Q: 270-mL flask contains pure helium at a pressure of 759 torr . A second flask with a volume of 455 mL...
A: Given conditions Volume of He flask = 270mL Volume of Ar flask = 455 mL Pressure of He = 759 torr ...
Q: Analyzing Liquids (Pepsi, Ice Tea and 50% Glucose) Interpretation: (explain the figure shown)
A: A question based on IR spectroscopy that is to be accomplished.
Q: Find each part of this Metathesis reaction: Ammonium Carbonate and Phosphoric Acid Molecular equ...
A: Metathesis reaction is also called as double displacement reaction. In this cations and anions are ...
Q: 1. A gas mixture contains each of the following gases at the indicated partial pressures: N2, 226 to...
A: Given :- PN2 = 226 torr PO2 = 122 torr PHe = 107 torr Volume = 1.50 L Temperature = 25°C To ca...
Q: Question 16 Simple extractions are particularly useful for separations where only one component has ...
A: Liquid liquid extraction are also known as solvent extraction is defined as the method used to separ...
Q: The reaction A - C was performed and the concentration vs. time data below collected. time, s 1 2 3 ...
A:
Q: Aqueous sodium carbonate – Na2CO3 – and aqueous aluminum nitrate – Al(NO3)3 – combine to rapidly for...
A: Aqueous sodium carbonate – Na2CO3 – and aqueous aluminum nitrate – Al(NO3)3
Q: 3. Given the following set of fluorescence data for Fluorescein, solve for the following: A. LLOD (i...
A: To Calculate: A. LLOD B. LLOQ C. Linear range of quantification
Q: A 14.5-L scuba diving tank contains a helium-oxygen (heliox) mixture made up of 24.5 g of He and 4.4...
A: Given, A 14.5-L scuba diving tank contains a helium-oxygen ( heliox ) mixture made up of 24.5 g of ...
Q: Draw the structure(s) of the major organic product(s) of the following reaction. Dilute aqueous HCI ...
A:
Q: what is the pH of a solution obtained by mixing 225 mL of 0.680 M HCl with 565 mL of an HCl solution...
A: Here, we have to calculate the pH of a solution obtained by mixing 225 mL of 0.680 M HCl with 565 mL...
Q: 18. When 1 mole of ethene in the gas state reacts with 1 mole of water in the gas state, 1 mole of e...
A:
Q: What liquid is usually used and measured when calibrating glassware? a. Water with detergent O b. Br...
A: Answer What liquid is usually and measured when calibrating glasswar...
Q: Nitrosyl bromide, NOB,, is formed from NO and Br2: 2 NO(9) + Br2(9) – 2 NOB1(g) Experiments show tha...
A: Please find your solution below : Rate constant also known as rate equation is an expression that re...
Q: Which statement properly describe Roult's Law? Raoult's law states that the vapor pressure of a solv...
A: Roult's law says that the vapour pressure of the pure solvent decreases when some amount of non-vola...
Q: CH3 CH3 CI CI- H CI CI- CH3 ČH3 These two structures represent: O a pair of structural isomers O a p...
A:
Q: A 20.00-ml sample contains 2.39 g of KHC, H,O,, known as KHP. This sample is used to standardize an ...
A:
Q: What is the major product of the following reaction? * cO,Me heat cO,Me CO,Me CO,Me CO,Me CO,Me CO-M...
A:
Q: A and B react to produce C and D: A +B=C+ D At a certain temperature, the equilibrium constant K has...
A:
Q: Use the energy diagram for the reaction A D to answer Im the questions. How many transition states a...
A:
Q: 200 mL Ethanol (d= 0.789 g/ml) was used to dissolve a certain 1.000 g of organic compound. The boili...
A: Volume of ethanol = 200mL Mass of organic compound = 1.000g B.P of mixture = 78.47C Kb = 1.20 C /m
Q: sample of gas has a mass of 38.9 mg. Its volume is 226 mL at a temperature of 57 ∘C and a pressure o...
A: Assuming the gas to be an ideal gas, ideal gas equation will be used to calculate the molar mass of ...
Q: QUESTION 6 If you have 4 moles of CH4 molecules, how many atoms do you have? O 4 20 4 moles O 5 mole...
A: Any molecule is consists of number of atoms and these atoms can be same or different to form a parti...
Q: Give the synthesis mechanism for following molecule from methane, ethane or benzene. give clear hand...
A: Here we have to synthesize the target compound from starting material benzene or methane and ethane ...
Q: When the substance is soluble in both liquids, the substance will be distributed to the two liquids....
A: There are given two statements - When the substance is soluble in both liquids,the substance will b...
Q: 11 5, de /188795/assignments/3601808 Homework Ch16 - Attempt 1 Problem 16.61 3 of 15 Constants | Pe...
A: Given- Concentration = 0.13M pKa = 2.32
Q: One equivalent of 1,3-butadiene reacts with the quinone below to produce a single product. Why is th...
A: In Diels alder reaction,diene reacts with dienophile.
Q: 2. Write balanced net ionic equations describing each of the following reactions: Precipitation of t...
A: Solution: (1) Precipitation of tin(IV) sulfide with H2S: Sn4+(aq) + H2S(aq) ----> SnS2(s) + 4H+(a...
Q: Convert 469 mm Hg to atm. • Your answer should be in units of atm. Do not include the units on your ...
A: mm Hg and atm are unit of Pressure. Relationship between mm Hg and atm, 1 atm = 760 mm Hg Or, 1 mm ...
Q: Question 9 Given the volume of 0.2000 M NaOH used determine the concentration of 10.00 ml sample eac...
A: Given, Concentration of NaOH = 0.2000 M Volume of NaOH for aqueous (Aq) layer = 12.55 mL Volume of N...
Q: Suppose that 6.56 moles of A at 4.10 atm and 298.15 K and 5.35 moles of B at 4.82 atm and 298.15 K a...
A: Given: For gas A Moles (nA) = 6.56 mol Pressure (PA) = 4.10 atm Temperature = 298.15 K For gas B Mol...
Q: What is the major product of the reaction below? * (1) 1 eq. Me MgCI - Major product (2) H2O HO Me M...
A:
Q: Simple extractions are particularly useful for separations where only one component has a favorable ...
A: Several important separation techniques are based on a simple extraction, including liquid–liquid, l...
Q: Calculate the rate constant, k, for a reaction at 63.0 °C that has an activation energy of 76.9 kJ/m...
A: The relation between rate constant, temperature and activation energy is given by Arrehenius equati...
Q: Water and sodium chloride are both considered substances as well as compounds, but water exists as m...
A: In this question, we have to give the reason why water and sodium chloride are both considered subst...
Q: NaSCH3 DMSO Mechanism Determine the anticipated ELIIMINATION mechanism for the reaction below. Using...
A: a) Here, substrate is secondary alkyl bromide and nucleophile is strongest that is NaSCH3 and moreov...
Q: (g) + 50,g) = 4NO(g) + 6H,Og) H.CO,(aq) = H¸O) + CO,(g) H,Og) + CG) 2 H(g) + CO(g) O(g) + 4NO(g) = 2...
A: Equilibrium constant expression includes concentration/partial pressure of products to reactants A) ...
Q: Temperature Dependence I The rate constant for a reaction relates to activation energy and temperatu...
A: Arrhenius EquationThe Arrhenius condition is an articulation that gives a connection between the rat...
Q: d) Calculate the pH, [H+] and [OH-] of a solution with a pOH of 5.36
A: Since you have posted multiple questions, the answer for first question is given below. Kindly repos...
Q: 1
A: Use the graphing tool above to make an appropriate plot and determine the activation energy, Ea for ...
Q: CH3 но -CH CH3 H3C 0-CH3 This compound has how many chiral carbons? 1 O 5 O 6 O 2 O 3
A:
Q: 7. The ka for acetic acid is 1.0 x 10.5. Is acetic acid a weak or a strong acid?
A: Strong acids completely dissociate in aqueous solution (Ka > 1, pKa < 1).
Q: What is the name of the following compound? Is Ethoxycyclopentane correct?
A: 1. Select the principle carbon chain 2. Numbering 3. Naming (prefix + word root + suffix)
Q: Hi can you please help At what temperature will the vibrational partition function for I2(IODINE)...
A: The vibrational partition function is given as: qv=11-exp-hνkT (1) Where h is the Planck's co...
Q: When writing a complete atomic symbol, there are four numbers used – in the upper-left, upper-right,...
A: In this question, we have to answer that When writing a complete atomic symbol, there are four numbe...
Q: 2. A solution that may contain Cu", Bi?", Sn*, or Sb2* is treated with thioacetamide in an acidic me...
A: Thioacetamide with molecular formula C2H5NS is an organosulfur compound, which acts as a supplier of...
Q: Given the cell notation: Fe/Fe2+ (0.100 M) (E0 = -0.44) Cd2+ (0.001 M)/Cd (E0 = -0.403) Write ...
A:
Q: What is the mass % of propylene glycol in a 1.89 M solution of propylene glycol (MM = 76.09 g/mol) i...
A: Given- Molarity = 1.89 M Molar mass = 76.09 gm/mole Density of solution = 1.03 g/ml
Q: Determine the amount of heat (in kJ) associated with the production of 1.76 × 104 g of NO2 according...
A: Molar mass of nitrogen dioxide is 46 g/mol
Q: or introduce the following topics of polymer : • Thermoplastic/thermoset elastomer •Addition/condens...
A: The process of formation of large molecules from repetitive addition of monomer units is called as p...
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

- Which of the following dienes have an s-cis conformation, and which have an s-trans conformation? Of the s-trans dienes, which can readily rotate to s-cis?Bicyclohexadiene, also known as Dewar benzene, is extremely stable despite the fact that its rearrangement to benzene is energetically favored. Explain why the rearrangement is so slow.Cycloheptatrienyl radical (C7H7∙) contains a cyclic, completely conjugated system of π electrons. Is it aromatic? Is it antiaromatic? Explain.
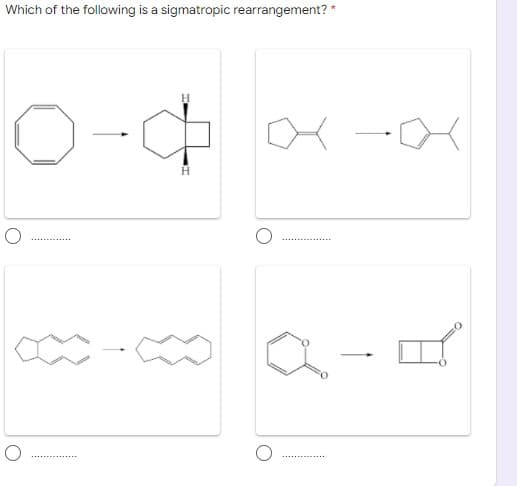
- Examine the following pericyclic reactions. For each reaction, tell whether it is an electrocyclic reaction, a cycloaddition reaction, or a sigmatropic rearrangement.What is the unsaturation of the following molecule?Which of the following carbocations is(are) likely to rearrange? a. I b. II c. III d. II and III e. I and II
- Is the following alkene E,Z, or neither? a. E b. Z c. neither d. It is one or the other, but which is impossible to determine from the information givenCarbocations are carbons bearing a formal positive charge. a. Carbocations can be stabilized by induction and hyperconjugation. Explain each of these forms of stabilization.If a trans alkene is reacted with OsO4 in H2S, the product is: Meso compound Hoffman product Markovnikov product Racemic mixture When 2-methylpropene is reacted with HCl in ether, the product is: 1-chloro-2-methylpropane 2-chloro-2-methylpropane 2-chloromethylpropene 3-chloro-2-methylpropane Which of the following is not true for the hydroboration reaction of alkenes? Anti-markovnikov Syn addition No incorporation of solvent rearrangements are observed
- Is the structure, transition state and relative stereochemistry of the products is correct? Is the curly arrow mechanism is correct ?For the following dienes, identify whether the molecules has any cis or trans stereochemistry and idenfity the conformation as either s-cis or s-trans, where appropriate. Please also note whether the compound would be reactive in a Diels -Alder reactionWhich of the following statements about cycloaddition reactions is not true? Cycloaddition reactions form a cyclic product with two new bonds. The course of the reaction is determined by the symmetry of the molecular orbitals of the products. Cycloaddition reactions are concerted. Cycloaddition reactions are stereospecific.

