found…
A: Due to the lack of mitochondria, there is no TCA cycle occurring in the RBCs. So, RBCs obtain the…
Q: Please explain what happened in the reaction. How did H2SO4 and 3H2O reacted with the glucose?
A: Carbohydrates are polyhydroxy aldehydes or ketones or compounds that yield them on hydrolysis.…
Q: c. (i) Which enzyme in prokaryotes synthesizes the primers? On which strand (leading or lagging…
A: DNA replication, or copying of a cell's DNA, is semiconservative, which means that each strand of…
Q: Which statement is correct about expression of a gene regulated by Gal4? O Galactose increases gene…
A: In yeast the transcriptional activator GAL4 binds to the upstream activating sequence of the gal…
Q: Which statement does NOT describe a general function of the pentose phosphate pathway? Group of…
A: The pentose phosphate (hexose monophosphate shunt) process is more complicated than glycolysis.…
Q: Question 4 Match the following descriptions to the given choices v Synthesized from a steroid…
A: Vitamins are essential nutrients that are important for the protection of body from diseases.…
Q: Review method used to increase the solubility of a drug under the following headings co solvents PH…
A: Bioavailability is a powerful determinant of drug absorption. It represents the administered dose…
Q: is used in gluconeogenesis that bypasses step 1 of Glycolysis in the production of free glucose from…
A: Gluconeogenesis is a process that transform non-carbohydrate substrate into glucose.the principal…
Q: What is the difference between Raman Spectroscopy and SERS ( Surface Enhanced Raman Spectroscopy).
A: Introduction: It is the most powerful tool to study the interactions between matter and…
Q: Using a 1 cm cuvette, the absorbance at 260nm of your double-stranded DNA sample is 0.15. What is…
A: Lambert-Beer's law can be used to calculate the concentration of DNA in a sample. It states that the…
Q: Indicate at what step number in the glycolysis pathway the following event occur and why? a. First…
A: Isomerisation is defined as the reaction which alters a compound into its simple isomers which…
Q: How to calculate the amount of myoglobin in grams from a 2.0 ml sample of protein extract???
A: Beer Lamberts law states that value of Absorption at a particular wavelength of light by an analyte…
Q: (continued) B. From a metabolic engineering perspective, why did the researchers heterologously…
A: Zymomonas mobilis is a gram negative facultative anaerobic bacteria. It ferments the pyruvate into…
Q: An individual developed a condition characterized by progressive muscular weakness and aching muscle…
A: This shuttle functions in the transporting the fatty acids present in the cytosol to the…
Q: Which amino acid(s) are more commonly found at the At which position(s) are amino acids limited to…
A: Proteins are composed of amino acids, which are bound together by peptide linkage. Amino acids…
Q: do aklaloids minic neutrotransmitters in our bidy? amines contain: a) nitrogen atom b) an acid c)…
A: Note : Hi ! Thank you for the question. We are authorized to answer one question at a time. Since…
Q: 2. Explain the difference between saturated and unsaturated fats.
A: Fats often refers to triglycerides which is esters of glycerol and fatty acids. One glycerol can be…
Q: Question 6 Match the following lipids with their functions v Bile acids A. signaling molecules…
A: Lipids are biomolecules that include fats, waxes, oils, hormones, and certain components of…
Q: Which among the following The colored solution formed as a positive result for the Biuret test is…
A: A polypeptide chain has amino acids linked together by a peptide backbone.
Q: Given Tagatose, Briefly explain its expected reaction (based on their structural formula) to the…
A: The Molisch's test, Fehling's test, and Bial's test are the qualitative tests for carbohydrates. In…
Q: Look at the structure of the disaccharide shown. Name the type of bond which is present. CH2OH H он…
A: Disaccharides exist in more than one chemical conformational structure. The alpha and beta forms of…
Q: In the RBCs of the patient described above, which of the following would be expected? And give the…
A: Pyruvate kinase enzyme deficiency typically can manifest clinical symptoms on red blood cells…
Q: Direct methanol fuel cells (DMFCS) have shown some promise as a viable option for providing "green"…
A: The value of Delta G indicates whether a reaction has a tendency to go forward. From this value, we…
Q: 1. If the Km of an enzyme for substrate A is 1 x 106 and for substrate B, is 4 x 10°, it means a.…
A: The Michaelis constant, Km, varies considerably from one enzyme to another, and also with different…
Q: On the right the Hill plot com- (b) pares the O2 binding properties of Hb Ya- kima with those of HbA…
A: The Hill plot given in the diagram shows the allosteric regulation and affinity of the different…
Q: the structure of a soap molecule, use the concept of intermolecular forces to explain why we use…
A: Soap molecules has hydrophilic head and a hydrophobic tail, they are composed of long chains of…
Q: A solution has a pH of 5.4. What is its pOH O5.4 8.6
A: The hydrogen ion concentration [H+] and hydroxide concentration [OH–] in a aqueous solution have an…
Q: c. Ribulose 5-phosphate levels would decrease. d. NADH to NAD+ ratios would decrease. e.…
A: In the RBC's of the patients certain changes are observed. Ribulose 5 Phosphate levels would…
Q: For you, what is the contribution around 18-1900 of golden era in biochemistry? Explain
A: Introduction: The term biochemistry refers to the study of the chemistry of living things including…
Q: find stereochemistry, (from the structure) , rotational bond and geometrical / optical isomer for…
A: Phenyotin is a synthetic compound and also called Diphenylhydantoin and it is a potent…
Q: Question #1: Please describe the specific enzymes that you are familiar with that are involved in…
A: " Since you have asked a multiple question , we will solve the first question for you. If you want…
Q: Explain which of the following substances ATP, CoA-SH, FAD and NAD+ have the subunits in their…
A: Adenosine triphosphate (ATP) serves as the energy currency of the cell while FAD/FADH2, NAD+/NADH,…
Q: Indicate what step each of the events in the glycolysis pathway the following takes place: a. First…
A: The first step in breaking down glucose to obtain energy for cellular metabolism is glycolysis.…
Q: how allosteric regulation is fundamentally different from competitive/uncompetitive/mixed inhibition…
A: Some categories of enzymes exhibit kinetic properties that cannot be studied using Michaelis-Menten…
Q: Consider the Michaelis-Menten equation, below: Vmar S V. k + [S] %3D What is the relationship…
A: [S] : Substrate concentration V= Vmax[S]/(Km+[S]) Vmax: Maximum velocity Km: [S] at which V is…
Q: Why some of the dissolved plant matter travels faster with the alcohol up the filter paper than the…
A: Answer: Ethanol has high eluting nature, and also has higher viscosity through which it can travel…
Q: 4. Synthesis of basic corticosteroids. Steroid: A. Testosterone. B. Aldosterone. C. Pregnenolone. D.…
A: Introduction: Corticosteroids are steroid hormones that are synthesized in the adrenal cortex. It is…
Step by step
Solved in 2 steps with 1 images

- Explain this quesiton in descrition format whether non-barbiturate drug classes are labeled at a lower or higher selection compared to barbiturates?Define the term sulfasalazine?What are the Respective Structure Activity Relationship(SAR) of Sedative-hypnotic drugs? Please briefly explain at your own words.
- what are the Classification (Chemical and Therapeutic) of the Gliclazidewhat are the Drug-drug or drug-food interactions of gliclazideFind the innovator brands of these drugs, leading brands in the Philippine market and the taxonomical source of these drugs: A. Biperiden B. Procyclidine C. Trihexyphenidyl D. Isopropamide E. Tropicamide F. Diphenamil G. Ethopropazine I. Papaverine J. Meclizine (Bonamine) K. Dicycloverine L. Oxybutynin M. Loperamide N. Diphenoxylate O. Morphine P. Pralidoxime Q. Diacymonoxime
- How do you synthesize Chloramphenicol and show the mechanism? Explain step by step please. (If you can 1 sentence is enough.) (Drug Chemistry)How can you determine the expiry date of a drug? Is there any role for the ICH on these stability studies? Discuss.What is the current position of the Food and Drug Administration regarding bisphenol A?

