Q: [References] For each example, specify whether the two structures are resonance contributors to the…
A: In resonance there is shifting of electrons. Only Movement of electrons takes place no shifting of…
Q: What is the correct IUPAC name of the following compound? CI Problem viewing the image. Click Here…
A:
Q: Bi(OH)3 + SnO2 →SnO² + Bi Match the following for the above reaction. Drag and drop options on the…
A: The species for which oxidation state is decreased gets reduced and termed as oxidising agent…
Q: Predict the major organic product of the following reactions or provide the reagents needed o…
A: Given : structure of reactant and products.
Q: Under standard state conditions, the complete combustion of a hydrocarbon, primarily produces: O…
A: -> Combustion is a chemical reaction between fuel and an oxidant that produces an oxidized…
Q: 8 a. The alpha decay of 232 Pu 94 b. The positron decay of C c. The beta decay of 437 23TC
A:
Q: 6. The decomposition of N₂Os (g) to NO2 (g) and NO3 (g) has a half-life of 2.81 sec at 298K and…
A: The decomposition of N2O5 is a first order reaction.N2O5(g)→NO2(g)+NO3(g)For a first order reaction,…
Q: d [Review Topics] [References] How many monochloro substitution products are produced when the…
A:
Q: For the standardization procedure, 0.2023 g of anhydrous sodium carbonate was dissolved in 100 mt…
A: mass of anhydrous sodium carbonate, Na2CO3(s) taken for standardization = 0.2023 g Molar mass of…
Q: 2. MS: a. Identify and give the m/z for the base peak and molecular ions b. Determine n appropriate…
A: This question is from mass spectrometry. Base peak : The tallest peak in the spectrum is known as…
Q: 6. What forces compounds to take on specific shapes based on the number of atoms and lone pair…
A: we have to explain the fact that compounds take specific shapes based on the electron pair…
Q: Enough base was added to an amount of water to produce an aqueous solution with [OH-] = 2.4 x 10-5.…
A: Given, Concentration of Hydroxide ion = [OH-] = 2.4 × 10-5 M Concentration hydronium ion = [H3O+] =…
Q: The solubility of CaF, in water at 25°C is 2.05 x 10 mol. dm³. What is the of Ksp at 25°C?
A: It is given that Solubility of CaF2= 2.05 ×10-4 moldm-3
Q: Which of the following molecules has an R configuration? COOH CH CH OH З H₂C CH₂ HOCH COOH CH cooH…
A: We have to determine the configuration and select the molecules having R configuration
Q: What is the number of equivalents per mole, neq. of cupric oxide in the reduction-oxidation reaction…
A: Recall the given redox reaction, CuO s + H2 g → Cu s +…
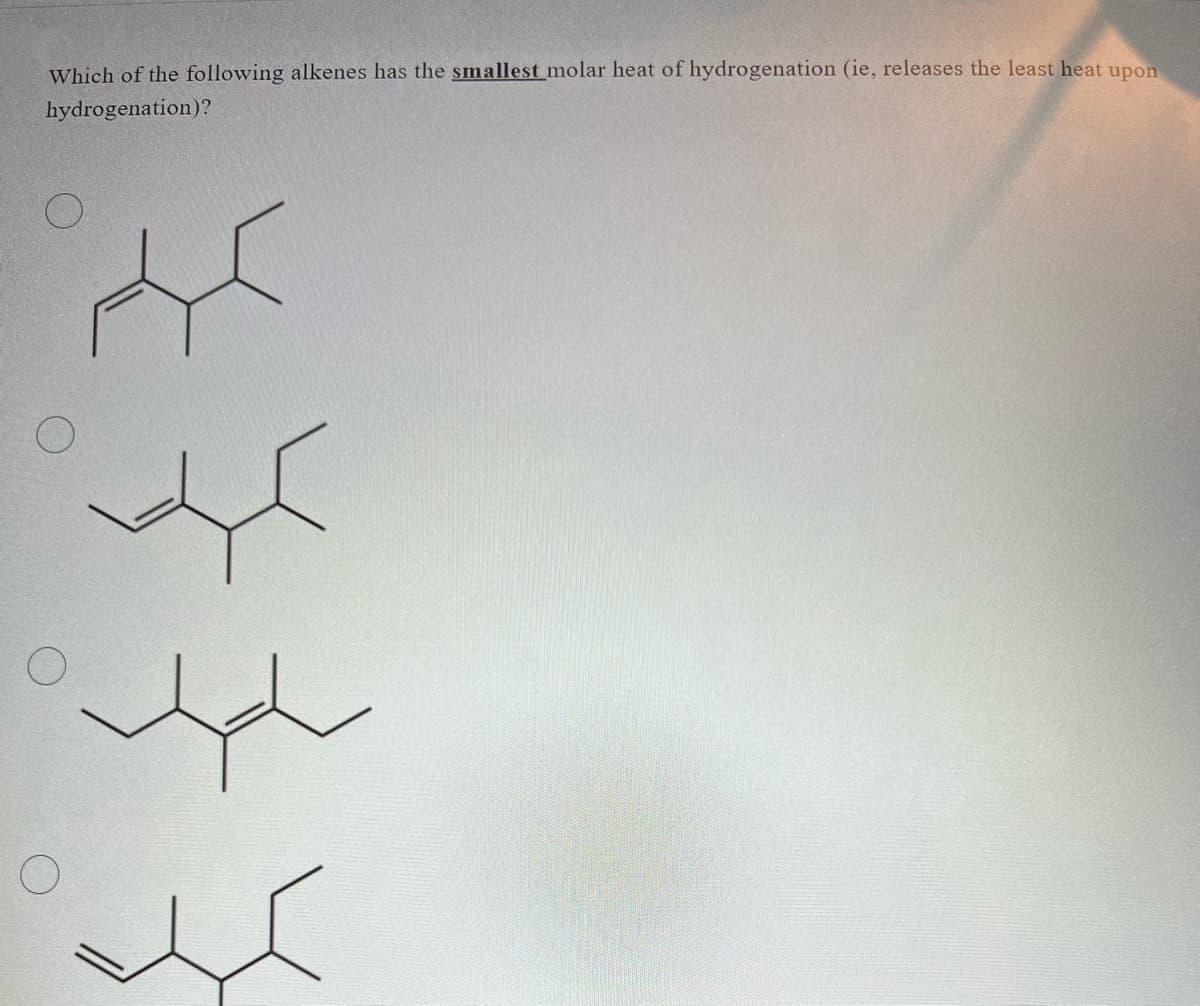
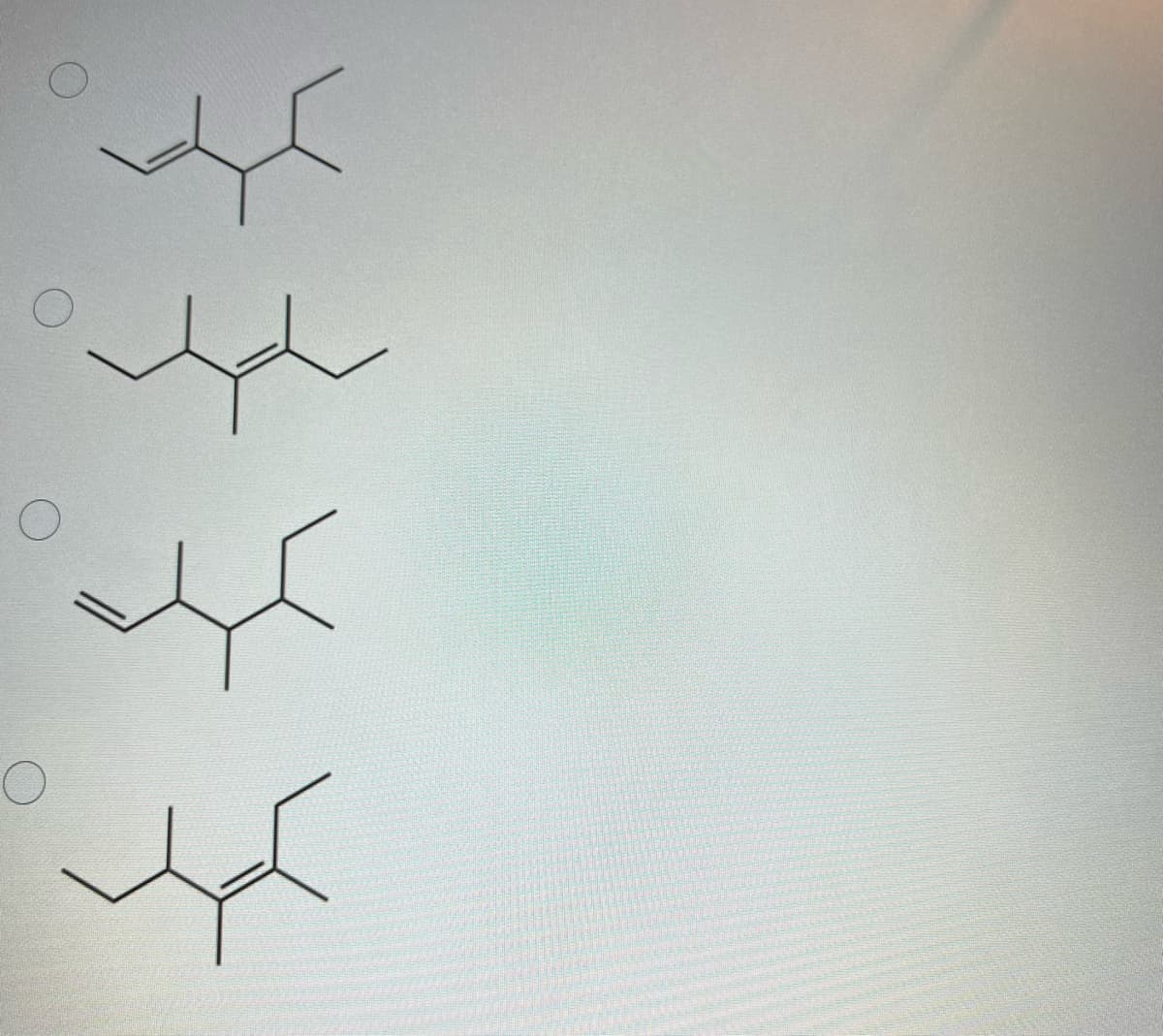
Q: Two isomers, A and B, of molecular formula C5H8 undergo catalytic hydrogenation with hydrogen gas…
A:
Q: Use VSEPR theory to predict the electron-pair geometry and the molecular geometry of XeF5+.
A:
Q: Unshared, or lone, electron pairs play an important role in determining the chemical and physical…
A: Elements have electrons in valence shell called valence electrons . In formation of compounds…
Q: [References] Identify the molecules or lons below as Lewis acids, Lewis bases, or neither. If there…
A: Here, we have to identify the given molecules as Lewis acids, Lewis bases, or neither.
Q: D. Provide a reasonable mechanism for the Diels-Alder reaction between two molecules of…
A:
Q: C. Show the product formed in each step of the following synthetic sequence. CH₂=CH₂ HBr, H₂O₂ A…
A:
Q: Rank the following according to their boiling point (lowest boiling first): A. CH3CH₂OCH₂CH3 B.…
A: Boiling point is the the temperature at which atomospheric pressure of liquid becomes equal to…
Q: The pKa of acetic acid is 4.74. What is the pH of a buffer solution that is prepared from 0.010 M…
A: Buffer solution have tendency to resist pH change on addition of small amount of strong acid or…
Q: The phase diagram of a substance is given below. The region marked "x" represents: super critical…
A: Phase diagram gives the information regarding phase transition of any substance.
Q: This is a three-part question about a tripeptide. Answer all three sub-questions. The pKR of side…
A:
Q: What are the coefficients?
A: Given that, a redox reaction is H5IO6 + Cr → IO3- + Cr3+. We have to write the coefficients of the…
Q: For Water at 15 bar and 400°C What is the specific internal energy in kJ/kg?
A:
Q: Which compound below contains both covalent and ionic bonds?
A:
Q: Arrange the following compound in the order of increasing acidity (least acidic first) I)…
A: Given compounds: I) Cyclohexanol II) 4-Nitrophenol III) Phenol IV) 4-Chlorophenol The acidity…
Q: How would this chemical react with (CH2=CH)2CuLi?
A: Organic reactions are those in which organic reactant react to form organic products.
Q: The meals-ready-to-eat (MREs) in the military can be heated on a flameless heater. You can purchase…
A:
Q: Cyclopropane is a highly strained molecule. Why? Select all that apply. Why is cyclopropane highly…
A: The carbon atoms in cyclopropane is SP3 hybridized, so the bond angle should be 109o28/ . But due to…
Q: H3C НО OCH3 ОН тос H3C НО LOCH 3 OH
A: Requirement from question: Four questions. Identify whether they are stereoisomers, constitutional,…
Q: 2. What do you think the bubbles are from the reaction between vinegar and baking soda?
A:
Q: Fe | Fe²+ (0.100M) || Cd²+(0.001) | Cd I. Write the half-reactions and cell reaction 2. Calculate…
A:
Q: Use electrode potential data in Table 19.1 to determine A,Gº for the reaction: 2 Al(s) + 3 Br₂(1)→…
A:
Q: Et3N ** Et₂O H Find out the product A, B and C. i. i. Me NaNH, NH3 NaNH, B ii. ii. Br H₂O (Weak…
A: Note - The given question is a multiple questions , hence I solved first question according to rules…
Q: When the following molecule is subjected to free radical chlorination, how many different chiral…
A:
Q: What product will be formed when the chemical undergoes reaction with NBS and hv ?
A:
Q: Glucose from molasses is fermented to alcohol using S. cerevisiae where 500 ml of CO2 (at standard…
A:
Q: a) CH3 -CH-CH-COOH + Ca → CH, NH, b) CH₂ - CH-CH-COOH + CH₂OH- CH, NH, c) CH3 -CH-CH-COOH + Ba(OH)₂…
A:
Q: Arrange the following compound in the order of increasing acidity (least acidic first) I)…
A: Phenol is more acidic than cyclohexanol due to stability of phenoxide ion. The conjugate base of…
Q: Two gas mixtures, A and B, are compared for their carbon dioxide content. Mixture A has 50%…
A: Gas-A is given moles basis,so we need to convert to mass basis, by using molar mass of the…
Q: Compound A of molecular formula C8H₁4 is reduced by sodium in liquid ammonia to give compound B of…
A:
Q: An engineer turns the temperature of a gas down, holding the volume constant at 1,250 L. Initially,…
A:
Q: answers clearly. 1.1) From the concept of reversed SN2 Disconnection, draw the best set of positive…
A:
Q: What is/are the reagent(s) for the following reaction? Problem viewing the image, Click Here O…
A: The conversion of a functional group to the other functional group is carried out in the presence of…
Q: What is the correct IUPAC name of the following compound? Problem viewing the image. Click Here…
A: -> For IUPAC naming following steps are required - First of all identify the longest carbon…
Q: Do the molecules below have a permanent electric dipole moment?
A: Dipole Movement The permanent electric dipole moment (EDM) of a particle or molecule is a separation…
Q: C. a A galvanic cell consists of a chromium metal electrode in a solution of Cr(NO) and a zinc metal…
A:
Step by step
Solved in 3 steps with 2 images

- What alkene is the major productwhen 2-bromo-2-methylpentane is treated with sodium ethoxide in ethanol?Choose the two words which complete the following sentence to make a true statement. As the number of conjugated double bonds in an alkene increases, the difference between electronic energy levels becomes ______ and the λ(max) wavelength _______. A.) larger, increases b.) larger, decreases c.) smaller, increases d.) smaller, decreasesFor the following reactions, determine the type of reaction (either combustion, hydrogenation of alkenes, or hydration of alkenes) & please make sure to show the products for each of the reactions
- For each of the following alkenes, assign the configuration of the double bond as either Z or Ewhich of the following reagents can oxidize an alkene? H2/Pt, HCl, H2SO4, KMnO4How many moles of Bra are required to completely halogenate the alkene?A. One moleB. Two molesC. Three molesD. Four moles What is the expected arrangement of the bromine atoms relative to each other amongthe carbon involved in pi bonding?A. anti-conformationB. syn-conformationC. trans-configurationD. cis-configuration What happens to bromine when it is adjacent to an alkene during a chemical reaction?A. Bromine becomes stable. (? kasi before brown siya/acidic tas naging colorless? Jk ewan)B. Bromine becomes polarized.C. Bromine becomes hybridized.D. Bromine becomes acidic. The relative arrangement of bromine atoms in the product is primarily due to:A. ElectronegativityB. RepulsionC. Hydrogen bondingD. Atomic weightWhat is your observation after the reaction?A. A yellow flame is produced.B. Bromine water decolorizes.C. The alkene becomes denser.D. A brown precipitate forms.

