Chapter87: Steam Distillation
Section: Chapter Questions
Problem 2P
Related questions
Question
100%
Justify your answer:
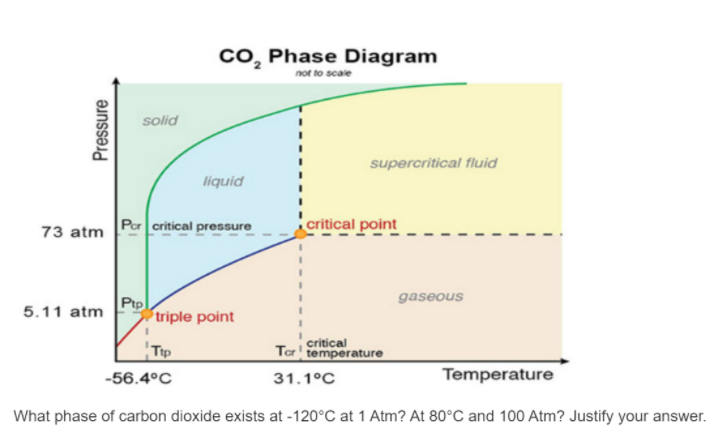
Transcribed Image Text:cO, Phase Diagram
not to scale
solid
supercritical fluid
liquid
Por critical pressure
critical point
73 atm
Ptp
triple point
gaseous
5.11 atm
Tip
critical
Tar' temperature
-56.4°C
31.1°C
Temperature
What phase of carbon dioxide exists at -120°C at 1 Atm? At 80°C and 100 Atm? Justify your answer.
Pressure
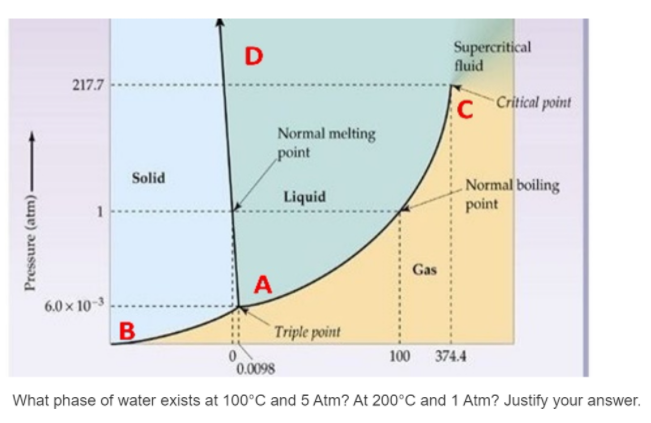
Transcribed Image Text:D
Supercritical
fluid
217.7
Critical point
Normal melting
point
Solid
. Normal boiling
point
Liquid
1
Gas
A
6.0 x 10-3
B
Triple point
100 374.4
0.0098
What phase of water exists at 100°C and 5 Atm? At 200°C and 1 Atm? Justify your answer.
Pressure (atm)
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Recommended textbooks for you

EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:
9781305446021
Author:
Lampman
Publisher:
CENGAGE LEARNING - CONSIGNMENT

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:
9781305446021
Author:
Lampman
Publisher:
CENGAGE LEARNING - CONSIGNMENT

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning