* You can keep extra decimal places through most of this problem, but round your final estimate to one decimal place* You wish to quantify your favourite compound in an experimental solution using spectrophotometry. First, you use standard solutions of your favourite compound to collect the following standard curve data. Calculate the slope of the line of best fit. Standard curve data
* You can keep extra decimal places through most of this problem, but round your final estimate to one decimal place* You wish to quantify your favourite compound in an experimental solution using spectrophotometry. First, you use standard solutions of your favourite compound to collect the following standard curve data. Calculate the slope of the line of best fit. Standard curve data
Principles Of Radiographic Imaging: An Art And A Science
6th Edition
ISBN:9781337711067
Author:Richard R. Carlton, Arlene M. Adler, Vesna Balac
Publisher:Richard R. Carlton, Arlene M. Adler, Vesna Balac
Chapter36: Bone Densitometry
Section: Chapter Questions
Problem 4RQ
Related questions
Question
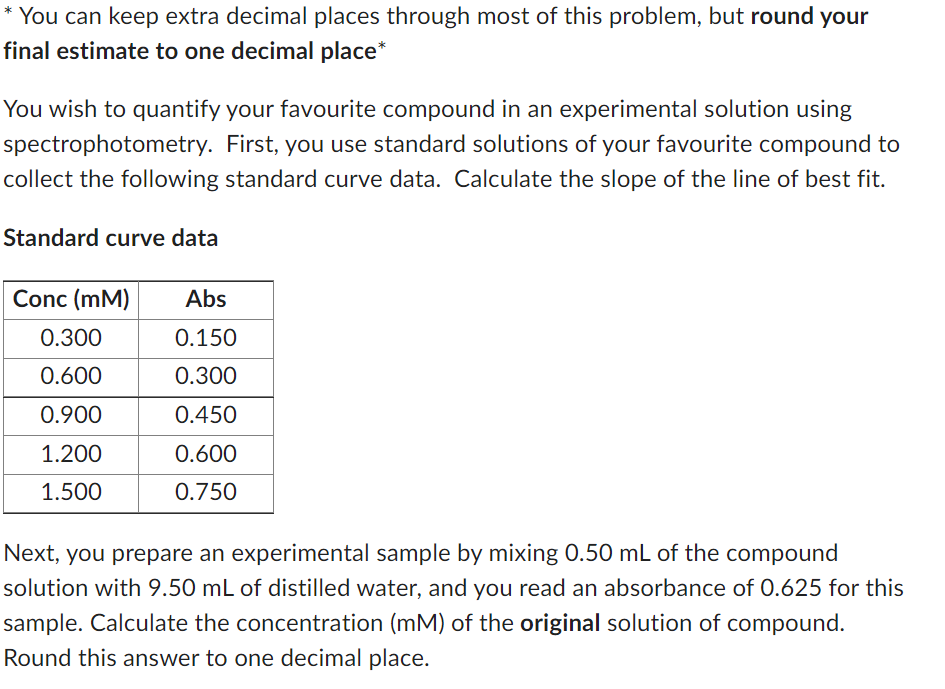
Transcribed Image Text:* You can keep extra decimal places through most of this problem, but round your
final estimate to one decimal place*
You wish to quantify your favourite compound in an experimental solution using
spectrophotometry. First, you use standard solutions of your favourite compound to
collect the following standard curve data. Calculate the slope of the line of best fit.
Standard curve data
Conc (mM)
0.300
0.600
0.900
1.200
1.500
Abs
0.150
0.300
0.450
0.600
0.750
Next, you prepare an experimental sample by mixing 0.50 mL of the compound
solution with 9.50 mL of distilled water, and you read an absorbance of 0.625 for this
sample. Calculate the concentration (mM) of the original solution of compound.
Round this answer to one decimal place.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

Recommended textbooks for you

Principles Of Radiographic Imaging: An Art And A …
Health & Nutrition
ISBN:
9781337711067
Author:
Richard R. Carlton, Arlene M. Adler, Vesna Balac
Publisher:
Cengage Learning

Principles Of Radiographic Imaging: An Art And A …
Health & Nutrition
ISBN:
9781337711067
Author:
Richard R. Carlton, Arlene M. Adler, Vesna Balac
Publisher:
Cengage Learning