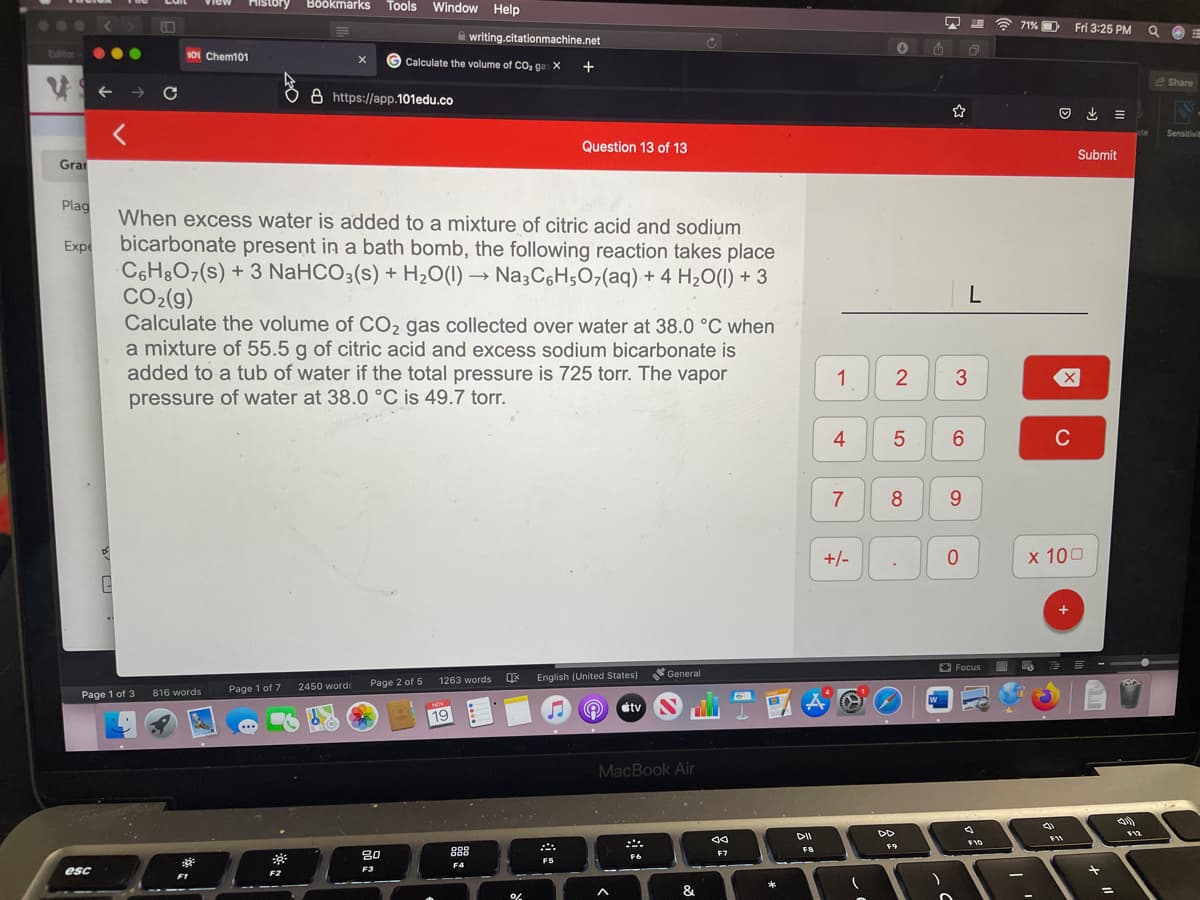
When excess water is added to a mixture of citric acid and sodium bicarbonate present in a bath bomb, the following reaction takes place C6H&O7(s) + 3 NaHCO3(s) + H20(1) → Na3C6H507(aq) + 4 H2O(1) + 3 CO2(g) Calculate the volume of CO2 gas collected over water at 38.0 °C when a mixture of 55.5 g of citric acid and excess sodium bicarbonate is added to a tub of water if the total pressure is 725 torr. The vapor pressure of water at 38.0 °C is 49.7 torr.
When excess water is added to a mixture of citric acid and sodium bicarbonate present in a bath bomb, the following reaction takes place C6H&O7(s) + 3 NaHCO3(s) + H20(1) → Na3C6H507(aq) + 4 H2O(1) + 3 CO2(g) Calculate the volume of CO2 gas collected over water at 38.0 °C when a mixture of 55.5 g of citric acid and excess sodium bicarbonate is added to a tub of water if the total pressure is 725 torr. The vapor pressure of water at 38.0 °C is 49.7 torr.
Chapter79: Solubility
Section: Chapter Questions
Problem 1P
Related questions
Question
Transcribed Image Text:Tools Window Help
View
History
Bookmarks
...
71% D
Fri 3:25 PM
A writing.citationmachine.net
Editor
101 Chem101
Calculate the volume of CO, ga: X
+
e Share
->
8 https://app.101edu.co
ate
Sensitivit
Question 13 of 13
Submit
Gra
Plag
When excess water is added to a mixture of citric acid and sodium
Expe
bicarbonate present in a bath bomb, the following reaction takes place
C6H&O7(s) + 3 NaHCO3(s) + H2O(1) → Na3C6H;07(aq) + 4 H20(1) + 3
CO2(g)
Calculate the volume of CO2 gas collected over water at 38.0 °C when
a mixture of 55.5 g of citric acid and excess sodium bicarbonate is
added to a tub of water if the total pressure is 725 torr. The vapor
pressure of water at 38.0 °C is 49.7 torr.
L
4
6
C
7
8
+/-
x 100
O Focus
General
书
1263 words
English (United States)
2450 word:
Page 2 of 5
Page 1 of 7
Page 1 of 3
816 words
tv
NOV
19
MacBook Air
DII
F10
F9
80
888
FB
F7
F6
F5
F4
esc
F2
F3
|
&
3.
1,
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:
9781305446021
Author:
Lampman
Publisher:
CENGAGE LEARNING - CONSIGNMENT


EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:
9781305446021
Author:
Lampman
Publisher:
CENGAGE LEARNING - CONSIGNMENT
