Chapter3: Mechanisms
Section: Chapter Questions
Problem 93EQ
Related questions
Question
Which reaction is the fastest, which is the slowest?
I want to check my work and see if I am correct
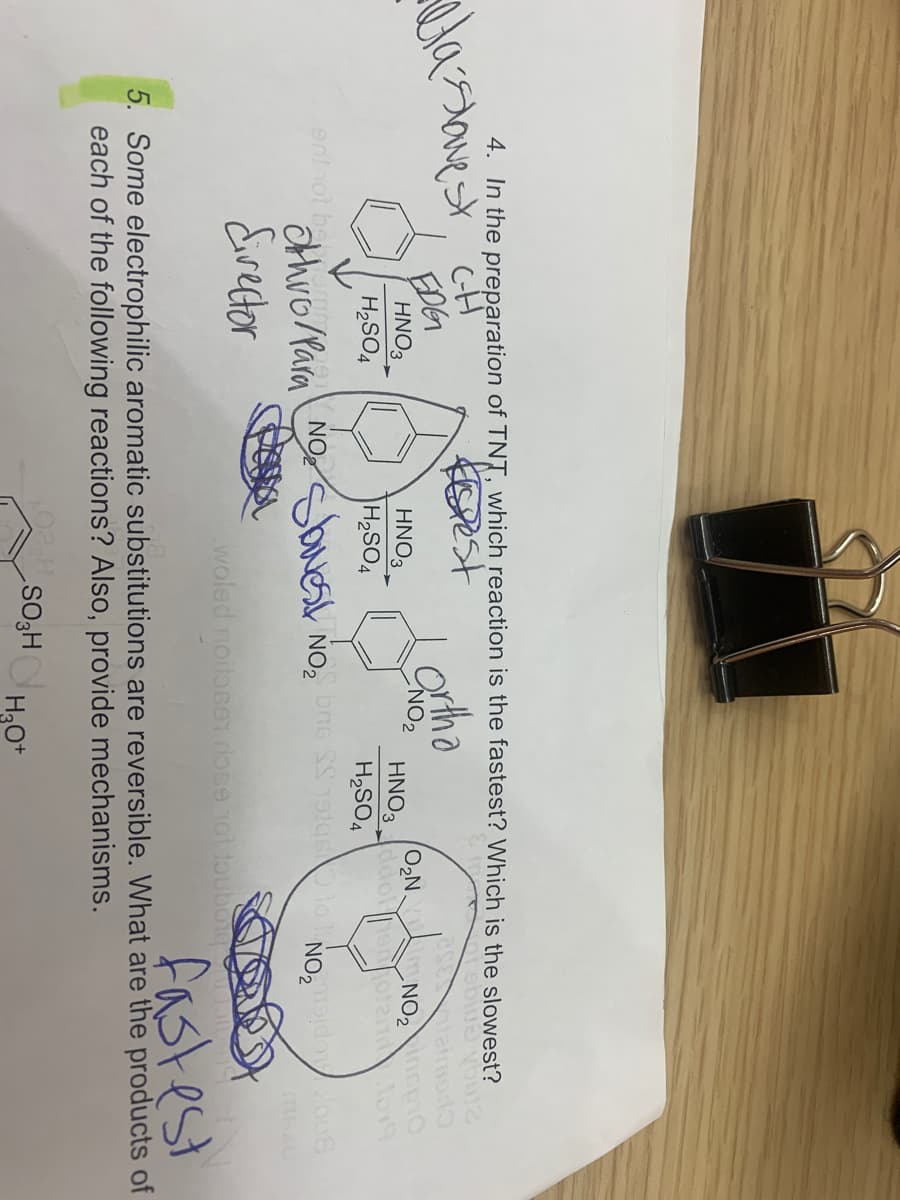
Transcribed Image Text:4. In the preparation of TNT, which reaction is the fastest? Which is the slowest?
Kest
EDG
HNO3
NO,
O2N.
NO2 0
HNO3
H2SO4
HNO3 do ooten o
H2SO4
H2SO4
othro/Para NObuest
Eirector
woled noilase dose tol loubo
fastest
5. Some electrophilic aromatic substitutions are reversible. What are the products of
each of the following reactions? Also, provide mechanisms.
SO3H H2O*
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Recommended textbooks for you



Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning



Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning