Q: Calculate ?H° for each reaction.a. HO• + CH4 -> •CH3 + H2Ob. CH3OH + HBr -> CH3Br + H2O
A: The chemical equation for the given chemical reaction is shown in equation (1).
Q: Select the major product of the following reaction. i. ii. HO2C. HO,C, -CO2H CO2H MeO2C. H3O*…
A: In this question, we want Identify the major Product. How we can identify, I will give details…
Q: Given the reaction P,O10 (s) + H2O → H3 PO4 (aq) ΔΗ -96 %3D .2 kJ what the AH for P4O10 (s) + 6 H2O…
A:
Q: NH4Cl + CaSO4 → (NH4)2SO4 + CaCl2 Δ Hf NH4Cl = -311.730 Δ Hf CaSO4 = -1434.5 kJ/mol Δ Hf…
A:
Q: Calculate ∆S° for the following reactions in J K-1 a. 2 C2H6(g) + 7 O2(g) → 4 CO2(g) + 6 H2O(g) b.…
A: The standard molar entropy of the compounds in reaction (a) can be listed as follows: ∆Soethane =…
Q: LIOH H,0 Reaction D
A:
Q: CH
A: The given reaction can not be achieved directly by Friedel Craft Alkylation due to carbocation…
Q: 15 Question: Complete the following reaction scheme. A H B H,O. H HBr. H,O2 Br.. H.O conc. H,SO4…
A:
Q: Given the following: NAOH (aq) + HCI (aq) --> H20 (1) + NaCI (aq) DHreaction = -55.83 kJ/mol %3D…
A:
Q: The following reaction will give a number of products. Choose from the list some of the likely…
A: ANS. option b.
Q: JA H;º of H2O(1) is -285.8 kJ/mol. calculate A H° of MgO. The relevant reactions for these two heats…
A: Heat of formation: For the heat of formation of compound from its stable elemental state, the…
Q: Predict the product(s): ö: HÖ HNO, H,SO,
A: The electron-withdrawing group attached to the ring such as -COOH, -NO2, X, etc. direct the incoming…
Q: 4. From the enthalpies of reaction H2 (g) + F2 (9) → 2 HF(g) AH = -537 kJ C (s) + 2 F2 (9) - 2 C (s)…
A:
Q: Consider the attached two-step reaction: Question: How many bonds are broken and formed in Step [1]?…
A: In a chemical reaction, the bonds between atoms may either break or form. This process either absorb…
Q: a.Which value corresponds to a negative value of ΔGo: Keq = 10−2 or Keq = 102? b.In a unimolecular…
A: (a) Greater the equilibrium constant value the concentration of product formation is high. An…
Q: For which one of the following reactions the value of ΔH°rxn to ΔH°f for the product? N2(g)+O2(g)—>…
A: Answer First option. N2(g) + O2(g) → 2NO(g)
Q: Calculate the heat of reaction deltaH for the following reaction: 2CH4(g)+3O2(g)——>2CO(g)+ 4H2O(g)…
A: The reaction given is, => 2 CH4 (g) + 3 O2 (g) → 2 CO (g) + 4 H2O (g)
Q: For which of the attached reactions is ΔSo a positive value?
A:
Q: The H0 and S0 for the reaction N2O4 N2 + 2O2?
A: H0 and S0 for the reaction-- N2O4 -----> N2 + 2O2
Q: 6.) Predict the product of the following chemical reactions and state where the product (waste) sk…
A:
Q: Complete the following reactions: a. CH,-C-0-C-CH,+ CH,-OH -→ | CH, — ОН | b. CH,CH— С-ОН + H*, heat…
A: To complete the following reaction.,
Q: An important step in the synthesis of nitric acid is the conversion of ammonia to nitric oxide.…
A: The enthalpy change of the given reaction can be calculated using the expression: ∆H°rxn =…
Q: Calculate ΔH° for each reaction. a. HO• + CH4 → •CH3 + H2O b. CH3OH + HBr → CH3Br + H2O
A: The given reaction is: ∆H˚ for this reaction can be calculated as follows: Putting the values in…
Q: Consider the following reaction. H. H,SO, H. H-O c=C H,C CH3
A: Hydration of alkene: Alkene gives hydration reactions in presence of an acid catalyst. In this…
Q: Which of the following is NOT rearrangement reaction? for CH, CH, CH3 H,C CH3 H,C CH, + CH, CH, CH,…
A:
Q: At -29086 °C, Keq = 6.46 for the reaction: 1/2 N2(9) +1/2 02(9)NO2(g) (a) What is the value of Keg…
A: Given, Temperature = -29086◦c keq = 6.46
Q: -NO2 H3C-C-C-CH3. Magnesium mono peroxy phthalate (MMPP) O2N- -CH=CH-COOH CH,COONA/CH, COOH
A: The magnesium monoperoxypthalate (MMPP) is a peroxy acid used as oxidizing agent in organic…
Q: Calculate the heat of reaction deltaH for the following reaction: H2(g)+Cl2(g)——2HCl(g) Round your…
A:
Q: 1) Complote the following reactions CH;CH;O° OH" CH SNe CH;CH;OH LIAID. excess heat HO. OH
A: Hello. There are multiple subparts in the question. The first three subparts have been solved. To…
Q: It the 8pesitic heat of methanolis 8,51j/k-g how many joules are peguired t0a/se the temperature of…
A:
Q: 1. Using the bond energies on Handout 8, estimate the heat of reaction for each of the following:…
A: Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: 10. For which one of the following reactions is the value of ΔH0rxn equal to ΔH0f for the product?…
A: For a reaction to have equal standard enthalpies of reaction and formation, it must satisfy the…
Q: Which of the following is an energy diagram for a three-step reaction? reaction coordinate reaction…
A: 20) To select an option where the value of ΔG is -ve.
Q: For which of the following reactions is ΔS° a positive value?
A:
Q: Q7. Fill in the missing intermediates, products and reagents in the reaction scheme shown below. 1.…
A: Concept 1) Hydroboration reaction involved formation of less substituted alcohol. 2) addition…
Q: Reaction AH at 298 K 3 Y + 2 X2O → 2 X2Y + YO2 146.9 kJ Y + O2 → YO2 - 296.4 kJ O -149.5 kJ O 443.3…
A:
Q: Calculate ΔHo for each attached reaction.
A: GIVEN: Reaction: To solve: ΔHo for each reaction= ?
Q: Et Et Me. LEt Et Me and Me H. H Me
A:
Q: 18) How Namy isan d ara posible for Cu Ha F? C-C-C-C- -C-C-C; -C-C-C-C 1
A: Isomers are molecules that have the same molecular formula, but have a different arrangement of the…
Q: MAC=350b7672c69b4a6f9ade516b8d521100#1 Part A Give the major product for the following reaction.…
A:
Q: If a given reaction is A + B --> 2C with ΔH = -8.8 lJ and the needed reaction is 2A + 2B -->…
A: Given:When 1 mole of A react with 1 mole of B, then ΔH = -8.8 KJ
Q: O3(g) ⇌ O2(g) + O(g) To which direction will the reaction shift if additional O3 is added?
A:
Q: Which of the following organic equations has the highest heat of reaction AH° ? CH3CH2- Br + H20…
A: Since you have asked multiple question, we will solve the first question for you. If you want any…
Q: Given the product/s of reaction, provide the reactants а. оо H--C-H b. -CH;Br + HBr с. CH,CH, Br
A: The reactants for above transformation is given below
Q: The reaction below is: * Cl2 hy 2 :Cl•
A: The given reaction is a free radical reaction where the reaction proceeds in the following three…
Q: Estimate the temperature at which (a) CaCO3 decomposes spontaneouslyand (b) CuSO4.5H2O undergoes…
A: Decomposition occurs when a complex molecule of organic as well as inorganic matter is decomposed…
Q: O3(g) ⇌ O2(g) + O(g) To which direction will the reaction shift if additional O2 is added?
A: Le Chatelier's principle: This principle is used to predict the effect of a change in conditions on…
Q: stratosphere converts UV light energy into heat. Which of these reactions release heat? (A) O3 +…
A: The oxygen atom which is produced in the ist reaction that is A when combines with oxygen molecule…
Q: Give the major product(s) of the following reaction. Cl2 (1 mole) OMe heat OMe CI CI OMe OMe CI CI…
A: The methoxy group, OCH3, is ortho-para directing group and thus the formation of meta substituted…
Q: Heat (c) ? Heat (d)
A:
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

- 2H2S(g) + SO2(g) <--> 3S(s) + 2H2O(g) Would this reaction be favored at a high or low temperature?For each reaction, decide whether substitution or elimination (or both) is possible, andpredict the products you expect. Label the major products. chlorocyclohexane + NaOC(CH3)3 in (CH3)3COHDraw the structure(s) of the major organic product(s) of the following reaction. NH₂ O₂N NaNO₂ aqueous HCI at 0°
- Draw the intermediate carbocation that is formed when each is protonated, say, with H+.These occur when adding HBr, HCl, HI, or H+/H2O to a double bond. The rich get richer. Will it rearrange?Questão 10A certain hydrocarbon had the molecular formula C16H26 and contained two triple bonds. Ozonolysis resulted in CH3(CH2)4CO2H and HO2CCH2CH2CO2H as the only products. What is the reasonable structure for this hydrocarbon? Hexadec-6,10-dino undec-1,5-dino Hept-1,5-dino hex-1,5-dino nah(CH3)3CHOH is reacted with CH3-O-C--C6H5 in a transesterification reaction. What is the M+ of the product? || O 102 80 178 None of these
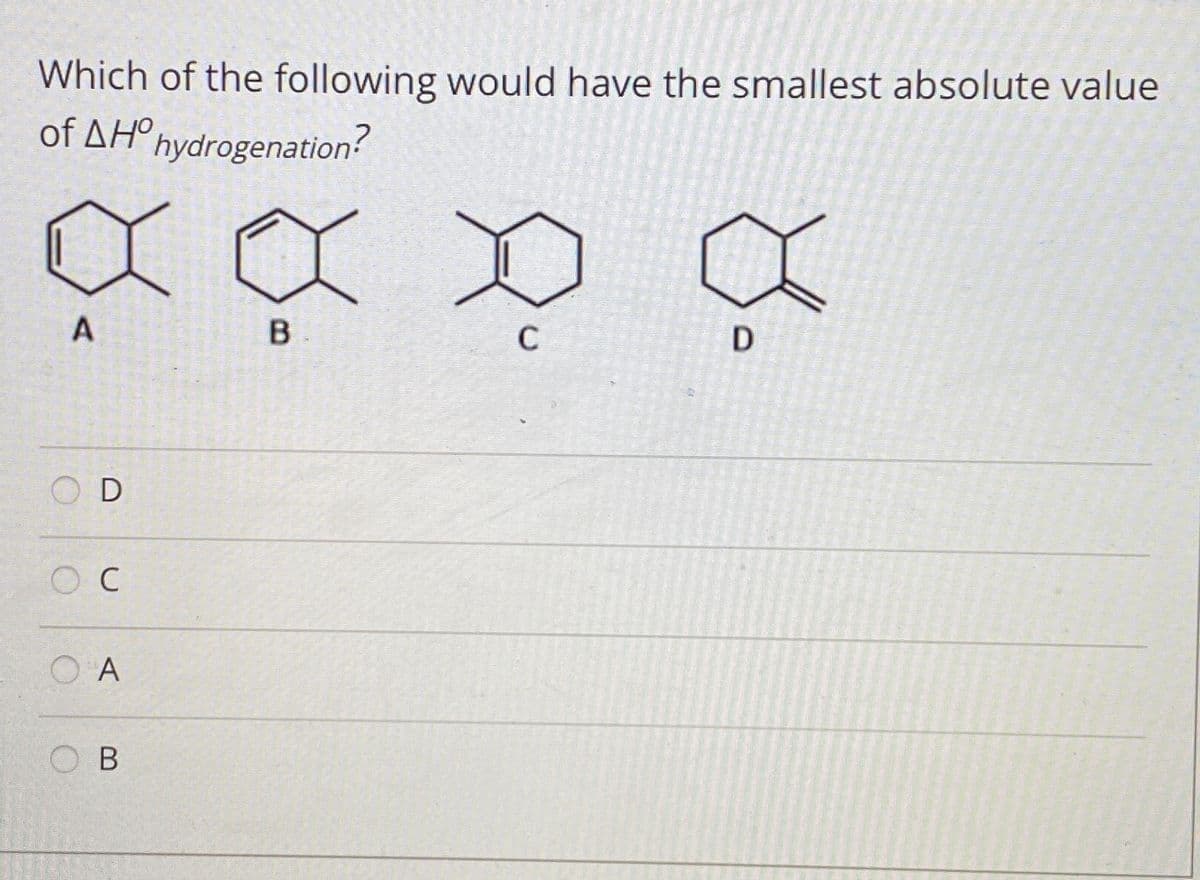
- Draw all resonance structures for the carbocation formed by ortho attack of the electrophile +NO2 on each starting material. Label any resonance structures that are especially stable or unstable.Match each alkene to its heat of hydrogenation.Alkenes: 3-methylbut-1-ene, 2-methylbut-1-ene, 2-methylbut-2-ene?H° (hydrogenation) kJ/mol: –119, –127, –112Q7 Consider the reaction, where the alkane shown is subjected to radical bromination at 25 °C. Br₂ light Describe the major monobromination product. product
- Which reaction corresponds to the Kb for HSO4− ? a. H2SO4 ⇌ H+ + HSO4− b. SO42− + H2O ⇌ OH− + HSO4− c. HSO4− + H2O ⇌ OH− + H2SO4 d. HSO4− ⇌ H+ + SO42− e. HSO4− + OH− ⇌ H2O + SO42−Q10. Which of the following is more stable?What are the major products obtained from the following reactions? Indicate the geometry (cis/trans)?



