Q: 4. Urea, CO(NH₂)2, is manufactured on a large scale for use in producing urea-formaldehyde plastics…
A:
Q: ноч HO to HO
A: Dehydration of alcohol in the presence of strong acid ( like H2SO4) to give alkene. Alkene undergo…
Q: The temperature of a 500.mL sample of gas increases from 150. K to 350. K. What is the final volume…
A:
Q: Compound A (CH₁0O2) works well in color reaction with FeCl3 solution. Compound A also can reaction…
A:
Q: The atmospheric pressure in Denver, CO is 633 mmHg. What is this pressure in atm?
A: Given, The atmospheric pressure in Denver, CO= 633 mmHg. Atmospheric pressure of CO = ? atm
Q: 37. Which carbocation is likely to undergo a 1,2-hydride shift? CH3 CCH₂ (A) CHCH3 (B) (C) CH3 CH₂…
A:
Q: What is the AG for the following reaction from the equilibrium constant at the temperature given?…
A: R = 8.314 J/K.mol , T = 900°C = 1173 K
Q: Table 2 Weighing by Difference Mass of Empty Beaker Mass of Beaker + Sugar Mass Measurement 69.787 g…
A: Given, Mass of empty beaker = 69.787 g Mass of beaker + Sugar = 75.230 g Calculate the mass of…
Q: Ozone (03) in the atmosphere can react with nitric oxide (NO): 03 (g) + NO(g) -> NO₂(g) + O₂(g).…
A: Given: standard change in enthalpy of formation of reactants and products.
Q: Draw the product of the E2 reaction shown below. Ignore any inorganic byproducts. Br KOtBu Draw the…
A: E2 reaction is a type of biomolecular elimination reaction. In this type of reaction both carbon…
Q: Draw the major product of this reaction. Ignore inorganic byproducts.
A: Given : structure of reactant. Step 1 : formation of carbocation Step 2 : attack of Nucleophile…
Q: 48. What product is obtained from hydroboration-oxidation of 1-heptyne? OH (A) (B) 49. What is the…
A: The first reaction is the hydroboration oxidation reaction of the alkyne . In this hydroboration…
Q: For the reaction PCl₅(g) ⇌ PCl₃(g) + Cl₂(g) Kp = 1.45 × 10⁻⁴ at 160 °C. A 1.00 L vessel at 160 °C is…
A:
Q: Synthesize the following compounds from any of the given starting material, any necessary inorganic…
A: The formation of the final product needs a lot of steps that can be divided into 4 major steps.…
Q: Core Chemistry Skill: Using Prefixes ▼ Part A What are the abbreviations for a milliliter, a…
A: Millimeter, megameter and centigram are all units of physical properties such as length and mass.
Q: A solution contains 8.61x10-3 M calcium nitrate and 8.96x10-3 M chromium(III) acetate. Solid…
A: From the possible precipitation reactions, the concentration of Cr3+ and concentration of Ag+ can be…
Q: 2. Draw the Lewis dot structure for each of the following molecules or ions. Determine the number of…
A:
Q: 05 Burning of natural gas (x) in excess of cer (0₂) proceeds with release also two gares y and I are…
A:
Q: A pharmacist reading doses needs to convert the requested dose to the units reported for each…
A:
Q: A 0.150 M solution of a weak acid has a pH of 2.75. What is the Ka value of this acid?
A: Given that, The concentration of a weak acid is C = 0.150 M. The pH of that 0.150 M solution of warm…
Q: What is the heat, q, when 25.0g of acetonitrile (CH3CN) reacts completely with excess water? Molar…
A:
Q: Use the following Lewis diagram for ethyl methyl ether to answer the questions: H +HI 1.. 2 Н H H- H…
A:
Q: (3) Vapor pressure of liquid ethanol is 7291 Pa at 20°C. (a) one student reports that the vapor…
A: #3: Given the vapor pressure of ethanol at 20 oC is 7291 Pa Atmospheric pressure, Patm = 101325 Pa
Q: Mass of Sample Trial 1 Trial 2 Trial 3 Weighing bottle Initial mass 163.757g 129.203g 167.423g…
A:
Q: Consider the structure of one enantiomer of warfarin (top right), an essential drug that exhibits…
A: Warfarin is an essential drug that exhibits anticoagulant activity by inhibiting vitamin-k epoxide…
Q: (5) Which compound is most acidic? ( ) A. H₂CC CH B. CH₂COOH C. C6H5OH D. C₂H5OH (6) Which compound…
A: 5) (B) Acetic acid is most acidic because conjugate base is stabilized by resonance
Q: 2. State CLEARLY how you would differentiate between primary, secondary, and tertiary alcohols shown…
A:
Q: Select the reagent that you would use to convert an alkene to a Markovnikov (more substituted)…
A:
Q: Propose an efficient synthesis for the following transformation. ㅅ HBr, ROOR The transformation…
A: First we are going to make anti markonikov product then to increase the number of carbon we made…
Q: 14. How do you know you have the right compound? Comment on the test you have done or would have…
A:
Q: 3. Please draw out the major product or reaction conditions for the following reactions. (1) OH (2)…
A: As per Guidelines , Since you have posted multiple question as per guidelines we can solve only 3…
Q: 2. In the molecule shown, what are the expected geometries around the indicated carbon atom and…
A: Note- The given question is a multipart question, hence I solved first three questions according to…
Q: 57. Which structure is expected to emerge as the product of the reaction between the given alkyl…
A: For an E2 elimination the stereo electronic requirement is the leaving group and the -H atom must be…
Q: 1. A. Pick one of the molecules below that you would expect to undergo Sn1 substitution the fastest.…
A:
Q: Could you explain questions 1 annd 7? Thank you
A: Hybridization is the mixing of atomic orbitals to give rise to new set of orbitals with similar…
Q: p.101edu.co Question 49 of 50 Two chair conformers of a structure are shown below. Identify the more…
A:
Q: Carvone is the major constituent of spearmint oil. What product would you expect from reaction of…
A:
Q: Which of the following set of quantum numbers can match an electron in the 3d orbital?
A: We have to tell the correct sets of quantum number for an electron in the 3d orbital. So, for an…
Q: Draw the structures of the two chair conformations of trans-1-tert-butyl-4-ethylcyclohexane. Which…
A: The solution is given below -
Q: Predict the product(s) of the following reaction: OCH₂CH3 1. CH3CH₂MgBr 2. H30*
A: When ethylcyclohexanoate treated with Grignard reagent (ethyl magnesium bromide) and simultaneously…
Q: What is the symbol (including the atomic number, mass number, and element symbol) for the oxygen…
A:
Q: reaction to complete the following questions #7 and #8: CaCl2 (s) + H2O (l) → CaO (s) + 2 HCl (aq)…
A:
Q: Determine the Ksp of an acidic salt through titration. Using a .10M solution of NaOH titrate a…
A: Solubility product of KHT can be determined from its solubility, deduced from the titration with…
Q: Draw the major product of this reaction. Ignore inorganic byproducts. Na2Cr2O7 OH H2SO4, H2O
A:
Q: (a) rate at which the following alcohols undergo dehydration to form alkenes in sulfuric acid OH OH…
A: Given : structure of molecules Rate is directly proportional to the stability of formed carbocation.…
Q: In the production of a bottled alcoholic mudslide, 3 ingredients are mixed together and bottled:…
A:
Q: compare and constract the nucleophilic of HCL with propanol, pronanone, and propan-1-ol
A: We are given some compounds and we have to compare and contrast the nucleophilic character of these…
Q: A box contains a gas composed of 12 atoms each corresponding to the different stable isotopes of Tin…
A:
Q: Identify the numbers in each of the following statements as measured or exact. Drag the appropriate…
A: Measured number are those which have some uncertainty present in them but exact number have no…
Q: Which of the following substances do not follow the octet rule in their Lewis structures? W. SF4 Z.…
A:
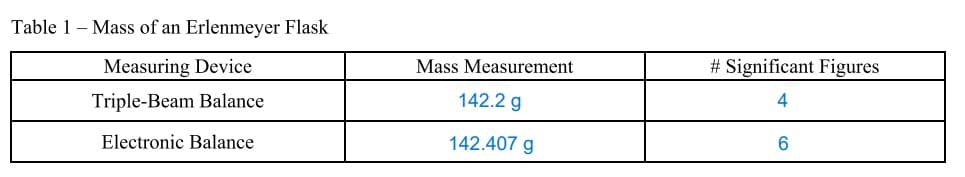
Which balance, triple-beam or electronic, provides the more precise measurement? Explain.
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

- Show the gravimetric calculation of the ff:No need guideline answer if u provide guideline answer I will dislike ok Module Code & Title: CHM9011M Chromatography and Sep Sciences Description of Assessment Task and Purpose: To prepare an SOP for the GC-MS InstrumentI have struggled to get the answer. A classmate said it was 159, though I would like to see the steps in demensional analysis, with correct sig figs. thank you! Provide only typed solution answer no handwritten solution needed
- Table 1. Data for the Concentration of the KMnO4 Unknown Solution. Abs of Unknown Solution is 0.362 Absorbance Concentration Trial 1 0.120 0.00145 Trial 2 0.066 0.00104 Unknown Solution: __________ What is the Concentration of the Unknown Solution. _____________ Show statistical treatment of data.Help with q=mcchange in temp ?Good day I have asked this question already this question is from the lesson about refractometry and I belive that its about linear regression which I don't know how to get the missing value. 28-31. A calibration curve was created, and the results has beenestablished and showed in the table, Assume the following data, Supply the missing value. n 1.55 1.65 1.70 1.80 ??? 1.62 Conc. 10% 15% 25% 35% 22% ??? 28-29.a. 1.66 c. 1.70b. 1.68 d. 1.72 30-31.a. 14.24 c. 16.24b. 15.24 d.17.24
- The original solution was diluted by a factor of three, and then the absorbance of the diluted cereal sample solution was measured and recorded as (2.0x10^-1). Use the following trendline and dilution factor to determine the concentration of iron, in unit of M, in the original cereal sample solution. The trendline (linear regression) as displayed on the plot is y = 11898x - 0.0112.chemistry help need please make sure of sig-fig1. Which of the following types of filter are made to operate continuously? a. rotary drum b. gravity c. filter press d. leaf filter 2. The filter medium resistance is controlled by I. pressure drop II. flowrate a. only a is correct b. only b is correct c. none are correct 3. In filtration, which of the following conditions will yield to a higher resistance? a. none of these b. fluid with low viscosity c. slurry with high solid concentration d. large are of filter e. low cake compressibility
- Hello, can someone please help me? I have already hand graphed 1 and 2 and found the v max, however I cannot find the Km value. I am confused because the plots on the linear weaver burk plot do not extend past the values that make up the line. So how am I supposed to estimate the value for the y-intercept in order to find the Km valueWhich statements are TRUE regarding internal standards? I. The response factor is calculated by using a mixture containing known amounts of analyte and internal standard and by using detector response to the analyte and the internal standard. II. Internal standards are useful where instrument response or quantity of sample analyzed varies over time. III. Detector response is inversely proportional to concentration. IV. Detector response to analyte and internal standard is assumed to be constant over a range of concentrations A. I, II, and III B. II, III, and IV C. I and II D. I, II, and IVspectrofluorometry is potentially more sensitive and selective compared tospectrophotometry

