What kind of bond(s) is/are shown in the molecule below? NH2 N. O Covalent bonds only O Hydrogen bonds only O lonic bonds only Covalent and hydrogen bonds ZI
What kind of bond(s) is/are shown in the molecule below? NH2 N. O Covalent bonds only O Hydrogen bonds only O lonic bonds only Covalent and hydrogen bonds ZI
Chapter4: Forces Between Particles
Section: Chapter Questions
Problem 4.104E
Related questions
Question
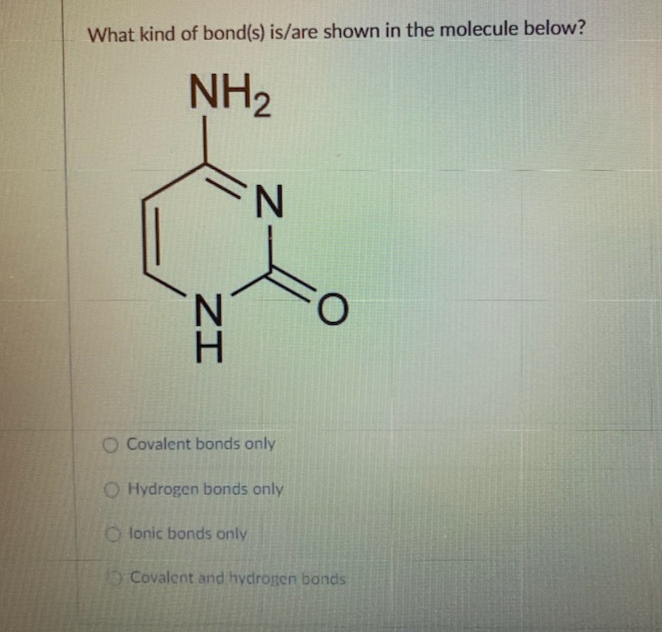
Transcribed Image Text:What kind of bond(s) is/are shown in the molecule below?
NH2
N.
O Covalent bonds only
O Hydrogen bonds only
O lonic bonds only
Covalent and hydrogen bonds
ZI
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Recommended textbooks for you


General, Organic, and Biological Chemistry
Chemistry
ISBN:
9781285853918
Author:
H. Stephen Stoker
Publisher:
Cengage Learning

Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning


General, Organic, and Biological Chemistry
Chemistry
ISBN:
9781285853918
Author:
H. Stephen Stoker
Publisher:
Cengage Learning

Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning
