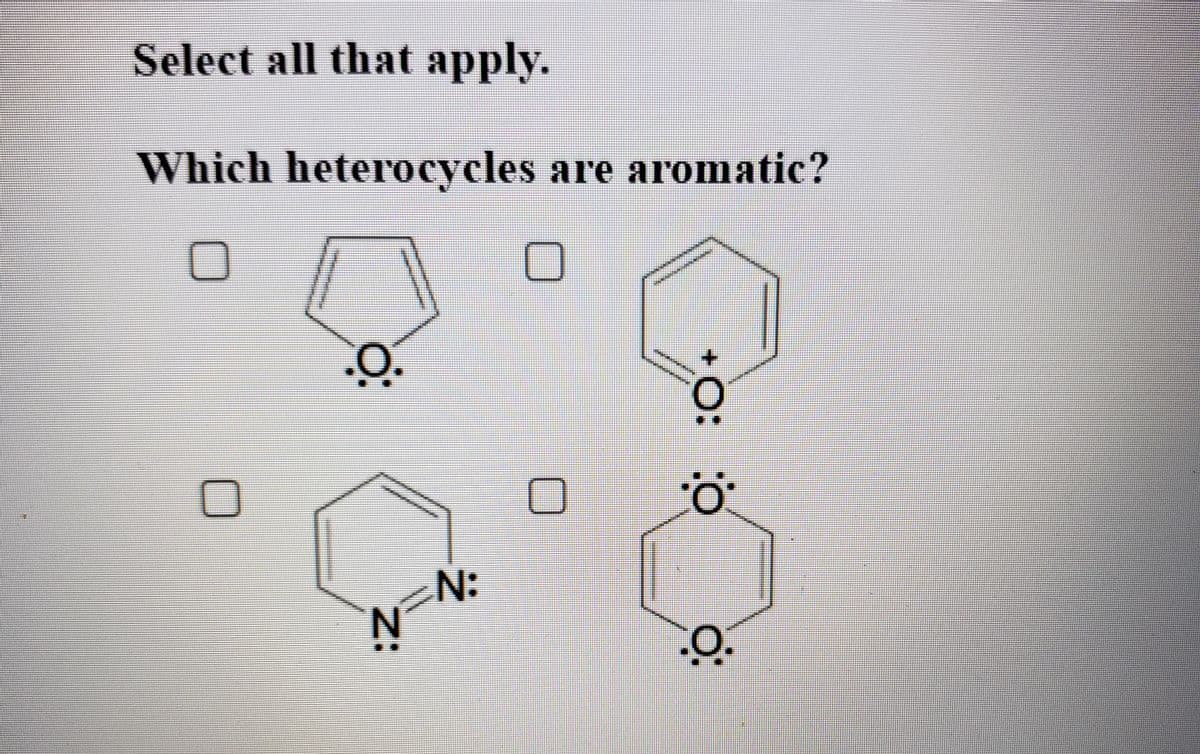
Q: Which heterocycles are aromatic?
A: Since we answer only upto 3 sub-parts, so we'll answer first three sub-parts. Please resubmit the…
Q: o electrophilic aromatic substitution
A: Electrophilic substitution reaction may be defined as the reaction in which electrophile is…
Q: Cyclooctatetraene is a not planar because it disrupts conjugation and avoids anti-aromaticity. b…
A: Choose the correct option for Cyclooctatetraene ---
Q: Label each heterocycle as aromatic, antiaromatic, or not aromatic.
A: The aromaticity of a molecule is governed by the following factors: Cyclic Planar Delocalization of…
Q: Which of the following compounds represent an acyclic compound? Select one: Oa. Ob.
A: Acyclic compounds are those compounds which don't contain cyclic ring. These are open chain. Cyclic…
Q: Which of the following anions is aromatic? A) B) C) D) CrO
A:
Q: Which (if any) lone pairs are participating in aromaticity?
A: Since the lone pair which is present in the single bonded atom is in conjugation with the ring,…
Q: Which is least reactive in electrophilic aromatic substitution? (A) (B) CH3 (C) CHO (D) C3
A:
Q: Rank the following compounds in order of increasing stability
A: According to Huckel's rule, a conjugated compound is said to be aromatic if it has (4n + 2)π…
Q: Rank the following compounds in order of increasing stability.
A: The stability of the given systems can be solved by the conjugation concept. Conjugation means to…
Q: (6) Which compound has aromaticity? ( А. В. С. D.
A:
Q: Which of the following is expected to show aromaticity? CH CH HC CH NH O none of the above is…
A:
Q: Rank the following substituted anilines from most basic to least basic:
A: Electron withdrawing group present in the phenyl ring increases the acidic strength. This results…
Q: Rank the following substituted anilines from most basic to least basic:
A: Amine is a functional group in which nitrogen atom is attached with two hydrogen and one aromatic…
Q: Label the alkene in each attached drug as E or Z. Enclomiphene is one component of the fertility…
A: If the relatively higher priority groups are on the same side then it is a (Z)-alkene. If they are…
Q: which of the foolowing Compounds whose structure/s involve a benzene ring and two substituents…
A: Draw the structure of all the compounds given, to determine which compound contains a benzene ring…
Q: Rank the following compounds in order of increasing stability. IV I II II
A: "Since you have asked multiple questions, we will solve the first question for you ( as per our…
Q: Which of the following best describes the lone pairs on oxygen in the following aromatic molecule.…
A:
Q: Which heterocycles are aromatic?
A: a. Fuarn is cyclic and planar. It has (4n+2)pi electrons which means it has 6pi electrons. Thus it…
Q: :S: (b) (c) (a)
A: Any molecule is said to be Aromatic if it follow the following the molecule must follow Huckel's…
Q: State which of the following compounds is/are aromatic and non-aromatic? Classify the compounds…
A: For a compound to be aromatic or anti-aromatic, the compound must follow the three conditions which…
Q: What is the main difference between an aromatic and an anti-aromatic compound? O None of these O…
A: Aromaticity
Q: Electrophilic aromatic substitution involves a cationic intermediate in which positive charge is…
A: Mechanism is given below
Q: w compound Y?
A:
Q: Arrange the compounds in set in order of decreasing reactivity (fastest to slowest) toward…
A: Electrophilic substitution is the substitution of H of an aromatic ring by an electrophile. The…
Q: B. C. D. A.
A: Some of the properties of aromatic compounds are: The ring must be conjugated It should be a closed…
Q: Which of the following is anti aromatic?
A: The anti aromatic compound has to be identified.
Q: Given that syn addition of H2 occurs from both sides of a trigonal planar double bond, draw all…
A: The objective of the question is to find all the stereoisomers obtained after the syn addition of H2…
Q: What compound undergoes metathesis to form each of the following compounds?
A:
Q: Rank group of compounds from most reactive to least reactive toward electrophilic aromatic…
A: An aromatic compound is more reactive toward electrophilic substitution reaction when it contains an…
Q: 1.2 Use the criteria for aromaticity to determine if the following compounds are aromatic,…
A:
Q: Classify each substituent as electron donating or electron withdrawing.
A: Electron donating group: The group that increases the electron density on the ring. Electron density…
Q: Explain why electrophilic aromatic substitution in Pyrrole takes place at C-2 positions whereas, in…
A: Pyrrole is more reactive towards electrophilic reagents in comparison to pyridine because of its…
Q: Is the following carbocation aromatic ? +) Select one: VES
A:
Q: Which of the following ions is aromatic? 0000 B
A:
Q: Circle aromatic compound(s)
A: Consider the following properties for a compound to be aromatic; The compound to be cyclic and…
Q: Arrange the compounds in set in order of decreasing reactivity (fastest to slowest) toward…
A: If the compounds having similar structure, then the general trends of compounds reactivity in…
Q: Label each compound as aromatic, antiaromatic or not aromatic. Assume allcompletely conjugated rings…
A: Aromatic compounds are those that obey the huckle rule of aromaticity (4n+2 pie electrons). The…
Q: Label each compound as aromatic, antiaromatic, or not aromatic. Assume allcompletely conjugated…
A: The first compound A is non-aromatic because the compound is not planar. It consists of one sp3…
Q: Which statement is incorrect as far as pyridine is concerned? OA. Aromatic. O B. it is planar. Oc.it…
A:
Q: What bromo-substituted compound would be required to react with (CH2 = CH)2CuLi in order to form…
A: In order to get the product by using Bromo substituted compound, delete the (CH2 = CH2) group and…
Q: Which of the following best describes the lone pairs on oxygen in the following aromatic molecule.…
A: The given molecule is shown below. In order for a molecule to be aromatic, it should follow…
Q: Arrange in decreasing order according to their reactivity towards an electrophile. H A В
A:
Q: Label each compound as aromatic, antiaromatic, or not aromatic. Assume all completely conjugated…
A: Since you have posted a question with multiple sub-parts we will solve the first three for you. To…
Q: select the expected major organic product.
A:
Q: Which heterocycles are aromatic? N: 'N +O: Z:
A: Aromaticity - It is a property of conjugated cycloalkenes, which leads to excess stabilization of…
Q: Explain why electrophilic aromatic substitution in Pyrrole takes place at C-2 positions whereas, in…
A: To find: The electrophilic aromatic substitution in Pyrrole takes place at C-2 positions whereas, in…
Q: Iow would you carry out each of the following syntheses: ? а) b) c)
A: Given are organic reactions
Q: Which of the following lone pairs of purine participate in its aromaticity? N. C
A:
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

- Identify each one Which one is aromatic antiacromatic or nonaromatic ( need explaination)Determine whether the following molecules are aromatic (A), non-aromatic (NA), or anti-aromatic (AA), and show the use of a Frost circle in determing aromaticity. You may assume planarity.which ones are aromatic? (select all that apply )

