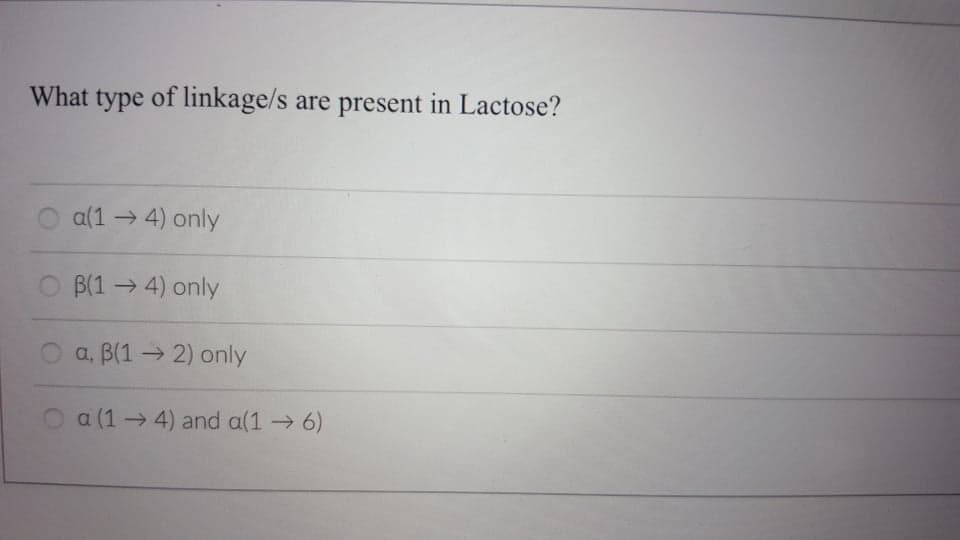
What type of linkage/s are present in Lactose? a(1 → 4) only O B(1→4) only O a, B(1→ 2) only O a (1→ 4) and a(1 6)
Q: What is the effect of the NDM gene?
A: NDM (New Delhi Metallo-β-lactamase) gene, found in Enterobacteriaceae, is a genetic element encodin...
Q: With respect to the above molecular outline, what chemical properties would you expect for the (a) h...
A: A fatty acid is a carboxylic acid with an aliphatic chain. Soap is sodium salt of fatty acid.
Q: Solute contributes to the movement of water betweencells and the surrounding medium by ________.a. o...
A: Living beings are made up of cells. The cells contain several organelles and the cytoplasm in which ...
Q: A two-step pathway that is activated by the secretion of glucagon and adrenaline
A: Glucagon and adrenalin, both induce an increase in hepatic glucose levels. However the mechanism of ...
Q: 1. How do you prepare 25 mL of a 0.4X solution of SDS from a ...
A: Using formula C1V1= C2V2, we can calculate the Molarity, w/v, %, or any other concentration of the s...
Q: Will a decrease in elevation shift the binding curve of hemoglobin? Will it shift the curve to the ...
A: Binding curve of haemoglobin: this is also called as oxygen dissociation curve which is plotted bet...
Q: 2. The standard state in biochemistry is slightly different than in Physical Chemistry. In biochemis...
A: "Since you have posted a question with multiple sub-parts, we will solve first three subparts for yo...
Q: 1. The diagram below shows the chemical structure of two sugar molecules. СНО b. CHZOH а. НО- H. C=0...
A: As you have asked multiple sub part questions ,we are are supposed to solve only first three sub pa...
Q: Calculate the pI value of aspartate. (iii) Calculate the pI value of arginine.
A: Amino acids are monomers that make proteins. The amino acids have a central carbon to which a variab...
Q: What is the effect of cholesterol when they are embedded in membranes? 1. Cholesterol alters the...
A:
Q: 1. Amino acids are not stored in the body. Describe how excess amino acids are processed in the cell...
A: Amino Acids are organic compounds that combine to form proteins, which is why they are called protei...
Q: Which of the following statements are true about stacking and separating gels? a. The holes is t...
A: In the SDS-PAGE technique, stacking gel is a low concentration polyacrylamide gel that is placed on ...
Q: After sufficiently long and intense exercise, EPOC can be observed for more than a day True False
A: Excessive Postexercise Oxygen consumption is the phenomenon by which our body continues to burn calo...
Q: are all the protein separated properly using isoelectric focusing? why or why not?
A: The proteins are composed of amino acids that have ionizable side chains with characteristic pKa val...
Q: Which statement is NOT true of sterols? Question 8 options: Sterols have a structure that incl...
A: Sterols are fat molecules that are commonly found in cells. Sterols are non-polar molecules that are...
Q: 1. Cite five diseases that could be diagnosed with the determination of enzyme concentration. 2. Giv...
A: Note : Hi ! Thank you for the question. We are authorized to answer one question at a time. Since yo...
Q: (a) Propose a biosynthetic pathway to the secondary metabolite, B. OH O H. HO Metabolite B
A: The given metabolite structure is derivative of Salicylaldehyde derivative and synthesized by plant ...
Q: Explain why the maximum initial reaction rate cannot be reached at low substrate concentrations
A: Multiple questions asked. I will answer the first question, as allowed by guidelines. Please repost ...
Q: You are a dietitian working in a hospital. You noticed that the resident physician typically orders...
A: Malnutrition is a risk factor for patient morbidity and mortality on its own. It has been linked to ...
Q: What is the Amino Acid Sequence of 3'-AUG GUA CUC UUA ACA CCA GUG GAA AAA-5'
A: Proteins are polymers made of amino acids. The cellular process by which proteins are synthesized by...
Q: Indicate the COOH-pKa value of the HAY tripeptide on the titration curve. Titration curve of tripept...
A: The titration curve of peptides indicates the buffering zones and the pKa value of each ionic specie...
Q: Question 6 options: Cholesterol is a sterol that is commonly found in mammals. Sterols are pre...
A: Cholesterol is a steroid that constitutes of a hydrocarbon tail, four hydrocarbon rings and a hydro...
Q: CAMP Ga-O GTP OFF GDP Ligand GDP Adenylate Cyclase Ga O ON PKA GTP Mobilie Glycogen stores through g...
A:
Q: 11. Which of the following descriptions of 24:6 n-3 fatty acid is correct? A. It is an omega-6 fatty...
A: The essential fatty acid cannot be synthesized by our body but they are required for the maintenance...
Q: Which of these are soluble in water, vitamin C or vitamin K? Explain your answer.
A: Carbohydrates, lipids, and proteins are macronutrients that the body requires in greater quantities ...
Q: Some cancers are detectable with a simple blood test (if the cancer cells secrete a chemical that ci...
A: Hi thanks a lot for asking the details of the solution. The solution is correct, and amount of chemi...
Q: How protein activity can be modulated by alternative splicing ?
A: The process of selecting distinct combinations of splice sites within a messenger RNA precursor (pre...
Q: Compare and contrast the location and pH sensitivity of receptor-ligand interaction in the LDL and t...
A: Endocytosis occurs when a coat protein (typically clathrin) on the cytoplasmic side of the plasma me...
Q: What Determine the amount of mRNA that is available for translation ?
A: A single-stranded RNA molecule called messenger RNA (mRNA) is complementary to one of a gene's DNA s...
Q: The median size protein in humans is 8600 amino acids. How does this compare to if our genome was ra...
A: Proteins are polymers of amino acids linked by peptide/amide bonds. It is expressed inside the cells...
Q: QUESTION 7 The monosaccharides shown below are H. HO H -HO- HO H H- HO H но HO- HO- CH,OH CH,OH O En...
A: Isomers are molecules with the same molecular formula, but different arrangement of atoms. Isomers a...
Q: (i)Define initial velocity (ii) Describe how you would calculate initial velocity from a graph of pr...
A: Initial velocity of a reaction is a very important parameter of an enzyme. It is denoted by Vo which...
Q: In regards to the protein structure, explain what is meant by “tertiary structure.” What are the mai...
A: There are four levels of protein structure are important to attain the final 3- dimensional folded s...
Q: Choose the plot that best reflects the activity of an enzyme inhibited irreversibly. 100% A B C D ti...
A: Enzyme inhibitors are the substances that bind to enzyme either at active site or allosteric site so...
Q: Please write primary amino acid sequence with minimum number of amino acids that leads to formation ...
A: The primary amino acid sequence is simply the linear arrangements of amino acids that make up that ...
Q: In less than 3-5 sentences, explain the following attributes of water that makes it a polar solvent:...
A: Introduction: A water molecule is made up of three atoms which includes one oxygen and two hydrogen ...
Q: H. Draw a plot showing reaction velocity as a function of substrate concentration for Ks = 50 µM, Vm...
A: First we need to find the values of (V0) at various substrate concentrations ([S0]). For this we dra...
Q: Referencing the image above, you are designing an experiment to determine which food source your yea...
A: In retrospect, the fact that the rate of fermentation is dependent on yeast concentration is not sur...
Q: G1: CASE ANALYSIS A 28 year old woman is seen with an easy fatigue for a couple of months. She is c...
A: Symptoms mentioned here for 28 years old lady with two babies in 2 years Symptom 1: Taste of eating ...
Q: Calculate the pH of a solution containing 0.0314 M malic acid and 0.020 M potassium hydrogen malate....
A: Given Values: Ka1 = 3.48×10-4 Ka2 = 8×10-6 Conc. of malic acid = 0.0314 M Conc. of potassium hydroge...
Q: INTRUCTIONS: - Do not copy here in BARTLEBY or GOOGLE - PLEASE ANSWER PROPERLY Failed to follow inst...
A: The most prevalent lipids in nature are fats and oils. They give life energy, protect bodily organs,...
Q: THE MAIN BUFFERS OF THE CELLULAR FLUIDS ARE EXCEPT a. HPO4 & H2PO4- b. НСОз- & Н2СОЗ c. NH40H & NH4C...
A: Introduction: The buffering system in body fluids provide an immediate response in fluctuations in p...
Q: Please explain in detail a chemical reaction between rRNA and another biomolecule
A: rRNA are the structural components of ribosomes. Hence rRNA does not generally participate in chemic...
Q: Please give the color of each analysis indicating positive result and substance formed causing the c...
A: Since we only answer up to 3 sub-parts, we’ll answer the first 3. Please resubmit the question and s...
Q: Its principal function is to increase the concentration of glucose in the blood by speeding the glyc...
A: Glucose is the simplest source of energy for the body. The glucose which is obtained through diet is...
Q: Analyzing: Analyze the following pair of monosaccharides and determine which of the following is an ...
A: Since there are multiple questions and they are not interlinked, as per our company guidelines only ...
Q: how would you identify whether or not complete resolution of the three amino acids has been achieved
A: An amino group and an acid group-containing organic molecules are called Amino acids. when ...
Q: An E. coli replication fork is shown in Figure 2.2. III 3' IV 5 II Figure 2.2 (i) Identify the laggi...
A: The nucleic acid polymer has nucleotide as its monomeric unit. synthesis of nucle...
Q: Give an example and explain why and how changing just one amino acid in a protein's sequence can aff...
A: Proteins (polymers) are macromolecules made up of subunits of the amino acid (the monomers ). These ...
Q: 10. Which of the following statement(s) is/ are true for the molecule below? (1) It can be found on ...
A: Structure of the monosaccharides can be represented using the Fischer projection or Haworth projecti...
Step by step
Solved in 2 steps with 1 images

- Hi what does this have to do with lactose or lactose intoStarting from glutamine, glycine, aspartate, N 10-formyi-THF, how many ATP- equivalents are required for purine nucleotide synthesis? 2 8 4 6Many people who are lactose intolerant can eat yogurt, which is prepared from milk curdled by bacteria, without any digestive problems. Give a reason why this is possible. (Hint: Read the label on each of several yogurt containers. Do the ingredients make a difference?)
- In N-linked glycoproteins, the sugar molecule is usually bound to a Asp b Ser c Asn d Thrwhere is a reducing end in this image? I initially thought it'd be on the middle oxygen connecting the lactose molecules since it's the only reducing sugar, but that was incorrect for some reason.All sugar residues are in their D-isomeric forms. The second sugar residue is an epimer of: A. altrose at C2. B. glucose at C3. C. altrose at C4. D. glucose at C4. E. talose at C4.
- Unbranched homopolymer of N-acetyl glucosamine is : 1.Cellulose 2.Chitin 3.Curcumin 4.Concanavalin AWhen a purine or pyrimidine is linked through a b-Nglycosidic link to C-1 of a pentose, the molecule is calleda _________________________.Draw the Haworth projection of the following sugar molecules: D-galactose D-fructose D-mannose
- There is energy requirement for every amino acid added in a growing polypeptide chain. Is this true or false?Why do lactose intolerant people have problems when they consume dairy products? Be sure to frame your answer within the context of the genetics and biochemistry involvedDraw both possible disaccharides of D-psicose and D-mannose that are joined by an α(1→4) glycosidic linkage. Are either of them now a reducing sugar?



