Which of the following aqueous solutions are buffer solutions? (Select all that apply.) ()0.21 M HCN + 0.18 M KCN U 0.32 M NaBr + 0.20 M Nal 0.23 M HBr + 0.20 M NaBr 0.30 M NH4NO3 + 0.38 M NH3 00.20 M KOH + 0.23 M KC1 Submit Answer to access important values if n Retry Entire Group 4 more group attempts remaining
Which of the following aqueous solutions are buffer solutions? (Select all that apply.) ()0.21 M HCN + 0.18 M KCN U 0.32 M NaBr + 0.20 M Nal 0.23 M HBr + 0.20 M NaBr 0.30 M NH4NO3 + 0.38 M NH3 00.20 M KOH + 0.23 M KC1 Submit Answer to access important values if n Retry Entire Group 4 more group attempts remaining
Chemistry: An Atoms First Approach
2nd Edition
ISBN:9781305079243
Author:Steven S. Zumdahl, Susan A. Zumdahl
Publisher:Steven S. Zumdahl, Susan A. Zumdahl
Chapter14: Acid- Base Equilibria
Section: Chapter Questions
Problem 102CWP: Consider the following acids and bases: HCO2H Ka = 1.8 104 HOBr Ka = 2.0 109 (C2H5)2NH Kb = 1.3 ...
Related questions
Question
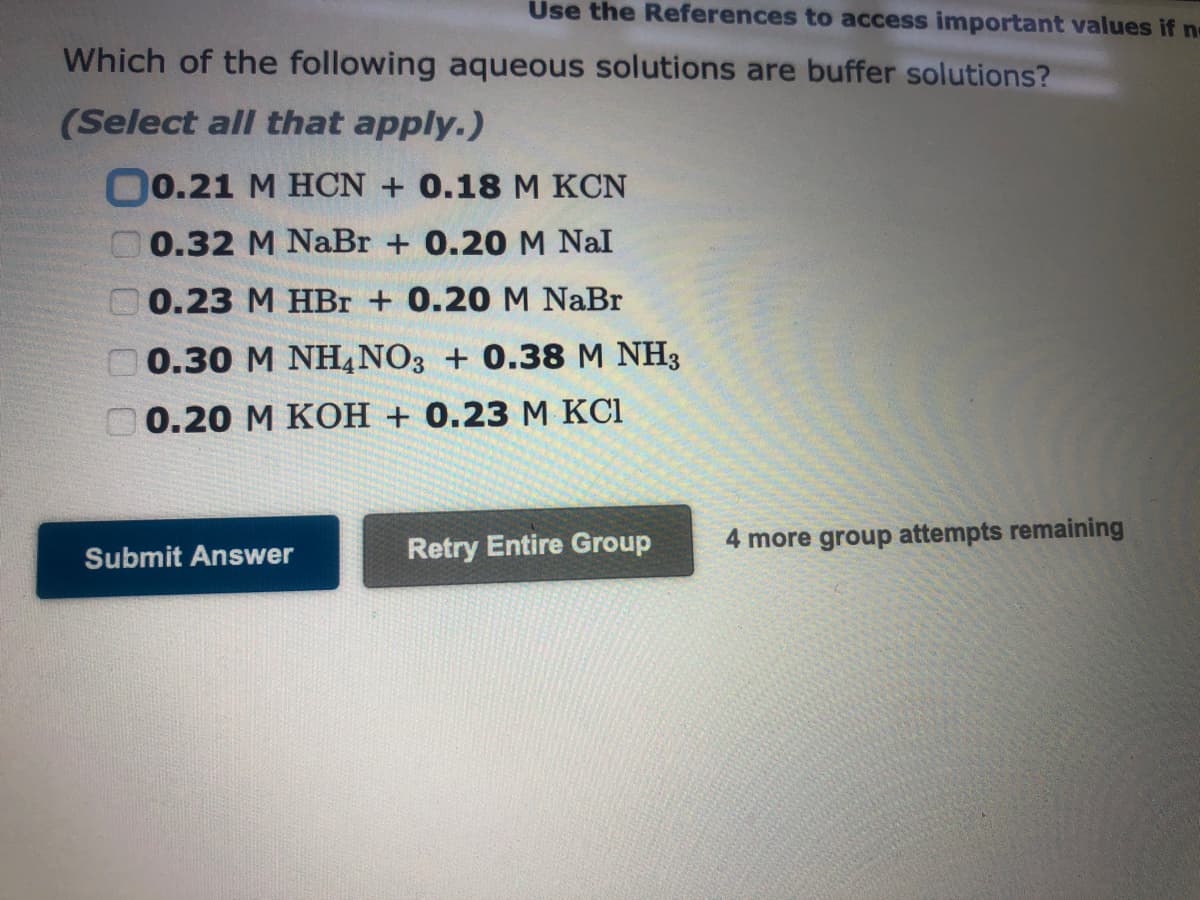
Transcribed Image Text:Use the References to access important values if no
Which of the following aqueous solutions are buffer solutions?
(Select all that apply.)
( 0.21 M HCN + 0.18 M KCN
G 0.32 M NaBr + 0.20 M Nal
0.23 M HBr + 0.20 M NaBr
0.30 M NH4NO3 + 0.38 M NH3
0.20 M KOH +0.23 M KC1
Submit Answer
Retry Entire Group
4 more group attempts remaining
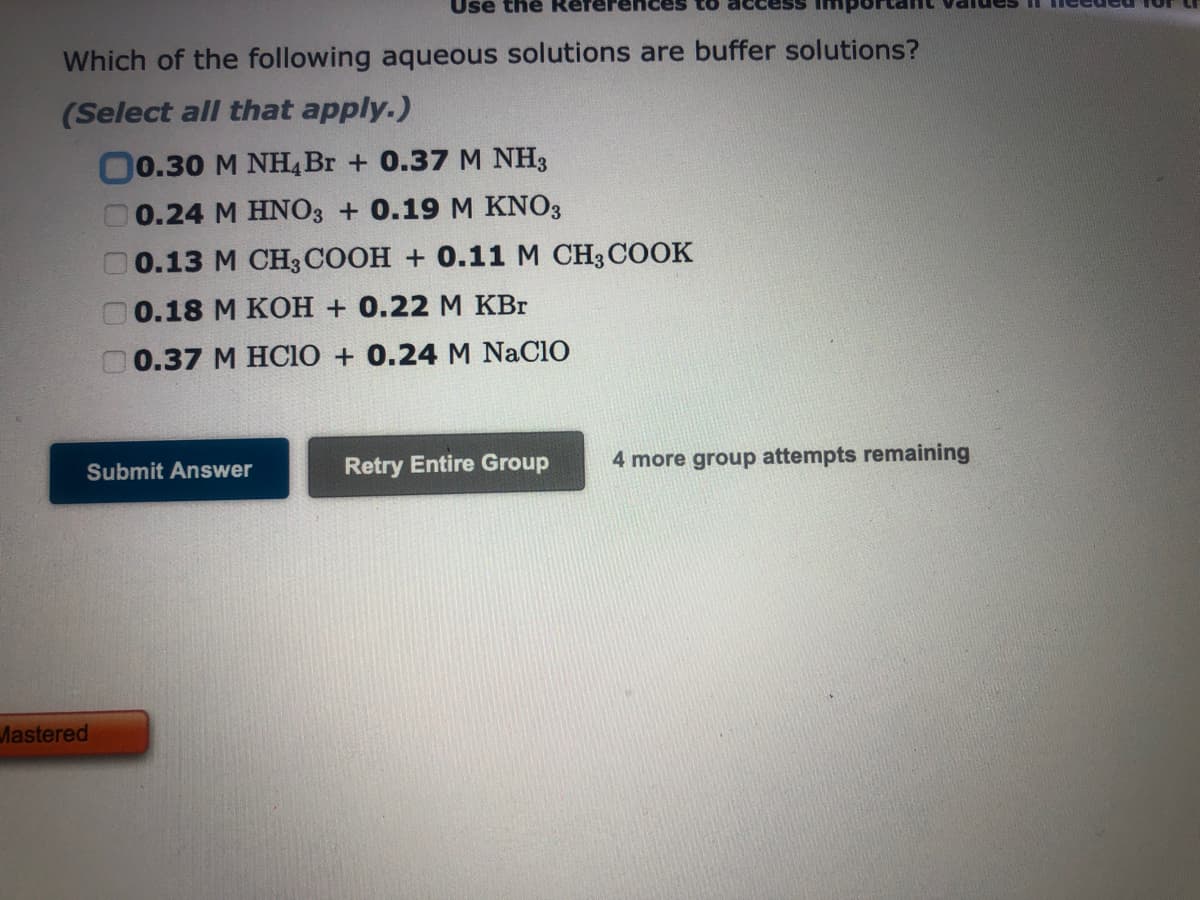
Transcribed Image Text:Which of the following aqueous solutions are buffer solutions?
(Select all that apply.)
00.30 M NH4 Br + 0.37 M NH3
0.24 M HNO3 + 0.19 M KNO3
0.13 M CH3COOH + 0.11 M CH3COOK
0.18 M KOH + 0.22 M KBr
0.37 M HCIO + 0.24 M NACIO
Submit Answer
Use the
Mastered
Retry Entire Group
4 more group attempts remaining
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning
