Which strong acid and which strong base would react in aqueous solution to produce the following salts? a. Sodium perchlorate, NaClO4 (Select all that apply.) Osodium carbonate sodium hydroxide Operchloric acid iron(II) perchlorate Cammonium perchlorate Ocobalt(II) perchlorate b. Sodium nitrate, NaNO3 (Select all that apply.) nitric acid sodium chloride Dammonium nitrate Diron(II) nitrate Osodium hydroxide Ocobalt(II) nitrate c. Potassium chloride, KC1 (Select all that apply.) Chydrochloric acid Opotassium carbonate Opotassium hydroxide Ocopper(II) chloride Dammonium chloride cobalt(II) chloride Show Hint Previous
Which strong acid and which strong base would react in aqueous solution to produce the following salts? a. Sodium perchlorate, NaClO4 (Select all that apply.) Osodium carbonate sodium hydroxide Operchloric acid iron(II) perchlorate Cammonium perchlorate Ocobalt(II) perchlorate b. Sodium nitrate, NaNO3 (Select all that apply.) nitric acid sodium chloride Dammonium nitrate Diron(II) nitrate Osodium hydroxide Ocobalt(II) nitrate c. Potassium chloride, KC1 (Select all that apply.) Chydrochloric acid Opotassium carbonate Opotassium hydroxide Ocopper(II) chloride Dammonium chloride cobalt(II) chloride Show Hint Previous
Principles of Modern Chemistry
8th Edition
ISBN:9781305079113
Author:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Chapter15: Acid–base Equilibria
Section: Chapter Questions
Problem 58P
Related questions
Question
100%
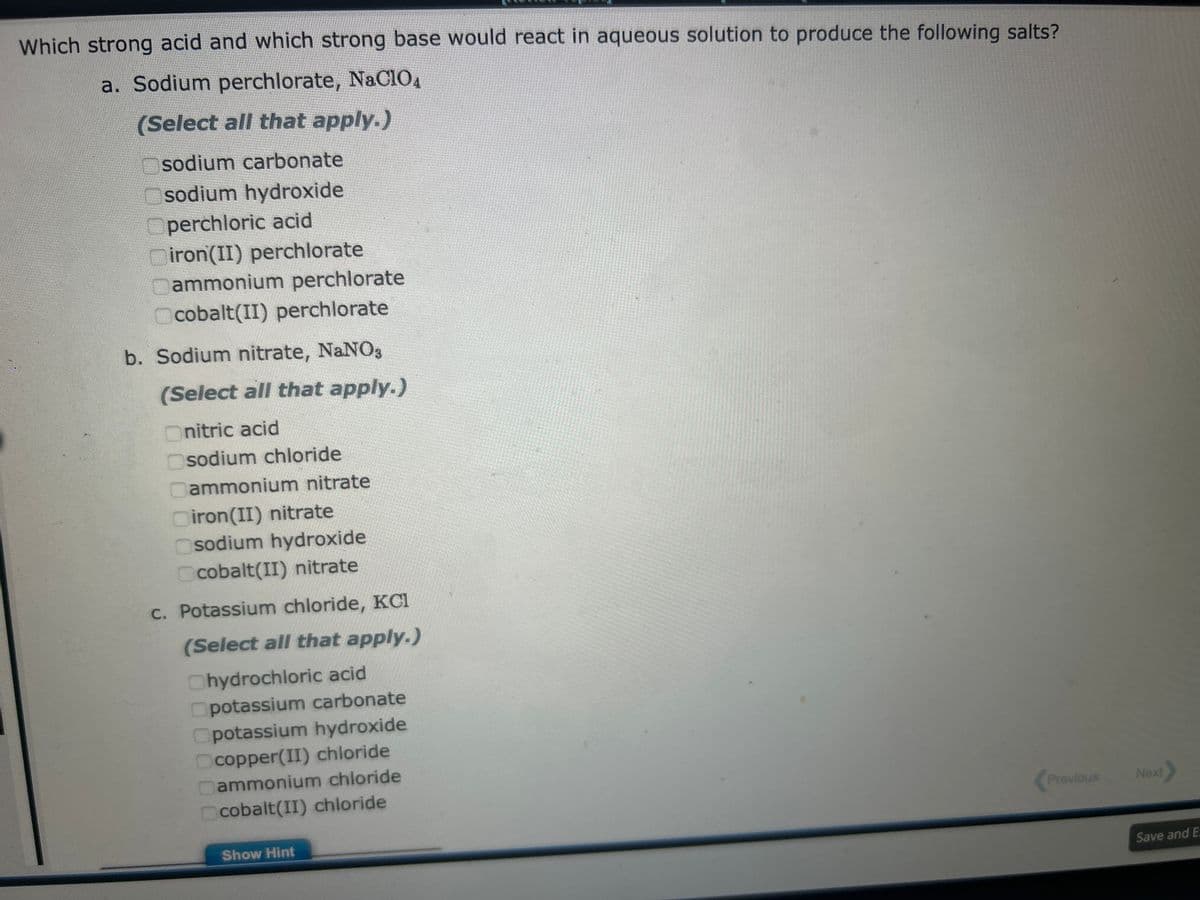
Transcribed Image Text:Which strong acid and which strong base would react in aqueous solution to produce the following salts?
a. Sodium perchlorate, NaC104
(Select all that apply.)
sodium carbonate
Osodium hydroxide
Operchloric acid
Diron(II) perchlorate
ammonium perchlorate
Ocobalt(II) perchlorate
b. Sodium nitrate, NaNO3
(Select all that apply.)
Onitric acid
sodium chloride
ammonium nitrate
iron(II) nitrate
Osodium hydroxide
Ocobalt(II) nitrate
c. Potassium chloride, KCI
(Select all that apply.)
Chydrochloric acid
potassium carbonate
Opotassium hydroxide
Ocopper(II) chloride
ammonium chloride
cobalt(II) chloride
Show Hint
Previous
Next>
Save and E
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 4 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning