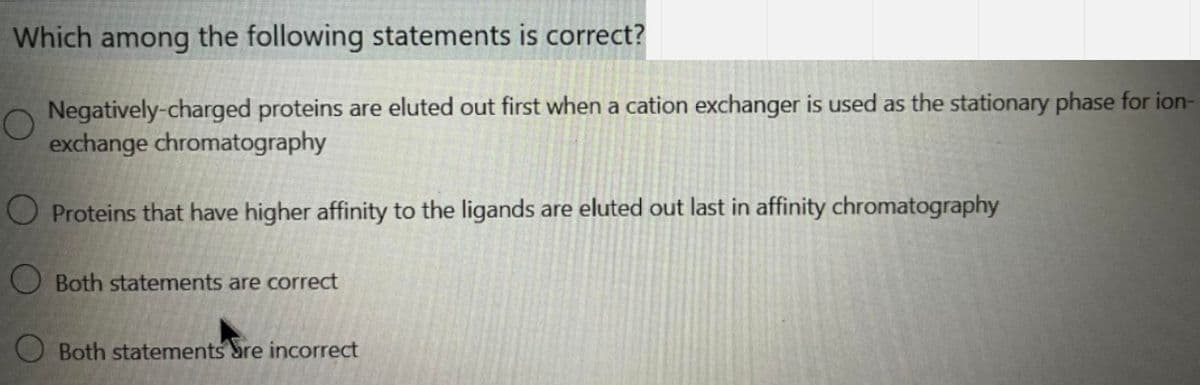
Which among the following statements is correct? Negatively-charged proteins are eluted out first when a cation exchanger is used as the stationary phase for exchange chromatography O Proteins that have higher affinity to the ligands are eluted out last in affinity chromatography OBoth statements are correct O Both statements are incorrect
Q: For you, what is the contribution around 18-1900 of golden era in biochemistry? Explain
A: Introduction: The term biochemistry refers to the study of the chemistry of living things including…
Q: Describe surroundings at home which reminds you about biochemistry and relate the situation to…
A: Food is the source of different biomolecules, vitamins, and minerals for the body. Some of the food…
Q: 10. Messenger RNA often encodes more that one biological activity. These activities can then be…
A: Introduction: Messenger RNA (mRNA) is a single-stranded RNA that carries hereditary information from…
Q: Sample Results Remarks Glycine A Egg white B Casein Tyrosine D
A: In a polypeptide chain the amino acids are linked together via peptide linkages.
Q: Indicate at what step number in the glycolysis pathway the following event occur and why? a. First…
A: Isomerisation is defined as the reaction which alters a compound into its simple isomers which…
Q: What does protein function in the human body?
A: Proteins are made up of amino acids or we can say that amino acids are the building block of…
Q: Step by Step process of Embden-Meyerhof pathway of RBC metabolism Pls Explain as simple as…
A:
Q: Why is it difficult to accurately estimate Km and Vmax values from a Michaelis-Menten plot?…
A: Introduction: The study of reaction rates and how they change in response to changes in the…
Q: The process of protein decomposition by pepsin enzymes in stomach is assumed and modeled as a batch…
A: The reaction kinetics given in the pepsin digestion reaction follows Michaelis-Menten Kinetics. The…
Q: What type of tertiary interactions do we observe for the two amino acid residues in the box? Draw…
A: It is the interaction between the R groups in the protein that determines tertiary structure. These…
Q: Describe surroundings at home which reminds you about biochemistry and relate the situation to…
A: related to this : proteins A situation to describe : Cooking
Q: Which of the following is the process where there is transfer of phosphate group to produce ATP or…
A: ATP and GTP are nucleoside triphosphates that are important for cellular function. They are produced…
Q: The "D" in DNA stands for which of the following?
A: DNA : Chemical name for molecule which carries genetic instructions in the living organisms.
Q: You are working in a factory that is producing chemicals from the bacterium Corynebacterium…
A: Corynebacterium glutamicum is a facultative anaerobic gram-positive, catalase-positive, rod-shaped…
Q: Reactions and Thermodynamics of Glycolysis
A: Third step of glycolysis, fructose-6-phosphate is converted to fructose- 1,6-bisphosphate by…
Q: Explain the mechanism of Warburg effect and how it benefits cancer cells
A: Cancer means uncontrolled cell growth. This uncontrolled cell growth may cause a lump of cell or…
Q: 5. Protein tyrosine phosphatase-1B (PTP1B) is an important enzyme regulating insulin signaling be-…
A: PTP1B enzyme in question catalyze the hydrolysis of phosphorylated tyrosine on Insulin receptor and…
Q: 4. Synthesis of basic corticosteroids. Steroid: A. Testosterone. B. Aldosterone. C. Pregnenolone. D.…
A: Introduction: Corticosteroids are steroid hormones that are synthesized in the adrenal cortex. It is…
Q: How to calculate the amount of myoglobin in grams from a 2.0 ml sample of protein extract???
A: Beer Lamberts law states that value of Absorption at a particular wavelength of light by an analyte…
Q: 1. Proteins act as structural components such as keratin of hair and nail, the collagen of the bone…
A: "Since you have posted a question with sub-parts we will answer the first 3 question for you. If you…
Q: Describe the roles of calcium in the cell, and the mechanisms that the cell uses to control…
A: Calcium is a vital nutrient that aids in the mineralization of the skeleton. Over 99 percent of the…
Q: How is a “committed step” defined in the context of a metabolic pathway and why are they important?…
A: A metabolic pathway is a chain of biochemical reactions linked together by their intermediates.…
Q: Question 5 In cholesterol, to which ring of the steroid system is the hydroxyl group attached? B A.
A: Cell membrane is made up of lipid bilayer. The composition of different types of lipids has very…
Q: In olfactory neurons, it is estimated that activation of the olfactory receptors results in an…
A: The olfactory epithelium of the nasal roof contains olfactory receptors. Each olfactory receptor is…
Q: Match the following descriptions to the given choices. A. Aldosterone The first molecule in the…
A: Steroids are biologically four ringed organic compounds that act as important components of cell…
Q: Which of the following statements is correct about oxidative pentose phosphate pathway? Group of…
A: Oxidative pentose phosphate pathway: . This is the first phase in pentose phosphate pathway in which…
Q: Retroviruses, like the HIV, contain an enzyme called reverse transcriptase. Explain the flow of…
A: Retroviruses are viruses with an enzyme known as reverse transcriptase. Transcription is the process…
Q: Review method used to increase the solubility of a drug under the following headings co solvents PH…
A: Bioavailability is a powerful determinant of drug absorption. It represents the administered dose…
Q: Match the following descriptions to the given choices. A. Aldosterone The first molecule in the…
A: 1. The first molecule in the biosynthesis of steroids that contain the…
Q: II. GENERAL INSTRUCTIONS Answer the following items. 1. Give an example of a catalyst and its…
A: Tri carboxylic acid cycle or the citric acid cycle, TCA cycle or the Krebs cycle is a series of…
Q: 3. (а) 0.0050 M operate at one-quarter of its maximum rate? At what substrate concentration would an…
A: To study an enzyme kinetics, Michaelis-Menten kinetics equation is best known mathematical…
Q: Question 3 Aldosterone regulates sodium, potassium, and chloride ions in tissues True False
A: Aldosterone is a hormone which is secreted by the adrenal glands. Aldosterone secretion is increase…
Q: Morphine (give structure) and enkephelin (give structure) both are antagonists of the m opioid…
A: Morphine is pain medication which is used in Treatment of pain.this is belongs to opiate family.…
Q: What type of monomers make up proteins? What are protein polymers called?
A: Note - Hi ! Thank you for the question. We are authorized to answer one question at a time. Since…
Q: why all amino acids except glycine have L and D forms and specify the type of isomerization…
A: Amino acids are the monomeric units of proteins. The general structure of an amino acid has a…
Q: What are the major functions of lipids?
A: Lipids are essential fats that play significant roles in the human body. It's a prevalent…
Q: Draw the two amino acids serine and alanine, and a dipeptide that could be formed by combining these…
A: Amino acids are the building blocks of proteins which are composed of amino group (NH3+), carboxyl…
Q: Food Sample marshmallows Pumpkin seeds cracker Dried cranberries рорсorn Rice cake almond
A: Calorific value of foods is based on their nutrient content. Calorific value of foods is the total…
Q: 5- Draw structure of products in the following metabolic reactions and name the enzyr involved and…
A: Introduction: The drug metabolism is needed to convert non-polar lipophilic compounds into polar…
Q: Classify the following amino acids base on their side chain if it's either -Non-polar aliphatic R…
A: Amino acids are the molecules that contain an amino group and a carboxylic group linked through the…
Q: 44. Roy's doctor recommended that he watch his blood cholesterol level because his of reading of…
A: In a person, the total level of cholesterol should be always less than 200 mg/dL for a healthier…
Q: (A) Generally, outline how you could produce or isolate such a species. (B) Once you have obtained a…
A: In the question, reaction presented is hydrolysis of a peptide bond of a dipeptide. The dipeptide…
Q: The activity of isocitrate dehydrogenase in E. coli is regulated by the covalent attachment of a…
A: Isocitrate dehydrogenase is an important Enzyme of TCA cycle for the conversion of isocitrate into…
Q: A solution has a pH of 5.4. What is its pOH O5.4 8.6
A: The hydrogen ion concentration [H+] and hydroxide concentration [OH–] in a aqueous solution have an…
Q: An unknown solution contains a single pure sugar. It gives positive results to Molisch’s and…
A: The qualitative tests are used to identify the presence of certain sugar in the given sample.…
Q: Indicate what step each of the events in the glycolysis pathway the following takes place: a. First…
A: The first step in breaking down glucose to obtain energy for cellular metabolism is glycolysis.…
Q: True or False? It takes Carbon dioxide and water in the presence of sunlight to produce…
A: Carbohydrates are formed in green plants by the process of photosynthesis.
Q: Direct methanol fuel cells (DMFCS) have shown some promise as a viable option for providing "green"…
A: The value of Delta G indicates whether a reaction has a tendency to go forward. From this value, we…
Q: [S] Vo vo with I 0.00 0.00 0.00 1. 15 2. 14 0. 60 0. 25 0. 50 1. 15 2. 14 3. 75 6. 00 8. 57 1.00…
A: The plot is given in step 2
Q: While fatty acids are most often formed by the condensation of_-carbon units, isoprenoids are…
A: Fatty acid and isoprenoid both are class of lipid and plays an important role in the metabolism of…
Step by step
Solved in 2 steps

- Which among the following statements is correct? Group of answer choices Both statements are incorrect Ion-exchange chromatography is dependent on the charge and size of the target component Both statements are correct Affinity chromatography is dependent on the affinity of the target component to the mobile phaseAn IV bag of NS containing 30 g KCl is being administered at a rate of 80 mL/hr using a macrodrip tubing with a DF of 15 gtts/mL. How many drops per minute is that? Group of answer choices a. 30 gtts/min b. 22 gtts/min c. 10 gtts/min d. 20 gtts/minIf Ca(NO3)2 solution is added to the saturated CaSO4 solution;Solubility product of CaSO4 (KÇ)II. Solubility of CaSO4III. The concentration of Ca + 2 ionWhich of these or which decreases at the same temperature? A) I only B) II only C) III only D) I and II E) II and III
- What is ion-exchange chromatography based on? moving a molecule with a net charge in an electric field high affinity of the antibody for the proteins separation of proteins by size affinity of proteins for specific chemical groups or specific molecules separation of proteins by their net chargeDefine the following terms:a. ligandb. motor proteinc. GTPd. macromolecular crowdinge. excluded volumeGiven these three statement- two of which are truths and one is a lie. Which of the following statements is a lie? Select one: a. Unless a channel of a carrier protein is open, cations remain outside. b. When we experimentally induce a positive charge in the inside of the cell, cations will be forced out. c. Equal concentrations are achieved in the ions involved in the electrochemical gradient. Given these three statements - two of which are truths and one is a lie. Which of the following statements is a lie? Select one: a.The primary active transport prepares the conditions of the membrane gradient by making the extracellular membrane have a less negative charge in the concentration gradient than the intracellular membrane. b. The primary active transport uses ATP while secondary transport helps replenish ATP. c. Both primary and secondary transport moves solutes against their concentration gradients.
- Define the following terms:a. supersecondary structureb. protomerc. phosphoproteind. denaturatione. ion exchange chromatographyDefine the following terms:a. chemiosmotic coupling theoryb. uncouplerc. ionophored. submitochondrial particlee. a,b hexamerSelect all the true statements about ion exchange chromatography Group of answer choices In anion exchange, negatively charged species are retained on the column, whereas positively charged ones go through the column unretained In cation exchange, negatively charged species are retained on the column, whereas positively charged ones go through the column unretained In anion exchange, a protein with a pI of 9.0 will not get retained if the mobile phase has a neutral pH In cation exchange, a protein with a pI of 9.0 will get retained if the pH of the mobile phase is acidic
- Membrane carriers resemble enzymes except for the fact that carriersa. are not proteins.b. do not have binding sites.c. are not selective for particular ligands.d. change conformation when they bind a ligand.e. do not chemically change their ligands.We have isolated a mixture of 2 protein of the same molecular weight but different isoelectric points. The isoelectric points are 7.5 for Protein A and 9.0 for Protein B. What would be the order of elution from a cation exchange column if a buffer of pH 7 is used? Group of answer choices Both proteins will remain in the column. Both proteins will elute simultaneously Protein B followed by Protein A Protein A followed by Protein BDesign an experiment to purify protein X on an anion-exchange column. Protein X has an isoelectric point of 7.0.

