Which of the following is TRUE regarding a precipitation reactioh. A) Both products must be soluble in water B) At least one of the products will be insoluble in water C) Spectator ions are always included in the reaction D) Electrons are gained and lost E) H+ ions are transferred from one molecule to another
Which of the following is TRUE regarding a precipitation reactioh. A) Both products must be soluble in water B) At least one of the products will be insoluble in water C) Spectator ions are always included in the reaction D) Electrons are gained and lost E) H+ ions are transferred from one molecule to another
Chapter18: Introduction To Electrochemistry
Section: Chapter Questions
Problem 18.7QAP
Related questions
Question
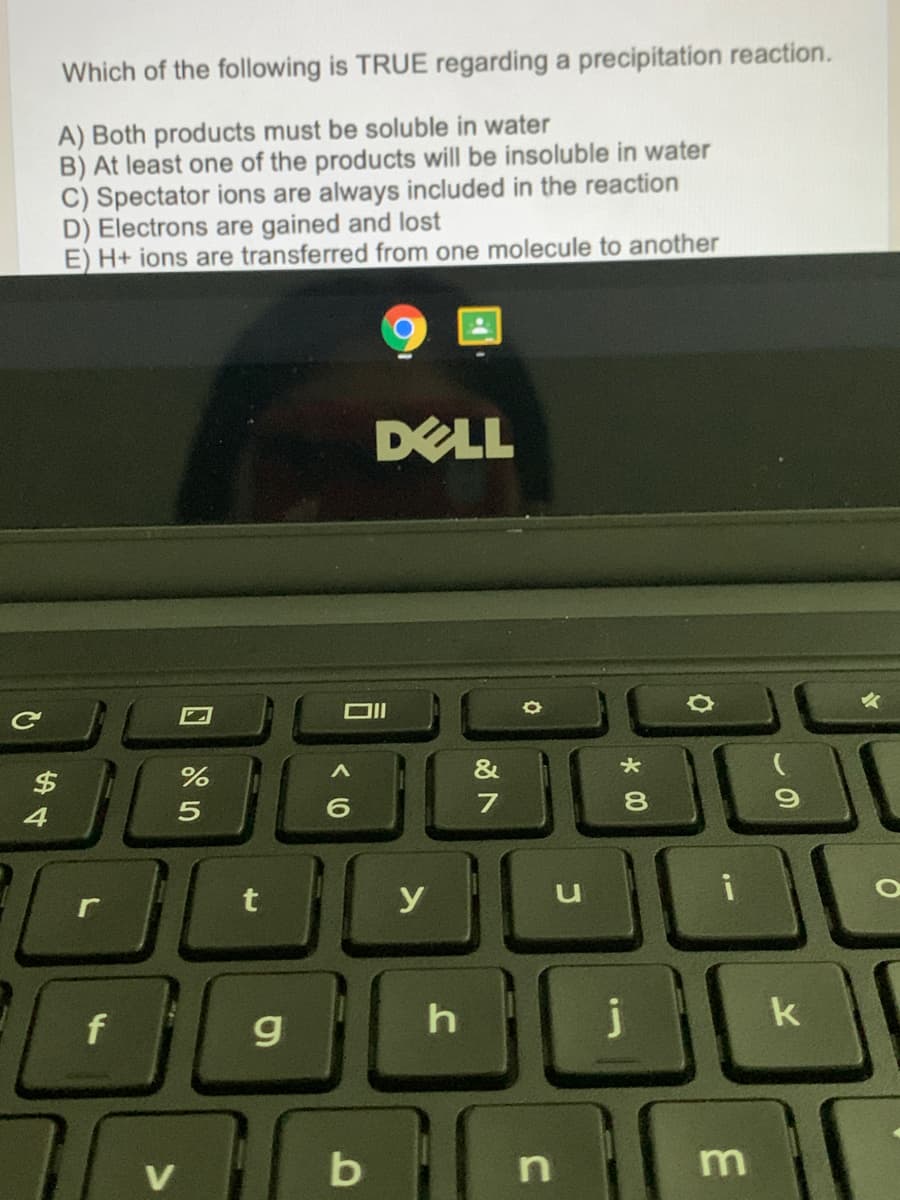
Transcribed Image Text:Which of the following is TRUE regarding a precipitation reaction.
A) Both products must be soluble in water
B) At least one of the products will be insoluble in water
C) Spectator ions are always included in the reaction
D) Electrons are gained and lost
E) H+ ions are transferred from one molecule to another
DELL
&
$
7
4
y
h
k
V
口 85
00 *
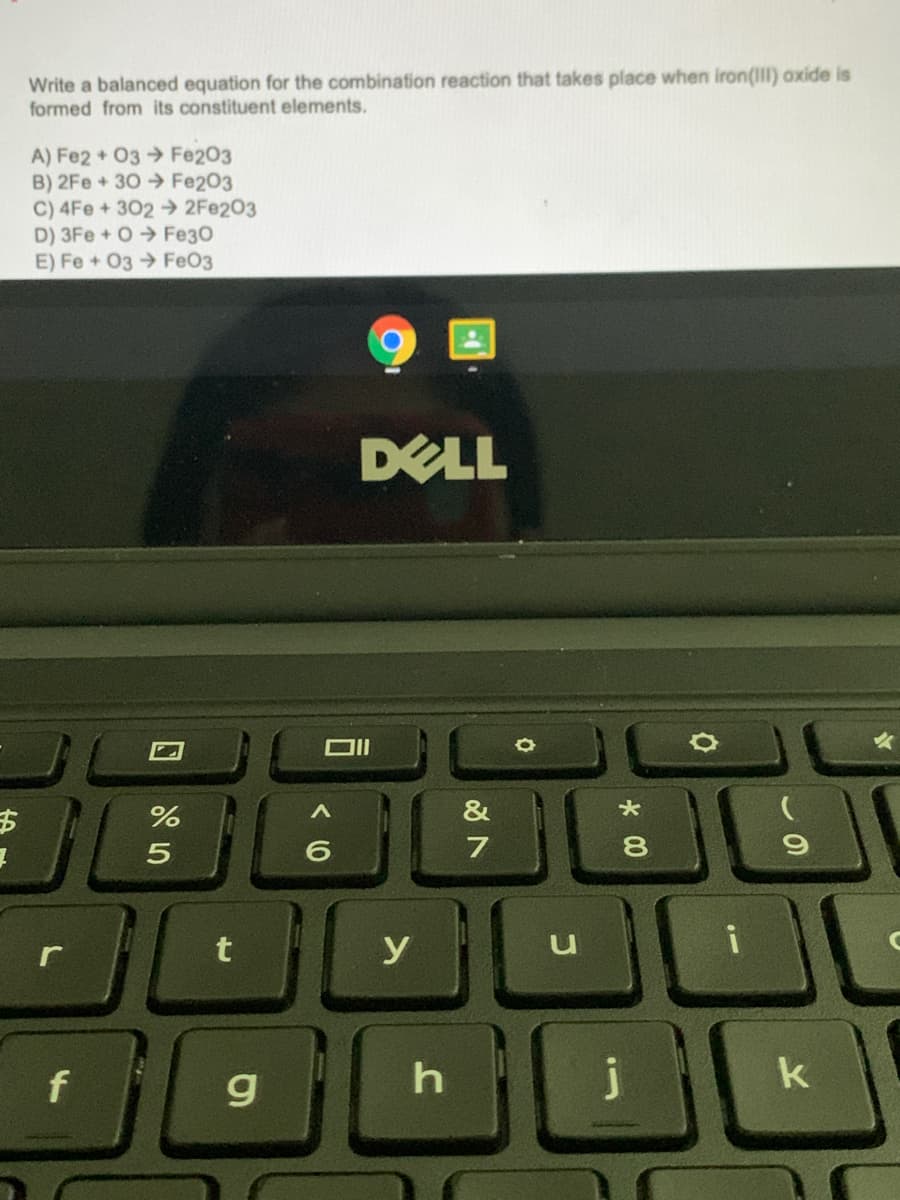
Transcribed Image Text:Write a balanced equation for the combination reaction that takes place when iron(III) oxide is
formed from its constituent elements.
A) Fe2 + 03 Fe203
B) 2Fe+30→ Fe203
C) 4Fe + 302 2FE203
D) 3Fe + O Fe30
E) Fe + 03 FeO3
DELL
&
大
7
y
f
g
h
k
* 00
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning



Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning


Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:
9780078746376
Author:
Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:
Glencoe/McGraw-Hill School Pub Co

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning