Q: Write chemical equations to show what happens when each of the following substances is mixed with…
A: Hello. Since your question has multiple sub-parts, we will solve the first three sub-parts for you.…
Q: Intermolecular Forces (what is the dominant IMF?) carbon tetrachloride N,H, Nitrogen gas Octane…
A: Intermolecular forces are the electrostatic forces that exist between the neighbouring molecules.…
Q: Which pairs of compounds will form a solution? a. benzene (C 6H 6) and hexane (C 6H 14) b. Na 2SO 4…
A: A solution is formed if both the components that is solute and solvent are completely miscible with…
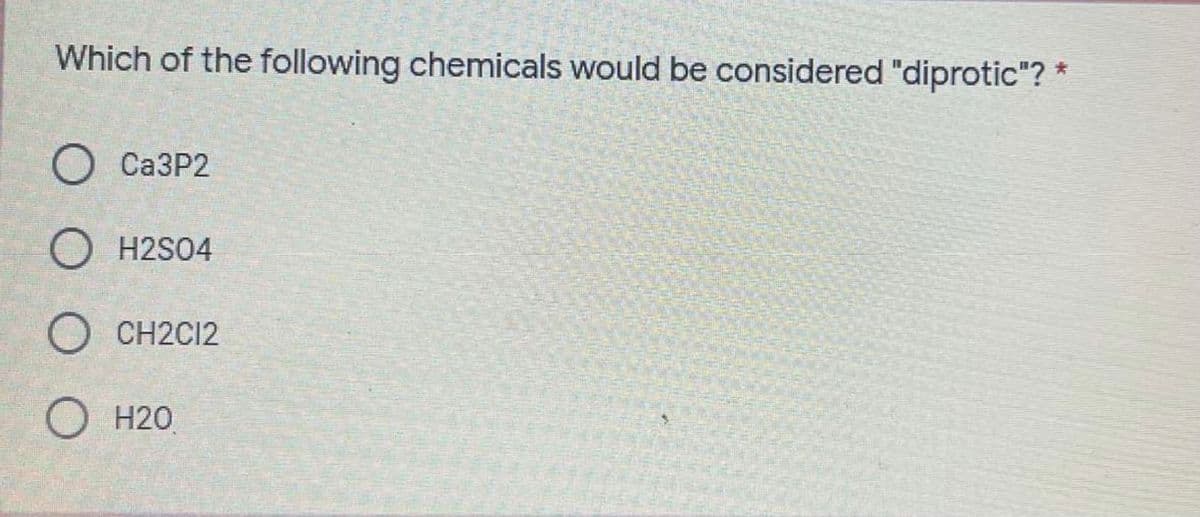
Q: Prepare 1 liter of 0.5M sulfuric acid from a concentrated aqueous solution. Sulfuric acid density is…
A: H2SO4 We have to determine the concentration of H2SO4 first and then dilute to prepare 1 L of 0.5 M…
Q: Which one of the following is characteristic of H2SO4? A. produces OH- in water B. has a…
A:
Q: How many grams of Mg(OH)2 will be needed to neutralize 25 mL of stomach acid, if stomach acid is…
A: 1- First calculate the moles of HCl : Moles of HCl = [ Molarity × Volume ] Moles of HCl = [ 0.10M…
Q: What is the volume of 0.05M HCl that is required to neutralize 50 ml of a 0.10M Mg(OH)2 solution? a-…
A: Answer:- This question is answered by using the simple concept of chemical reaction of acid and base…
Q: A solution has a concentration of 1.25g/l. what is its concentration in pp
A: For the expression of very dilute solutions, the unit term used is ppm which is parts per million.…
Q: What is the molarity of a 3.00% (m/v) solution of Levothyroxine?
A: Given that : We have to calculate the molarity of a 3.00% (m/v) solution of Levothyroxine. The molar…
Q: Draw the molecular structure of the polyvinyl alcohol molecules in the slime mixture? i.e., PVA +…
A: Given: Polyvinyl alchol molecule in slime mixture. To find: Molecular structure of Polyvinyl alchol…
Q: Which of the following compounds is insoluble in water? O K2CO3 O CaBr2 OAGNO3 BaSO4
A:
Q: Which of standard substance (or standardized solution) is correct for standardization of NaNO2:…
A:
Q: Of the following substances, which one is NOT expected to dissolve in water? Ca(OH)2 C2H5OH…
A: Solubility rule "Like dissolved like. Polar molecule dissolved in polar solvent and non-polar…
Q: Prepare 1.5L of a 2M sulfuric acid aqueous solution.
A: Amswe is given below
Q: What are ampholytes?
A: ampholytes are those molecules which can act like both acid as well as base i.e they show amphoteric…
Q: Which of the following compounds is insoluble in water? O Fe(CH3COO)2 O Fes O FENO3 O FeSO4
A: Water is a polar solvent. Polar and ionic compounds are soluble in water while nonpolar and non…
Q: Which of the attached molecules can hydrogen bond to anothermolecule like itself?
A: Given:- Which of the attached molecules can hydrogen bond to another molecule like…
Q: If the pH of a microorganism is 9.1, what is the [H+] concentration
A: By looking ph value we can tell us which compound is acid or basic or neutral. PH =7 -Neutral ph…
Q: Of the following substances, which one is NOT expected to dissolve in water? HNO3 C2H5OH CH3OH…
A: Solubility of any substance means the mixing of one component into the other component. If the…
Q: Which of the following are first aid measures for glacial acetic acid? Select one: a. If swallowed,…
A:
Q: Which one of these parameters measure amount of organic matters in water? Select one: O A. nutrients…
A: (a). Given statement is : Which one of these parameter measure amount of organic matter in water ?…
Q: 12. Write the balanced chemical reaction showing the neutralization of aqueous acetic acid with…
A:
Q: How many milliliters of 0.294 M KOH solution are required to completely neutralize 171.8 milliliters…
A: Given: Concentration(molarity) of KOH = 0.294 M Concentration of H3PO4 = 0.110 M Volume of H3PO4 =…
Q: Which of the following compounds is insoluble in water? Li2CO3 (NH4)2CO3 Na2CO3 K2CO3 FeCO3
A:
Q: Is NaH2PO4 a strong electrolyte?
A: NaH2PO4 is a salt of H3PO4.
Q: 2. A substance is A haa high meling poit R contains ocovalent bond C saqueo solution condacts an…
A: Electrolyte : It is substance when dissolved in polar solvent produces free ions and is able to…
Q: * 60% iPad 2:23 PM + openvellum.ecollege.com Search Textbook Solutions | Chegg.com 61%iPad2:20…
A: The conditions under which the process of crenation takes place are to be described.
Q: the questions by writing the correct letter of your answer. Explain your answer briefly in 1 or 2…
A: Given-> (d)-> bleach ,pH 12.5 - 13.5 (e) ->distilled water ,pH =7
Q: The concentration of 0.00400 M Al(OH)3 solution in terms of ppm is: Select one: 5.20 ppm 0.308 ppm…
A: Given concentration of Al(OH)3 = 0.00400 M we know that molar mass of Al(OH)3 = 78 g/mole ppm is…
Q: Explain how you could make a 0.01 M C6H12O6 solution from a 0.02 M C6H12O6 solution.
A: In the dilution process, the relationship between initial and final concentrations and volumes of…
Q: Ways or means that will help reduce or lessen the effects of these chemical reactions to avert…
A: One of the main reason of fish killing is eutrophication that is increased algal growth that cause…
Q: How many of the following compounds are insoluble in water? NaC2H3O2 SrSO4 BaS AlPO4
A: At a certain temperature, the dissolving property of any compound in a liquid in order to make…
Q: Which one of the following would be immiscible with water? H2C-CH2 O O H2C-CH O C2H5 OH H3C-S-CH3 Os…
A: Miscibility : Miscibility is the property of two substances to mix in all proportions ( to fully…
Q: Which of the following pairs of compounds form a solution? a. KCl and CCl 4b. 1-propanol (C 3H 8O)…
A: Solution is the homogeneous mixture of small particles. Liquid solution is transparent in nature.…
Q: 2. Which of the following molecules is the most vwater soluble? HO HO H. HO dopamine estradiol H. HO…
A: Solubility of compounds is based on the principle of like dissolves like. This means that a polar…
Q: Which of these interactions that hold hair strands together is/are pH sensitive? I. À salt bridge A…
A:
Q: The concentration of CaF2 in an aqueous solution is measured to be 0.056 M. Calculate the…
A:
Q: identify the products of the following reactions, a,b and c
A: A chemical reaction is symbolic representation of the conversion of substances to new substances. In…
Q: NH4NO3
A: Ca3(PO4)2 is calcium phosphate and is a white colored solid. It is present in our bones and can be…
Q: Which one of the following is insoluble in water? Na2Co3 Ca(NO3)2 Cu3(PO4)2 K2SO4 NH4Cl
A: Solubility rules: Salts containing Group I elements and ammonium ion are soluble. Generally…
Q: 13 which Of these aqueous SOIutions conduct eiectricity well? O acetone, CH3 COCH3 D AGNO3 caci2…
A: Which of the aqueous solution conduct electricity well and why they conduct electricity explanation…
Q: Which of the following compounds is insoluble in water? (NH4)2S CaCO3 NaOH Ag2SO4 Pb(CH3COO)2
A: Observing all five given compounds show that except Calcium carbonate all other are soluble in…
Q: How many of the following compounds are insoluble in water? KC2H3O2 CaSO4 SrS ΑΙΡΟ4 1 2 3 4
A: The solubility represents the dissolution of solute in a particular solvent in a specific…
Q: List three types of bonds found In a sodium chloride aqueous solution
A: A sodium chloride aqeous solution has Na+ , Cl- ions and water molecules
Q: An iron - salicylate solution has a concentration of 5.92 x 10-4 M. It was prepared by taking 2.00…
A:
Q: A student tests the electrical conductivity of a 2.0 M oxalic acid solution and compares it to the…
A: The acids given are oxalic acid i.e COOH-COOH and hydroiodic acid i.e HI. And the conductivity of HI…
Q: Draw and label the parts of a Clavenger Apparatus.
A: Clavenger apparatus is one of the setup to extract the volatile oils.
Q: Select ALL of the following compounds which can be dissolved in water to prepare a 0.2 M aqueous…
A: Concept: A solution in which water acts as a solvent is called as an aqueous solution. Here in the…
Q: Which of the following pairs would you predict to be insoluble/immiscible? A. Hexane (CeH14) &…
A: Polar solute are soluble in polar solvents while non-polar solute are soluble in non-polar solvents.
Step by step
Solved in 3 steps

- 4.Based on the boiling point and surface tension values obtained, which of the two liquids has stronger IMFA?Draw and label the parts of a Clavenger Apparatus.Give the functions of the following ingredients, then name a branded/commercial skin or hair care product where the said material is used. Ozokerite Wax Sodium Ascorbate Lactic Acid 90% Titanium Dioxide Magnesium Aluminum Silicate Mineral Oil 50 SUS Tween 80 Zinc Oxide Retinol DMDM HYDANTOIN
- At the largest solar thermal power plant in the world, what chemical is used as the working fluid? sodium acetate water proteins sodium chloride18,Which of the following compounds is insoluble in water? Group of answer choices A, (NH4)2CO3 B, FeCO3 C, K2CO3 D, Li2CO3 E, Na2CO3Which of the following salts are you going to use for the salting out or purification of the produced hard fatty acid salt?* A. Magnesium chloride, MgCl2 B. Potassium chloride, KCl C. Sodium chloride, NaCl D. Barium chloride, BaCl2 E. None of the given options
- The volume of an E. coli cell is approximately 1 µm^3. Calculate the number of water molecule that could fit inside this volume. Assume pho(H2O)= 1.00 g cm^-3What is the purpose of formaldehyde in embalming fluid? a. it acts as a surfactant b. it acts as a preservative c. it acts as a vehicle d. it acts as a bufferExplain in a more clinical detail the mode of actions of the following on the human skin cell or tissue: 1. Retinoids2. AHA/BHA

