Chapter30: Orbitals And Organic Chemistry: Pericyclic Reactions
Section30.SE: Something Extra
Problem 14MP: Plastic photochromic sunglasses are based on the following reversible rearrangement of a dye inside...
Related questions
Question

Transcribed Image Text:Question 36
The solubility of compound B is 0.15 g/100 mL of water or 0.45 g/ 100 mL of ether. Calculate the volume of ether needed to remove 95% of the compound B from 100 mL of a saturated aqueous solution in single extraction. Give your answer in zero decimal places and don't use scientific notation.
no
K =
V.
aq
n,
´aq
aq
V.
naq.
п,
KV, +Vaa
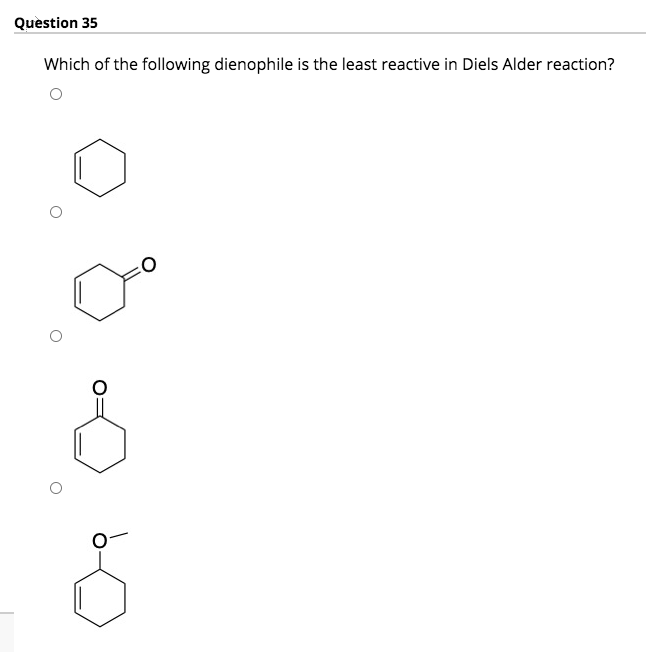
Transcribed Image Text:Question 35
Which of the following dienophile is the least reactive in Diels Alder reaction?
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

