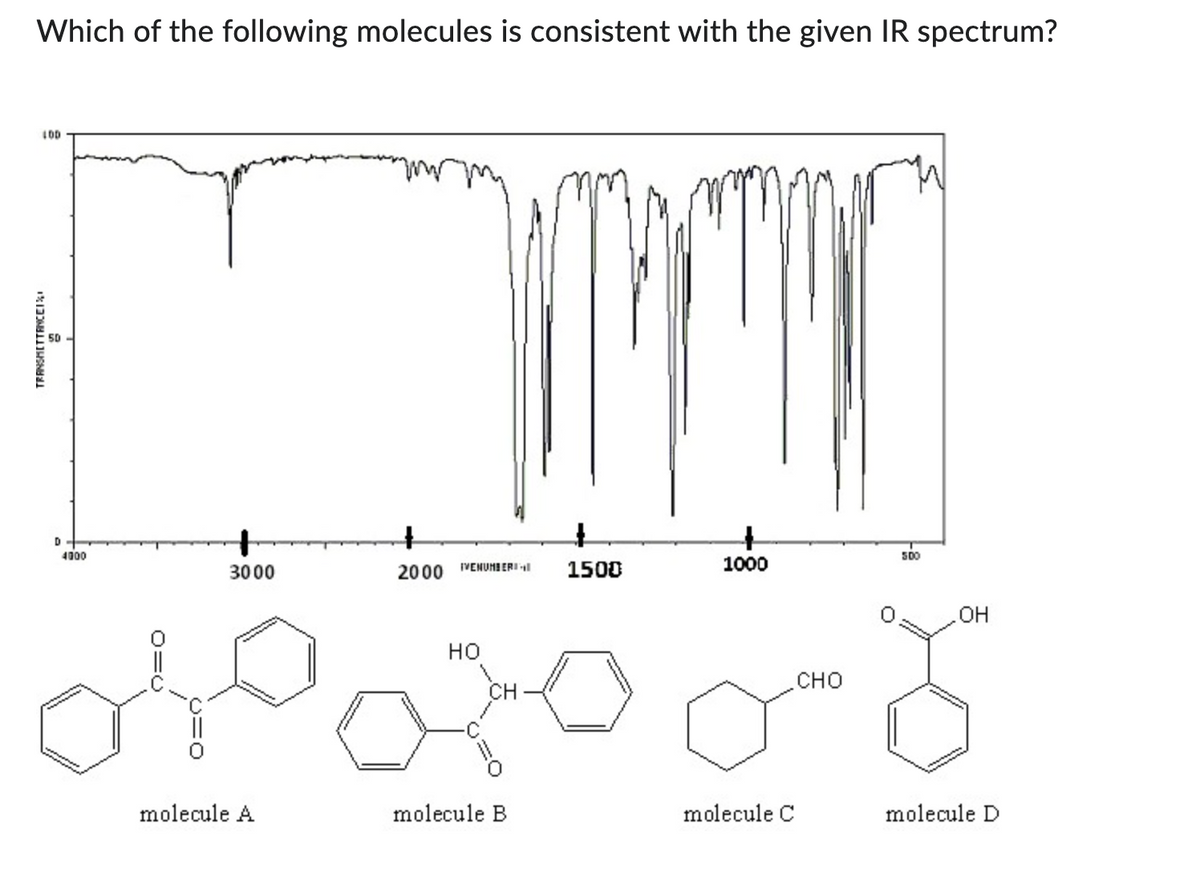
Which of the following molecules is consistent with the given IR spectrum? LOD TRANSMETTANCEI · 4000 3000 1000 2000 VENUMBER1 --1 1500 HO အဌဝတွင် molecule B molecule A molecule C 50 CHO .OH molecule D
Q: Which of the following compounds has the highest boiling point? 01 O III ONV NH₂ 11 ~ IV Br
A: To solve this problem we have to identify the compound having highest boiling point.
Q: CH2OH нон но-н но-н н-он CH2OH VS но- CH2OH -Н H—OH Н- -ОН но- -H CH2OH I I
A: A and B are given here and we have to determine relationship between these given molecule . As these…
Q: How much hydrochloric acid (in mol) should theoretically react with one antacid tablet containing…
A: Weight of CaCO3 = 750 mg = 0.750 g (1g = 1000 mg)Molecular weight= 100.089 g/mol
Q: 3D. Traditional Friedel Crafts acylation reactions often use water sensitive reagents and produce…
A: Friedel-Crafts acylation is an acylation of aromatics using an acyl halide and a Lewis acid…
Q: Consider the following structure as you answer the questions below: CH3 CH₂-C-N-CH3 CI Identify the…
A:
Q: 9) Draw the "hexagon" line structure of the following (be sure to indicate stereochemistry). FOLOH…
A: To draw the planar 2D structure of a cyclic ester given in chair form with their respective…
Q: 8. Which of the following compounds exhibits the strongest London dispersion forces?…
A:
Q: Please use a computer program to illustrate and/or type out the explanation. This cannot be hand…
A: 1H-NMR is mainly used for the identification of complete structure of the organic componds. From the…
Q: The reaction of LDA with acetophenone produces halogenation an ylide an enol an enolate alkylation
A:
Q: What reagent(s) would you use to convert the compound in part a to the product shown below? H…
A: Reactant -Product - The given reactant is an alcohol and the product to be formed is an aldehyde.…
Q: What is the major product of the following reaction? 1) LIAIHA 2) H₂O or H3O*
A: A question based on reactions of epoxides. 5 possible products of epoxide reduction are given, the…
Q: Gnereally speaking, which of the following is the most reactive to reactions with a nucleophile? A B…
A: We know priority order - Aromaticity > Mesomeric > Hyperconjugation > Inductive effect +I…
Q: Which of the following are products of the reaction below? Select all that apply. Labmaing obud A.…
A: This problem of organic reaction is based on use of an organometallic catalyst : Grubbs catalyst…
Q: Identify the relationship between the following pairs of molecules [constitutional isomers,…
A: Compounds having the same molecular formula but differently oriented atoms are called isomers. When…
Q: Which of the following compounds is(are) hydrolyzed to butanoic acid upon heating in H₂O, H₂SO4?…
A: -> Ester on hydrolysis give carboxylic acid and alcohol.-> Amide on hydrolysis give amine and…
Q: It took 5.7 minutes for 4.0 L helium to effuse through a porous barrier. How long will it take for…
A: Given,Volume of helium gas = 4.0 Ltime taken for effusion = 5.7 minutes.Volume of helium gas = 4.0 L
Q: Cl CI
A: Name of given compound is 1,2-dichlorocyclobutane.Now to determine the point group of a molecule we…
Q: What is needed to complete the following reaction? (CH3)2NH CH3NH2 (CH3)3N (CH3)4N+ H3C. CH3 H3C NCH…
A: When primary amines treated with carbonyl compounds produces imines.
Q: What is the name of the following?
A: IUPAC nomenclature is world wide accepted naming of organic compound using rules .
Q: What is the product of the reaction shown? 1. Li 2. Cul 3. CH3CH₂Br Br IV .Li V Br A) I B) II C) III…
A: Corey House synthesis is an organic reaction where an alkyl halide react with lithium in dry ether…
Q: please explain why A is S configuration and not R. I thought #1 priority would be the chain with OH…
A: Absolute configuration i.e. R/S configuration is decided on the basis of CIP (Cahn-Ingold Prelog)…
Q: Question: Express the quantity 546.3 x 10^-12 in each unit a) ms b) ns c) ps d) fs
A: Quantity= 546.3 × 10-12 s We know that ->1ms = 10-3 s 1 ns = 10-9 s 1 ps = 10-12 s1 fs = 10-15 s
Q: For the combustion of methane, the standard entropy change and the entropy change of the…
A: For combustion of methane the standard entropy change and the entropy change of the surrounding…
Q: CO₂Me MeO₂C dimethyl fumarate A 1,3-butadiene A CO₂Me CO₂Me dimethyl maleate
A:
Q: What is the equilibrium constant of the following reaction at 25˚C? NaCNO(aq) + HI(aq) ⇄ HCNO(aq) +…
A: The given chemical reaction present in the problem is-- NaCNO(aq) + HI(aq) ⇄ HCNO(aq) + NaI(aq)
Q: A volume of 500.0 mL of 0.160 M NaOH is added to 525 mL of 0.250 M weak acid (K₁ = 3.24 × 10-5).…
A: Volume of NaOH = 500.0 ml Molarity of NaOH = 0.160 MVolume of weak acid = 525 mlMolarity of weak…
Q: When plutonium-241 is bombarded with another nucleus, five neutrons and new isotope with an atomic…
A: In the process of radioactive decay, unstable nuclei emit particles such as alpha particles,…
Q: COMPOUND LEWIS DOT STRUCTURE HCN (use black sphere for C) SO3 (use black sphere for S) NO₂™ (use…
A: According to Q&A guidelines of Bartleby, we are supposed to answer only the first 3 sub-parts…
Q: Indicate the name of the parent for each of the compounds shown. НО. CI A Compound A is Compound B…
A: IUPAC name are based on certain rules, first the parent compound is to be found and named, then the…
Q: ly to read ons where the nuclear charge is less shielded and are thus er as nucleophilic or…
A: Since you asked multiple questions so as per Q&A guidelines of portal I solved first three parts…
Q: 10) Which compound is more acidic: phenol or 4-nitrophenol? Draw the mesomeric structures of the…
A:
Q: Draw the following esters a) ethyl butanoate b) pentyl propano ate c) propyl 3-ethylhexanoate d)…
A: As you have asked multiple questions, we will solve only first question for you according to our…
Q: The free energy change for the following reaction at 25 °C, when Cr³+ = 1.20 M and Zn²+ 2Cr³+ (1.20…
A: In the question, the reaction isWe need to find for this reaction. and
Q: When a lot of ammonium nitrate is added to a solution containing ammonia, the equilibrium will shift…
A: In the aqueous solution, ammonia NH3 remains as ammonium hydroxide (NH4OH) which dissociates…
Q: Provide the correct IUPAC name for the compound shown here. 6- 3- 4- 5- 7-2- cyclo tri di sec- tert-…
A:
Q: Draw the two possible enols that can be formed from 3-methyl-2-butanone:
A: In order to show keto-enol tautomerism, there should be at least one alpha hydrogen present in the…
Q: 1. The function of proteins in the body is/are a. energy b. signaling c. structure d. a thru c are…
A: Since you asked multiple questions so as per Q&A guidelines of portal I solved first question…
Q: A(s) + B(g) ⇌ C(g) + D(g) ΔGº = 2.7 kJ. If [B] - 0.50M and [C] = 0.30M, what must [D] be in order…
A:
Q: For each of the following molecules, predict two possible products that can be formed by reaction…
A: In the question NaCN react with α, β unsaturated carbonyl compound and α, β unsaturated eminesNaCN…
Q: What sequence of reaction can be used to accomplish the following synthesis? H3C CH3 ОН ? H3C Н CH3
A: To obtain Aldehyde from the Carboxylic acid, we have to treat it with a reducing agent. The reducing…
Q: 1A. Why do electrophilic aromatic substitution reactions require a very reactive electrophile to…
A: The given question has 2 parts and both the parts can be answered if we consider the stability of…
Q: A chemist measures the energy change AH during the following reaction: CH₂(g) +20₂(g) →…
A:
Q: Which of the following correctly represents the electron affinity of bromine? O Br(g)→Br+ (g) + e¯ O…
A: We need to tell among the given options which one correctly represents the electron affinity of…
Q: 2A. What is the theoretical yield and limiting reagent for the Friedel Crafts acylation reaction of…
A: IntroductionBy following this separation scheme, one can effectively isolate 4-methyl acetophenone…
Q: How many valence electrons are in Ar²+? 2+
A:
Q: What is the common name of the following compound? CH3 CH3CH₂CHOCH3
A: Common name of organic compounds are derived by regional name of organic compounds.
Q: Consider the following three compounds: bicyclo[3.1.1]heptane, bicyclo[2.2.1]heptane and…
A: We will draw the structure of all the given compounds here and will observe if there is any double…
Q: An ideal gas is contained in a cylinder with a volume of 5.1*102 mL at a temperature of 30.°C and a…
A: The combined gas law combines the three gas laws: Boyle's Law, Charles's Law, and Gay-Lussac's Law.…
Q: Arrange the solutions in order of increasing acidity. Rank solutions from least acidity to greatest…
A:
Q: Which organic starting material is needed to complete the following reaction? H₂C B ? A 1)…
A: oxymercuration–demercuration reaction, analkene is treated with mercury(II) acetate, Hg(OAc)2, and…
Step by step
Solved in 3 steps with 4 images

- Which amomg the fragments below will be dectected by mass Spectrophotometer? [CH3CH3]+ CH3CH3 •CH2CH3 [CH3CH4]- only ii i and iii only i ii and iv only iiI am talking about this one on the ir spectroscopy we have carbon carbon double bond on benzen the three double bond so if we have that cant we say we have to csp2-H on the graph around 3100cm-1 thank uIdentify all the peaks from the IR spectrum. Be sure to list the cm-1 and the bond that corresponds to each peak.
- Pls label IR spectrum for molecule Benzil. If there might be any impurities spotted please tell meA student identified an organic unknown based on the IR spectrum as an aliphatic carboxylic acid. Explain what signal on a mass spectrum will also confirm that the unknown organic compound is a carboxylic acid.Q1. Why is it common to use CCl4 as a solvent in IR spectroscopy? Q3. After completion of the previous reaction, you end up seeing the following peaks: 3300 cm-1 (broad intense peak), 3060-3040 cm-1 (2 medium intensity peaks), 3030-3020-3010 cm-1 (3 small peaks), 1640 cm-1 (low intensity peak), 1585 cm-1 (medium intensity peak), 1410 cm-1 (medium intensity peak), 1100 cm-1 (strong intensity peak), 1010 cm-1 (medium intensity peak), 900-690 (many peaks, medium intensities). Give the molecular structure of the actual final product for the previous reaction. (Hint: it is not a carboxylic acid, nor the ketone previously drawn.) Q4. You want to take the IR spectrum of acetylene. How would you prepare your sample? Mention all important details of the sample preparation (no need to describe the acquisition).
- Which amomg the fragments below will be dectected by mass Spectrophotometer? [CH3CH3]+ CH3CH3 •CH2CH3 [CH3CH4]-Please fully annotate this IR spectrum in the picture below, no need to annotate the fingerprint region (400-1500 cm-1). Thank you so much!!Calculate the IHD and identify the important peaks in the following MS spectral data and draw the structure of the important peaks in the following MS spectral data.
- 1. Write the IR spectra to the corresponding structures below and cite the applicable peaks A. Ethyl acetate B. Cyclohexanone C. Propionic acidA low-intensity peak at ?/? = 31 can be seen in the mass spectrum of methyl acetate in Figure 3. Propose a fragment that could originate from methyl acetate and be responsible for this peak.(A.) Name the (non-alkane) functionalgroup in the following compound. see attatched photo for structure. (B.) Write at least two IR peaks (frequencyranges, in cm-1) you would expect to find for thecompound in question (A.).

