1. The diagram below shows the chemical structure of two sugar molecules. СНО b. CHZOH а. НО- H. C=0 Но H. H HO-H НО —Н CH2OH CH;OH a. How many chiral carbons are in sugars a and b? b. Are they D- or L- sugars? Explain your answer. c. What is the name of this kind of representation of the sugars in the diagram?
1. The diagram below shows the chemical structure of two sugar molecules. СНО b. CHZOH а. НО- H. C=0 Но H. H HO-H НО —Н CH2OH CH;OH a. How many chiral carbons are in sugars a and b? b. Are they D- or L- sugars? Explain your answer. c. What is the name of this kind of representation of the sugars in the diagram?
Biology (MindTap Course List)
11th Edition
ISBN:9781337392938
Author:Eldra Solomon, Charles Martin, Diana W. Martin, Linda R. Berg
Publisher:Eldra Solomon, Charles Martin, Diana W. Martin, Linda R. Berg
Chapter3: The Chemistry Of Life: Organic Compounds
Section: Chapter Questions
Problem 5TYU: A monosaccharide designated as an aldehyde sugar contains (a) a terminal carboxyl group (b) an...
Related questions
Question
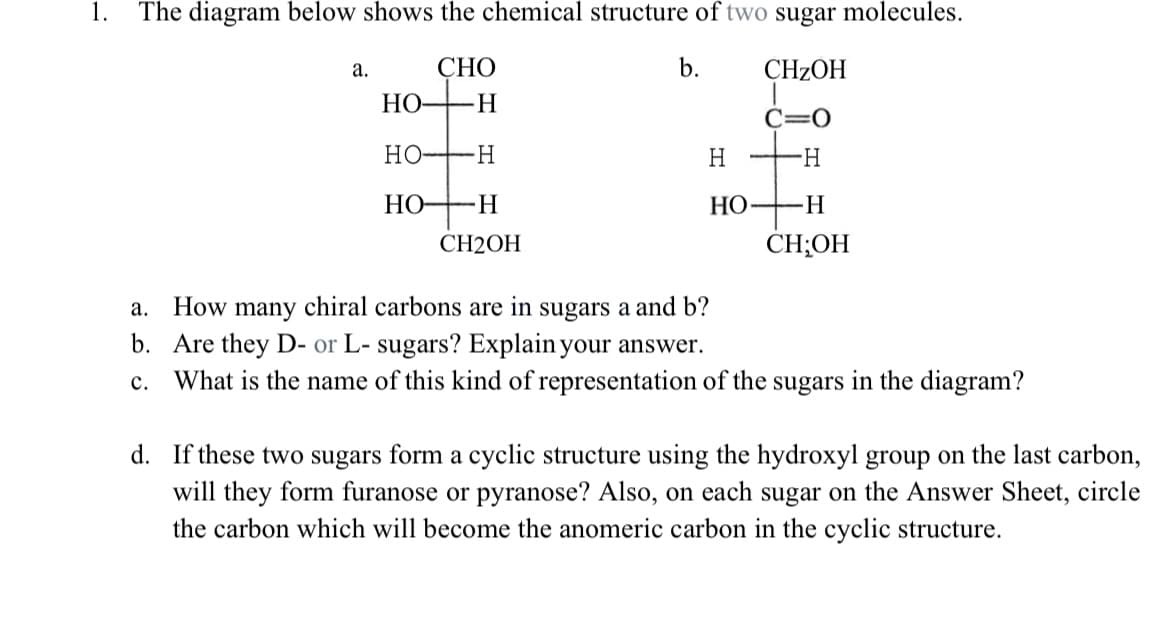
Transcribed Image Text:1.
The diagram below shows the chemical structure of two sugar molecules.
СНО
b.
CHZOH
а.
HO
C=0
НО—Н
H
-H-
Но-
НО —Н
CH2ОН
CH;OH
How many chiral carbons are in sugars a and b?
b. Are they D- or L- sugars? Explain your answer.
What is the name of this kind of representation of the sugars in the diagram?
а.
с.
d. If these two sugars form a cyclic structure using the hydroxyl group on the last carbon,
will they form furanose or pyranose? Also, on each sugar on the Answer Sheet, circle
the carbon which will become the anomeric carbon in the cyclic structure.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Recommended textbooks for you

Biology (MindTap Course List)
Biology
ISBN:
9781337392938
Author:
Eldra Solomon, Charles Martin, Diana W. Martin, Linda R. Berg
Publisher:
Cengage Learning

Biology: The Unity and Diversity of Life (MindTap…
Biology
ISBN:
9781337408332
Author:
Cecie Starr, Ralph Taggart, Christine Evers, Lisa Starr
Publisher:
Cengage Learning

Biology: The Unity and Diversity of Life (MindTap…
Biology
ISBN:
9781305073951
Author:
Cecie Starr, Ralph Taggart, Christine Evers, Lisa Starr
Publisher:
Cengage Learning

Biology (MindTap Course List)
Biology
ISBN:
9781337392938
Author:
Eldra Solomon, Charles Martin, Diana W. Martin, Linda R. Berg
Publisher:
Cengage Learning

Biology: The Unity and Diversity of Life (MindTap…
Biology
ISBN:
9781337408332
Author:
Cecie Starr, Ralph Taggart, Christine Evers, Lisa Starr
Publisher:
Cengage Learning

Biology: The Unity and Diversity of Life (MindTap…
Biology
ISBN:
9781305073951
Author:
Cecie Starr, Ralph Taggart, Christine Evers, Lisa Starr
Publisher:
Cengage Learning

Biology: The Dynamic Science (MindTap Course List)
Biology
ISBN:
9781305389892
Author:
Peter J. Russell, Paul E. Hertz, Beverly McMillan
Publisher:
Cengage Learning

Biology 2e
Biology
ISBN:
9781947172517
Author:
Matthew Douglas, Jung Choi, Mary Ann Clark
Publisher:
OpenStax