Q: Which of the following aqueous solutions will have the highest conductivity? * 0.5M NaCl 0.5M…
A: Conductivity nothing but number of conducting ions that are present in a solution As number of…
Q: Which ions can cause water hardness? Group IA cations such as Na*, Li* and K* O Ca2* and Mg2+ Ca2+…
A: There are two types of water, hard water, and soft water. This based on the difference in minerals…
Q: What are the uses of polyiodides? What are the importance of polyiodides?
A: The answer to the following question is-
Q: Rank the following solutions from brightest (1) to weakest/no light (6) when the solution's…
A: Equivalent conductance is inversely proportional to the normality of the solution and normality is…
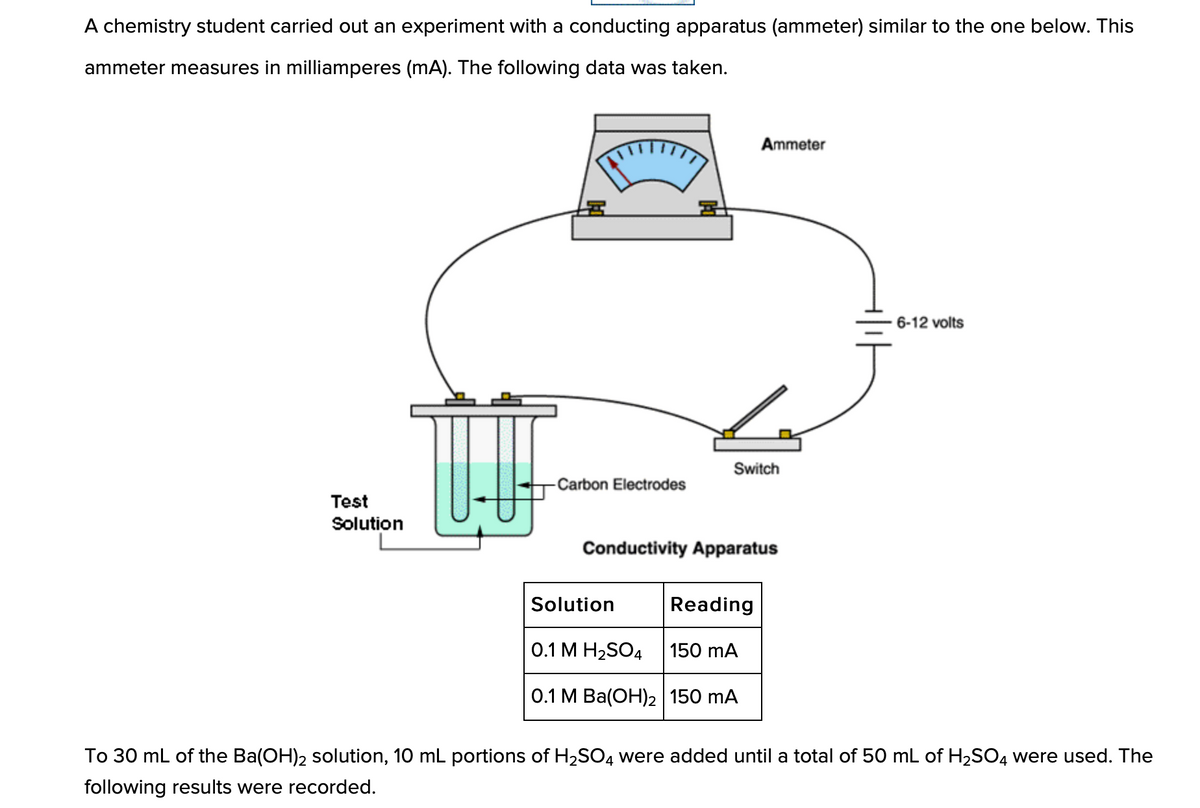
Q: The electrical conductivities of the following 0.100 M solutions were measured in an apparatus that…
A:
Q: What is the main purpose of preparing standard curve for nutrient determination in water? Draw a…
A:
Q: Rank these ions in order of increasing structuring (ordering, organizing) effect on water? a.)…
A: We have to rank the following given ions in order of increasing structuring (ordering, organizing)…
Q: Determine the pOH of a 0.625 M HClO solution. Ka = 3.0 x 10-8
A:
Q: H2PO4 can act as either an acid or a base. Species with this property are called... O Amorphic O…
A:
Q: Theoretical background oh how strength of acids affects electrical conductivity Min 150 words
A: The conductivity of an electrolyte solution is due to the presence of ions in the solution.
Q: 0.10 M NaCl, 0.10 M NaNO3, 0.10 M NaC2H3O2 These all have the same cation, Na+. Which anion…
A: The speed at which the ions move through a gas under the influence of unit electric field is known…
Q: 1. Why is pure dry NaCl a non-conductor of electricity but when dissolved in water, a good conductor…
A: Conductors are those compounds which allow the flow of current or heat through them. Free electrons…
Q: Conductivity increases with more charged ion and decreases when there are fewer ions. Explain how…
A: NaHCO3 is base with and HCl is a strong acid. They reacts together and neutralises to give salt.
Q: What are the major components in water that make hard water hard ? Ca2+&Mg2+, No3-& Mg2+, Na+&Fe3+,…
A: The major components in water that make hard water hard among these : 1) Ca2+ & Mg2+ 2) No3-…
Q: Write the equations that represent the first and second ionization steps for tellurous acid (H2TEO3)…
A: Acid is a substance which can easily lose proton and become stabilise and base gain that proton.
Q: Which of the following aqueous solutions will exhibit no conductivity at all? * 2 points 0.5M…
A: Electrolytes dissociates into ions in solution and will exhibit conductivity. Wheras a non…
Q: Which compound should have the greatest molar conductivity? O KBr BaBr2 GaBr3 O PbBr4 O They should…
A:
Q: True or False For strong electrolytes, such as hydrochloric acid, the molar conductivity decreases…
A: Strong electrolyte are those substance which dissociate completely. eg: HCl , KCl etc.
Q: Write the balanced equation if Vitamin C is oxidized by I2. That was generated by mixing KIO3 and KI
A: A chemical technique which can be used for the determination of amount of compound present in a…
Q: 1. The solution with the lowest electrical conductivity is .1OM H;S B. 0.10M HNO: C. 0.10M H2SO; D.…
A:
Q: 3. Explain the differences in conductivity between: a) aqueous solutions of acetic acid…
A: We have to explain the conductivity between the following solution
Q: Which one of these parameters measure amount of organic matters in water? Select one: O A. nutrients…
A: (a). Given statement is : Which one of these parameter measure amount of organic matter in water ?…
Q: uestion 8 For strong electrolytes, such as hydrochloric acid, the molar conductivity decreases…
A: The answer is as follows:
Q: State the chemical properties on which the following uses of baking soda are based: (ii) as a soda…
A: Baking soda is the common name of the compound sodium bicarbonate with a molecular formula NaHCO3.…
Q: What is ocean acidification and how is it impacting marine life
A: Ocean acidification is defined as the continuous decrease in the pH of the water in the ocean on…
Q: What type of salt is NH 41? A. neutral B. acidic C. basic
A: The corresponding acid or base can be predicted from a particular salt by the hydrolysis of salt.…
Q: on 2 Which of the following substances would be a poor choice for making conductive solution O NAOH,…
A:
Q: Based on these data, what is the value of the formation constant, Kr, of (Cu(NH3)4*? Express the…
A: Ans: 3x 10^13
Q: If the H+ concentration of a borewater sample was 1.00 x 10-8 M, what is its pH?
A:
Q: What is the pH of .03 M HC2H3O2 (aq)? O 10.07 O 1.52 O 4.74 O 3.13 O 6.27
A: Ka is acid dissociation constant which determines strength of acid. Stronger acid dissociates more…
Q: Which of the following chemicals would be considered "diprotic"? * О СаЗР2 O H2S04 O CH2C12 О Н20
A: Diprotic :- A chemical substance is said to be diprotic if it provides two protons (H+) after…
Q: Vhich of the following substances would be a poor choice for making conductive solutio O NaOH, KCI,…
A: The question is based on the concept of conductivity. We have to identify the solutions which are…
Q: What is the concentration of lithium ions in 0.215 M Lİ2SO4?
A:
Q: Which one of the following aqueous solutions shows the poorest electrical conductivity? A. 1 M NaCl…
A: Strong electrolytes: The strong electrolytes dissociate completely in the aqueous solution. Strong…
Q: Indicator of the acidity or the alkalinity of sewage/wastewater. pH Temperature Dissolved Oxygen…
A: Indicator is a device that tells whether given sample is acidic or alkaline by performing certain…
Q: Would you mix alkali metals and acids? Motivate.
A: Those elements of the 1st group of the periodic table which have electronic configuration ns2np6ns1…
Q: The concentration of charge contributed to a solution by [PO4°] is: O (PO1 [PO, ] O 1/2[PO4-] O…
A: Given: specie is PO43-.
Q: a.) What is [OH−] in a solution with a pH of 9.67? b.)How much heat does your body lose when 1.6 g…
A: [OH] is 4.7 × 10-5
Q: Write equations depicting the behavior of the following Arrhenius acids and bases in water. (Use…
A: HClO2 is an acid. In water it gives proton to water to produce hydronium ion and its anion
Q: Be sure to answer all parts. Enter your answer in scientific notation. Calculate the H30* |…
A:
Q: Write an equation to show how HC204 can act as an acid with HS¯ acting as a base.
A: Bronsted-Lowery acid base theory : Bronsed acid : Proton (H +) donar is acid Bronsted base :…
Q: a) A photon of ultraviolet light moves faster than a photon of microwave light in a vacuum.(TRUE OR…
A: Answer (a) True A photon of ultraviolet light move faster…
Q: The Lewis acidities of silicon halides follow the order SiI4< SiBr4<SiCl4< SiF4. Explain.
A: The Lewis acidities of silicon halides follows the order, SiI4 < SiBr4 < SiCl4 < SiF4.
Q: Calculate the pH at 25 °C of a solution that is 1.00 M in NaClO(aq) and 2.00 M in HClO(aq). For…
A: pH of the solution is 2.87.
Q: 5. What picks up the electrons from a metal which allows them to lissolve in water? H20 molecules…
A: Given statement is : What pick up the electrons from a metal which allows them to dissolve in water…
Q: A student tests the electrical conductivity of a 2.0 M oxalic acid solution and compares it to the…
A: The acids given are oxalic acid i.e COOH-COOH and hydroiodic acid i.e HI. And the conductivity of HI…
Q: Write the equations that represent the first and second ionization steps for telluric acid (H2TeO4)…
A: ⊙ Telluric acid ⇒ H2TeO4 ⇓
Q: Write the equation for the decomposition of the hydrate Na2SO4•10H2O. Label the anhydrous salt.
A: Chemical name of Na2SO4.10H2O is sodium sulfate decahydrate.It is also known as Glauber's salt.On…
Q: 5.1.3 Calculate the pOH of a 2.5 mol/L HCN solution. K 4.9 x 1010 %3D
A: Ka = 4.9 x 10-10 C = 2.5 mol/L
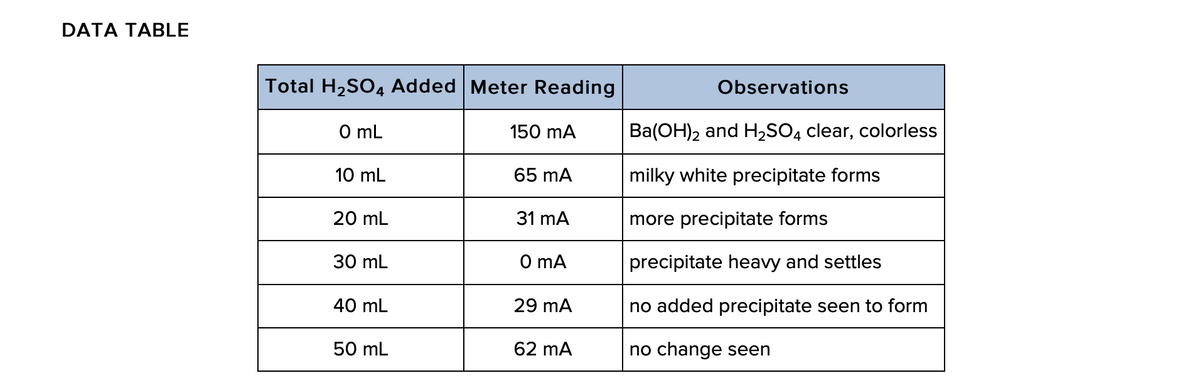
When the conductivity is at a minimum, what must be true about the amount of Ba(OH)2?
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

- A sample is analyzed for chloride by the Volhard method. From the following data, calculate the percentage of chloride present:Weight of sample = 6.0000 g dissolved and diluted to 200 mLAliquot used = 25.00 mL AgNO3 added = 40.00ml of 0.1234MKSCN for back titration = 13.20ml of 0.0930Mpls complete table given the data Determination of Ksp and Molar Solubility1. Add Ca(OH)2 to 250.0 mL distilled water with stirring until equilibrium is achieved. 2. Filter the undissolved precipitate. Measure out 50.0 mL of the supernate into a 250-mL Erlenmeyer flask using a pipette.3. Add a few drops of phenolphthalein indicator and titrate with standardized HCl solution until endpoint is achieved.4. Record the volume the HCl solution used. Perform two more trials.A local company sent you their green alternative for window cleaner to be tested for percent (w/v) acetic acid content. For your experiment, you first standardized your NaOH titrant with 0.8053 g of (99.80 % purity) KHP. You used 40.60 mL of NaOH for your standardization. After that you then analyzed a 10.00 mL sample and found that you needed 43.20 mL NaOH to reach the end point. Summary of results: Standardization Sample analysis KHP Weight (g) 0.8053 g Volume of sample 50.00 mL Purity 99.80% NaOH (mL) used 33.20 mL NaOH (mL) used 40.60 mL Determine the following: Molarity of NaOH % (w/v) acetic acid
- Sources of Error Determine the relationship between the observed/apparent value (EX) VERSUS that of the true value (ET) for the quantity being sought by writing either <, >, or = on the space provided TOPIC: Measured mass of the precipitate 1. Filter paper was dried prior to filtration. EX _____ ET TOPIC: Standardization of Titrant 2. Distilled water was not equilibrated to room temperature before the preparation of NaOH titrant. EX ______ ET TOPIC: Determination of Molar Concentration of each component (Double Indicator Titration) 3. No blank correction EX ______ ETAspirin powder = 0.8110g MW of Aspirin = 180g.mol-1 Volume of 0.5N HCl consumed in back titration = 23.50mL Volume of 0.5N HCl consumed in blank titration = 44.50mL Percent purity (USP/NF) = Aspirin tablets contain NLT 90.0% and NMT 110.0% of the labeled amount of aspirin (C9H8O4) What is the calculated weight (in grams) of pure aspirin?..A 50.00 (±0.02) mL portion of an HCl solution required 29.71(±0.02) mL of 0.01963(±0.0032) M Ba(OH)2 to reach an end point with bromocresol green indicator. ? of HCL = 29.71?? ? 0.01963 ???? ??(??)2 ?? ? 2 ???? ??? ???? ??(??)2/ 50.00?? = 0.02333 ? Calculate the uncertainty of the result (absolute error).Calculate the coefficient of variation for the result.
- Knowing that Ksp of PbI2 = 10-⁷ find out if precipitate will form when mixing 100ml of 0.01 N Pb solution (NO3) 2 and 200ml of 0.01 M NaI solutionMake an schematic diagram for procedure below: B. %SO3 determination Transfer all precipitate to the filter paper avoiding any loses during the filtration procedure. One may use a rubber policeman to scrape remaining precipitate in the beaker. Wash the precipitate in the beaker 3x with hot water before transferring to the filter paper. Test the washings with 0.1 M AgNO3 Cloudiness indicates presence of chloride ions; therefore there is a need to wash more the precipitate. Carefully lift the paper out of the funnel, fold it as demonstrated by your instructor and transfer it onto the constant weighed crucible. Dry the crucible cautiously with a small flam, as instructed by your instructor. The flame should be directed at the top of the container, and the lid should be off. Avoid spattering. After drying, char the filter paper by increasing the flame temperature. The crucible should have free access to air. The lid should be kept handy to smother the filter paper in case it catches fire.…I have gotten this wrong 8 times! Please help. See screenshot attached. Thank you! A solution containing a mixture of metal cations was treated as outlined. Dilute HClHCl was added and a precipitate formed. The precipitate was filtered off. H2SH2S was bubbled through the acidic solution. No precipitate formed. The pH was raised to about 9 and H2SH2S was again bubbled through the solution. A precipitate formed and was filtered off. Finally, sodium carbonate was added to the filtered solution and no precipitate formed. What can be said about the presence of each of these groups of cations in the original solution?
- A sodium thiosulfate solution is standardized using pure copper as the primary standard. A sample of copper weighing 0.2624 g is dissolved in acid, excess KI is added, and the liberated iodine was titrated with 42.18 mL of sodium thiosulfate solution. Calculate the molarity of sodium thiosulfate solution. 2Cu2+ + 4I- → 2CuI + I2 I2 + 2S2O32- → 2I- + S4O62-15. A 300.00 mL solution of 0.00165 M A2B5 is added to a 230.00 mL solution of 0.00380 M C2D3. What is pQsp for A2D5?Pre Lab Questions: (Each answer is to be written as a complete sentence) What is the reason for washing the precipitate with water in Step 9? Define precipitate. Define filtrate. In Step 2, what is the purpose for rinsing the stirring rod? read the Procedure to answer the questions Using a balance, mass between 1.50 – 2.00 grams of sodium carbonate in a pre-massed 150mL beaker. Add 20 mL of distilled water and stir thoroughly to make sure all the crystals are dissolved. Rinse the stirring rod with a little distilled water after stirring. Using a balance, mass between 1.50 – 2.00 grams of calcium chloride dihydrate in a pre-massed 50 mL beaker. Repeat Step 2 for the solution in the 50 mL beaker. Pour the calcium chloride solution into the 150mL beaker containing the sodium carbonate solution and stir. Mass a piece of filter paper. Fold the filter paper and place it into the funnel. Wet it with a little distilled water to ensure that it is stuck to the sides of the funnel. Slowly…

